
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 148
Title
EGFR Activation Leading to Decreased Lung Function
Short name
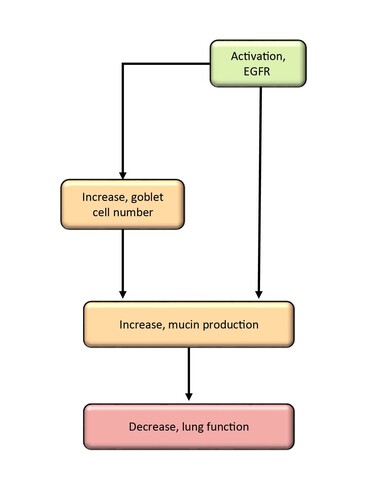
Graphical Representation
Point of Contact
Contributors
- Karsta Luettich
- Marja Talikka
- Hasmik Yepiskoposyan
- Cataia Ives
Coaches
- Rex FitzGerald
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.51 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Activation, EGFR | March 08, 2023 03:59 |
| Increase, Mucin production | March 09, 2023 01:55 |
| Decrease, Lung function | September 08, 2021 04:54 |
| Increase, goblet cell number | March 08, 2023 05:04 |
| Activation, EGFR leads to Increase, goblet cell number | March 16, 2023 04:09 |
| Activation, EGFR leads to Increase, Mucin production | March 09, 2023 05:26 |
| Increase, goblet cell number leads to Increase, Mucin production | March 17, 2023 09:30 |
| Increase, Mucin production leads to Decreased lung function | March 16, 2023 06:05 |
| Reactive oxygen species | August 15, 2017 10:43 |
Abstract
Increase in mucin production and consequent mucus hypersecretion in the airways are key attributes of many lung diseases, including asthma, cystic fibrosis and chronic bronchitis, all of which are characterized by decreased lung function (Yoshida and Tuder, 2007). Mucus hypersecretion is characterized by an increase in the number of goblet cells, mucin synthesis and mucus secretion which can result in airway obstruction and lung function decline (Kim and Criner, 2015, Yoshida and Tuder, 2007). Epidermal growth factor receptor (EGFR)-mediated signaling has been identified as the key pathway that leads to airway mucin production (Burgel and Nadel, 2008). This AOP for decreased lung function originates in EGFR activation in the airway epithelium. It describes the subsequent key events on the cellular and organ level that need to take place to culminate in the adverse outcome. The causal relationships in this AOP, including EGFR activation leading to increased number of mucin-producing goblet cells and to increased mucin production, are substantiated by multiple lines of evidence in studies performed using different model systems and approaches. Understanding how the inhaled toxicant-induced EGFR activation leads to pulmonary function impairment will be relevant to risk assessment of airborne pollutant exposure and how they contribute to the development and progression of the disease. Additionally, understanding the molecular underpinnings of these processes can aid in informing regulatory decision-making to assess the impact of inhalation toxicants on public health outcomes.
AOP Development Strategy
Context
This AOP delineates a sequence of key events initiating with stressor-induced activation of EGFR and resulting in decreased lung function through increased production of mucins. Excessive mucin production and consequent mucus hypersecretion are characteristic features of chronic diseases such as chronic obstructive pulmonary disease (COPD), cystic fibrosis, chronic bronchitis, and asthma, which pose a significant public health burden. Of note, exposure to cigarette smoke, occupational respiratory hazards, and air pollutants are clearly linked to the development of COPD, which is predicted to become the third leading cause of death worldwide by 2030 (Viegi et al., 2007, WHO, 2008). Mucus hypersecretion during the disease course can result in airway obstruction, decreased peak expiratory flow, respiratory muscle weakness, leading to decreased lung function (Kim and Criner, 2015, Yoshida and Tuder, 2007). Lung function decrease can have serious consequences and is associated with increased mortality (Panizza et al., 2006). This AOP is aimed to compile and organize the vast knowledge around molecular and cellular events and their relationships leading to lung function decrease with an overarching goal to facilitate the prediction and assessment of decreased pulmonary function. In vitro assays spanning from cell culture to organ system assays, with an aid of in silico methodology, all performed in human context, could be applied to measure each KE for inhaled toxicant assessments and adverse outcome predictions, and would contribute to eventual replacement of in-vivo tests in animals. This concept of hazard assessment and AO prediction aligns with integrated approach to testing and assessment (IATA) framework as a mechanistic support for regulatory decision-making (Clippinger et al., 2018).
