
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 1
Title
Uncharacterized liver damage leading to hepatocellular carcinoma
Short name
Graphical Representation
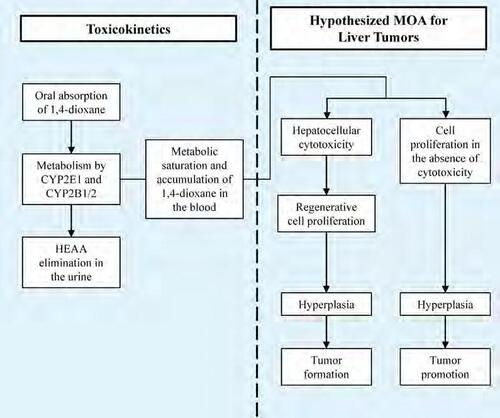
Point of Contact
Contributors
- Stephen Edwards
- Evgeniia Kazymova
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Hyperplasia, Hyperplasia | September 16, 2017 10:14 |
| Promotion, Hepatocelluar carcinoma | September 16, 2017 10:14 |
| Proliferation, Cell proliferation in the absence of cytotoxicity | September 16, 2017 10:14 |
| N/A, Unknown | September 16, 2017 10:14 |
| N/A, Unknown leads to Proliferation, Cell proliferation in the absence of cytotoxicity | December 03, 2016 16:37 |
| Proliferation, Cell proliferation in the absence of cytotoxicity leads to Hyperplasia, Hyperplasia | December 03, 2016 16:37 |
| Hyperplasia, Hyperplasia leads to Promotion, Hepatocelluar carcinoma | December 03, 2016 16:37 |
Abstract
1,4-Dioxane (also called dioxane) is a semi-volatile, colorless liquid with a faint sweet odor, produced in large amounts (1-10 million pounds in 1994, 1998, and 2002) in the United States.[1] For many years, it was primarily used as a stabilizer for 1,1,1-trichloroethane transport and storage, but that use is being phased out. It is also used as a solvent in the manufacture of household products such as detergents, soaps, lotions, shampoos, and cosmetics, in a variety of food manufacturing and food packaging processes, and is also used as a solvent in the manufacture of lacquers, paints, varnishes, waxes, resins, etc. 1,4-dioxane is water soluble and readily leaches into groundwater; thus,it has a high potential for entering the environment. Dioxane has affected groundwater supplies in many areas.
1,4-Dioxane has been classified by IARC as a Group 2N carcinogen, meaning that it is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity in experimental animals.[2] The United States Environmental Protection Agency classifies dioxane as a probable human carcinogen.[3] The compound has an LD50 of 5170 mg/kg.[4]
1,4-dioxane is absorbed rapidly following inhalation or oral exposure, with much less absorption occurring through skin contact. Toxicology data for exposure to 1,4-dioxane in humans is limited to a few inhalation exposure studies; no human data is available for oral or dermal exposures. The inhalation studies show that breathing 1,4-dioxane vapor for short periods of time causes irritation to the eyes, nose and throat, and that exposure to large amounts of 1,4-dioxane may cause kidney and liver damage. Accidental exposure of workers to high concentrations (unspecified) of 1,4-dioxane (via inhalation and dermal contact) have resulted in several deaths and the symptoms associated with those deaths suggest adverse nervous system effects and kidney toxicity. Studies in humans found no conclusive evidence for a causal link between occupational exposure to dioxane and increased risk for cancer; however, only two studies were available and these were limited by small cohort size and a small number of reported cancer cases.[5,6]
All exposure routes have been studied and described in animals. The majority of these studies have been subchronic and chronic studies of exposure to 1,4-dioxane administered in drinking water where oral exposure induced squamous cell carcinomas in the nasal turbinates and hepatocellular carcinomas in rats of both sexes, and increased the incidence of hepatocellular carcinomas in mice of both sexes. Liver and kidney toxicity were the primary noncancer health effects of subchronic and chronic oral exposure to 1,4-dioxane in animals.
There are only two subchronic inhalation studies and two chronic inhalation studies in animals. Inhalation exposure induced hepatocellular carcinomas in rats of both sexes. Liver and nasal toxicity are the primary noncancer health effects associated with inhalation exposure.
An oral reference dose (RfD) has been established at 0.03 mg/kg-day, with an overall confidence of medium. The oral cancer slope factor (CSF) is 0.10 (mg/kg-day)-1. The inhalation reference concentration (RfC) is 0.03 mg/m3, also with an overall confidence of medium. The IUR is 5 x 10-6 (g/m3 -1.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 294 | N/A, Unknown | N/A, Unknown |
| KE | 142 | Hyperplasia, Hyperplasia | Hyperplasia, Hyperplasia |
| KE | 57 | Proliferation, Cell proliferation in the absence of cytotoxicity | Proliferation, Cell proliferation in the absence of cytotoxicity |
| AO | 334 | Promotion, Hepatocelluar carcinoma | Promotion, Hepatocelluar carcinoma |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| rats | Rattus norvegicus | Moderate | NCBI |
Sex Applicability
Overall Assessment of the AOP
Consider the following criteria (may include references to KE Relationship pages): 1. concordance of dose-response relationships; 2. temporal concordance among the key events and adverse effect; 3. strength, consistency, and specificity of association of adverse effect and initiating event; 4. biological plausibility, coherence, and consistency of the experimental evidence; 5. alternative mechanisms that logically present themselves and the extent to which they may distract from the postulated AOP. It should be noted that alternative mechanisms of action, if supported, require a separate AOP; 6. uncertainties, inconsistencies and data gaps.
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
1. U.S. EPA (U.S. Environmental Protection Agency). (2002). Toxic Substances Control Act (TSCA) Inventory Update Database. Available online at http://www.epa.gov/iur/ (accessed February 22, 2010).
2. “Eleventh Report on Carcinogens” (http://ntp.niehs.nih.gov/ntp/roc/toc11.htm). United States Department of Health and Human Services’ National Toxicology Program. Retrieved 2 February 2006.
3. U.S. EPA (U.S. Environmental Protection Agency). (2005). Guidelines for carcinogen risk assessment [EPA Report]. (EPA/630/P-03/001F). Washington, DC: Risk Assessment Forum. http://www.epa.gov/cancerguidelines/
4. Surprenant, KS. (2002). Dioxane. In Ullmann's Encyclopedia of Industrial Chemistry (6th ed.). Weinheim, Germany: Wiley-VCH Verlag. http://dx.doi.org/10.1002/14356007.a08_545
5. Buffler, PA; Wood, SM; Suarez, L; Kilian, DJ. (1978). Mortality follow-up of workers exposed to 1,4-dioxane. J Occup Environ Med 20: 255-259.
6. Thiess, AM; Tress, E; Fleig, I. (1976). Arbeitsmedizinische Untersuchungsergebnisse von Dioxan-exponierten Mitarbeitern [Industrial-medical investigation results in the case of workers exposed to dioxane]. Arbeitsmedizin, Sozialmedizin, Umweltmedizin 11: 35-46.