Strategy
Chronic respiratory diseases such as COPD, cystic fibrosis, and asthma, characterized by increased mucin production and eventual lung function decrease, pose a substantial public health burden. EGFR-mediated signaling has been identified as the key pathway that leads to airway mucin production (Burgel and Nadel, 2004). Ligand binding in response to various toxicants and pathogens activates the EGFR tyrosine kinase on the surface of airway epithelial cells. Therefore, we determine the molecular initiating event (MIE) of this AOP the activation of EGFR which leads to adverse outcome (AO) of decreased lung function through key events (KE) increased mucin production and increased mucin-producing goblet cell numbers. In addition to increased mucin production, exposure-related decrease in lung function is also associated to ciliary function, the airway surface liquid height and mucociliary clearance efficiency, all of which are outlined and described in AOP411, AOP424, and AOP425.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 941 | Activation, EGFR | Activation, EGFR |
| KE | 2117 | Increase, goblet cell number | Increase, goblet cell number |
| KE | 962 | Increase, Mucin production | Increase, Mucin production |
| AO | 1250 | Decrease, Lung function | Decreased lung function |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Activation, EGFR leads to Increase, goblet cell number | adjacent | High | High |
| Activation, EGFR leads to Increase, Mucin production | adjacent | High | High |
| Increase, goblet cell number leads to Increase, Mucin production | adjacent | High | Moderate |
| Increase, Mucin production leads to Decreased lung function | adjacent | Moderate | Moderate |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult | High |
| Juvenile | Low |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific |
Overall Assessment of the AOP
Domain of Applicability
EGFR activation leading to increased mucin production and decreased lung function is predominantly studied in adults; however, it has been shown to also occur in pediatric asthma and bronchitis (Parker et al., 2015, Rogers, 2003). Nevertheless, the environmental exposures that induce EGFR activation and ultimately lead to lung function decline may apply more to adults who are more likely to be exposed to these stimulants over time (cigarette smoke, particulate matter).
The evidence presented here is derived from both human patient, cell culture and animal model biological systems. In vitro and in vivo studies in these systems have been performed to clarify the mechanisms of EGFR activation leading to mucus hyperproduction by studying the increase in goblet cells and upregulation in mucin transcript and protein expression. There are several clinical studies on mucus hypersecretion and how it affects lung function in humans with chronic bronchitis, asthma and other chronic lung diseases. The use of laboratory animals in human disease phenotype modelling enhances the understanding of disease mechanisms but also has limitations, e.g. due to anatomic differences between human and animal airways, differences in disease severity, difficulty of lung function measurements (Nikula and Green, 2000, Fricker et al., 2014). In summary, assembled data suggest that the KEs of this AOP are preserved across rodents and humans and there is good evidence supporting the occurrence of KERs in these species.
At times, clinical evidence linked to occupational exposures is derived from a majority of male subjects, which could be related to a male predominance in certain professions (Eng et al., 2011; Kennedy et al., 2007). Similarly, in most Western countries, cigarette smoking is still more prevalent in men than in women, although this gap has been closing steadily over the past decades (Syamlal et al., 2014; Hitchman and Fong, 2011). Nevertheless, the available in vivo and clinical evidence suggest that there is no remarkable gender difference.
Essentiality of the Key Events
MIE: EGFR activation
EGFR signaling is considered critical for mucin production (Vallath et al., 2014). Large amount of studies indicate that activation of EGFR through stressors and receptor ligands increase goblet cell numbers and mucin production while inhibition of EGFR decreases mucin production or goblet cell numbers (Barbier et al., 2012, Casalino-Matsuda et al., 2006, Choi et al., 2021, Deshmukh et al., 2008, Hao et al., 2014, Huang et al., 2017, Jia et al., 2021, Kato et al., 2022, Lee et al., 2000, Lee et al., 2011, Memon et al., 2020, Parker et al., 2015, Perrais et al., 2002, Shim et al., 2001, Song et al., 2016, Takeyama et al., 1999, Takeyama et al., 2001, Takeyama et al., 2008, Takezawa et al., 2016, Tyner et al., 2006b, Val et al., 2012, Wang et al., 2019, Yu et al., 2012a, Yu et al., 2012b). As for downstream AO, several studies indicate positive correlation between EGFR pathway activation and lung function decrease (Singanayagam et al., 2022, Feng et al., 2019, Lin et al., 2021). Taken together, and considering the strong causal link of EGFR activation on adjacent KEs, we propose high essentiality for the MIE “Activation, EGFR” in the AOP148.
KE: Increase, mucin production
Increased airway mucin production is a necessary condition for mucus hypersecretion. The stressor exposure maintenance and goblet cell number increase are important for sustained mucin production increase, otherwise the mucus hypersecretion resolves following re-establishment of airway homeostasis by anti-inflammatory mechanisms (Rose and Voynow, 2006). Mucus hypersecretion is a key feature of many lung diseases, including chronic obstructive pulmonary disease (COPD), asthma, cystic fibrosis and chronic bronchitis, all of which are characterized by decreased lung function. Evidences from literature indicate that sustained increased mucin production with consequent mucus hypersecretion correlate with lung function decrease (Caramori et al., 2009, Innes et al., 2006, Vestbo and Rasmussen, 1989, Vestbo et al., 1996, Ramos et al., 2014). Overall, increased mucin production is necessary but not always sufficient for leading to downstream events. Given the requirement of the KE for AO to occur we suggest high essentiality for the KE “Increase, mucin production” in the AOP148.
KE: Increase, goblet cell number
Goblet cells are specialized cells for mucin expression. Following stressor exposure, goblet cell numbers can increase which provides capacity to increase mucin production. Increased goblet cell numbers which can result from goblet cell hyperplasia and/or metaplasia sustain airway mucin overproduction contributing to airway obstruction and consequent lung function decline (Rose and Voynow 2006). Several studies show positive correlation between increase in goblet cell number and decrease in lung fuction (Innes et al., 2006, Raju et al., 2016, Ma et al., 2005, Celly et al., 2006). Considering above-mentioned, we propose high essentiality for the KE “Increase, goblet cell number” in the AOP148.
Evidence Assessment
Biological Plausibility
KER: EGFR activation leads to Increase, mucin production
Large number of studies using EGFR-activating ligands and EGFR inhibitors consistently show causal link leading from EGFR activation to increased production of mucin proteins (Barbier et al., 2012, Casalino-Matsuda et al., 2006, Choi et al., 2021, Deshmukh et al., 2008, Hao et al., 2014, Huang et al., 2017, Jia et al., 2021, Kato et al., 2022, Lee et al., 2000, Lee et al., 2011, Liu et al., 2013, Memon et al., 2020, Parker et al., 2015, Perrais et al., 2002, Shim et al., 2001, Song et al., 2016, Takeyama et al., 1999, Takeyama et al., 2001, Takeyama et al., 2008, Takezawa et al., 2016, Val et al., 2012, Wang et al., 2019, Yu et al., 2012a, Yu et al., 2012b). EGFR activation as a leading pathway for increased mucin production has broad acceptance in the scientific community and has been discussed also in review articles (Burgel and Nadel, 2004, Lai and Rogers, 2010). Therefore, we propose high biological plausibility for this KER.
KER: EGFR activation leads to Increase, goblet cell number
EGFR ligands and variety of stressors such as oxidative stress, cigarette smoke, allergens, viruses and bacterial endotoxins increase goblet cell number in an EGFR-dependent manner (Casalino-Matsuda et al., 2006, Gu et al., 2008, Hirota et al., 2012, Jia et al., 2021, Parker et al., 2015, Shatos et al., 2008, Song et al., 2016, Takeyama et al., 1999, Takeyama et al., 2001, Takezawa et al., 2016, Tyner et al., 2006a). Given the strong empirical evidence for involvement of EGFR in regulating the number of goblet cells and high reproducibility demonstrated in both in vitro and in vivo studies, we suggest high biological plausibility for this KER.
KER: Increase, goblet cell number leads to Increase, mucin production
Goblet cells are specialized cells for mucin production. The increase in the number of goblet cells is needed to accommodate the increased need for mucin production indicating that this KER is an inferred relationship, i.e. the occurrence of the downstream KE is inferred from the fact of occurrence of the upstream KE. Many studies demonstrate the correlation between increase in goblet cell numbers and mucin production (Zuhdi Alimam et al., 2000, Takezawa et al., 2016, Hao et al., 2012, Innes et al., 2006, Liang et al., 2017, Lee et al., 2000, Casalino-Matsuda et al., 2006, Tyner et al., 2006b), in fact the accepted measure of goblet cell number increase is the enhanced staining for mucus in the tissues. Thus, we judge the KER as highly plausible.
KER: Increase, mucin production leads to Decrease, lung function
Increased mucus production and hypersecretion is a physiological response to harmful exposures. This response is typically of short duration and does not pose a major problem to normal lung function. However, in the presence of sustained mucus production and secretion, maintained and promoted through increased number of mucin producing goblet cells, airways can become obstructed and result in lung function decline. In addition, impaired mucociliary clearance contributes to airway obstruction (Whitsett, 2018) and it is currently unclear whether chronic mucus hypersecretion alone is sufficient to elicit a decrease in lung function. Clinical studies and model animal research showed that MUC5AC production was inversely correlated with parameters of lung function (FEV1 (% predicted), FEV1/FVC ratio, inspiratory capacity) (Caramori et al., 2009, Innes et al., 2006, Raju et al., 2016), and epidemiological evidence indicates a link between mucus hypersecretion and decreased lung function (Allinson et al., 2016, Pistelli et al., 2003, Vestbo et al., 1996). As a cause-effect relationship cannot be conclusively proven, we suggest moderate biological plausibility for this KER.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
| Mucociliary clearance (MCC) | Impaired MCC contributes to decreased lung function (AOPs 411, 424, 425) | Increase, mucin production leads to Decrease, lung function |
Quantitative Understanding
There is good quantitative understanding of how EGFR signaling influences mucus production and goblet cell number increase. In the majority of these studies, the summary evidence indicates dose-response relationships, time-response relationships, and causality for EGFR activation leading adjacent downstream KEs, lending strong support for these KERs. However, data for increased mucin production and mucus hypersecretion leading to lung function decline at the organism level are mainly derived from surrogate measures, and while those may not adequately reflect quantitative mucus production, they are accepted in the clinical community as an indicator of lung diseases, such as COPD, chronic bronchitis and asthma.
Considerations for Potential Applications of the AOP (optional)
The future application of this AOP lies in its potential for predicting decreased lung function in humans exposed to potentially harmful inhaled substances. This becomes especially pertinent as impaired lung function carries a significant risk of morbidity and mortality. Owing to the long latency period between exposure and detectable decreases in lung function for most environmental pollutants, together with the fact that lung function tests alone may not be sufficiently sensitive to account for early lung damage that remains asymptomatic, means for early identification of potentially hazardous exposures are critical for the development of appropriate public health interventions. The AOP could provide a framework for mapping out suitable in vitro models and tests for evaluation of distinct KEs in different exposure contexts thus contributing to eventual replacement of in-vivo tests in animals. The predictive power of AOP aligns well with IATA framework to integrate diverse sources of information as a mechanistic support on chemical hazard characterization.
References
ALLINSON, J. P., HARDY, R., DONALDSON, G. C., SHAHEEN, S. O., KUH, D. & WEDZICHA, J. A. 2016. The Presence of Chronic Mucus Hypersecretion across Adult Life in Relation to Chronic Obstructive Pulmonary Disease Development. Am J Respir Crit Care Med, 193, 662-72.
BARBIER, D., GARCIA-VERDUGO, I., POTHLICHET, J., KHAZEN, R., DESCAMPS, D., ROUSSEAU, K., THORNTON, D., SI-TAHAR, M., TOUQUI, L., CHIGNARD, M. & SALLENAVE, J. M. 2012. Influenza A induces the major secreted airway mucin MUC5AC in a protease-EGFR-extracellular regulated kinase-Sp1-dependent pathway. Am J Respir Cell Mol Biol, 47, 149-57.
BURGEL, P. & NADEL, J. 2004. Roles of epidermal growth factor receptor activation in epithelial cell repair and mucin production in airway epithelium. Thorax, 59, 992-996.
BURGEL, P.-R. & NADEL, J. A. 2008. Epidermal growth factor receptor-mediated innate immune responses and their roles in airway diseases. European Respiratory Journal, 32, 1068-1081.
CARAMORI, G., CASOLARI, P., DI GREGORIO, C., SAETTA, M., BARALDO, S., BOSCHETTO, P., ITO, K., FABBRI, L. M., BARNES, P. J., ADCOCK, I. M., CAVALLESCO, G., CHUNG, K. F. & PAPI, A. 2009. MUC5AC expression is increased in bronchial submucosal glands of stable COPD patients. Histopathology, 55, 321-31.
CASALINO-MATSUDA, S. M., MONZÓN, M. E. & FORTEZA, R. M. 2006. Epidermal growth factor receptor activation by epidermal growth factor mediates oxidant-induced goblet cell metaplasia in human airway epithelium. Am J Respir Cell Mol Biol, 34, 581-91.
CELLY, C. S., HOUSE, A., SEHRING, S. J., ZHANG, X. Y., JONES, H., HEY, J. A., EGAN, R. W. & CHAPMAN, R. W. 2006. Temporal profile of forced expiratory lung function in allergen-challenged Brown-Norway rats. Eur J Pharmacol, 540, 147-54.
CHOI, W., YANG, A. X., SIEVE, A., KUO, S. H., MUDALAGIRIYAPPA, S., VIESON, M., MADDOX, C. W., NANJAPPA, S. G. & LAU, G. W. 2021. Pulmonary Mycosis Drives Forkhead Box Protein A2 Degradation and Mucus Hypersecretion through Activation of the Spleen Tyrosine Kinase-Epidermal Growth Factor Receptor-AKT/Extracellular Signal-Regulated Kinase 1/2 Signaling. Am J Pathol, 191, 108-130.
CLIPPINGER, A. J., ALLEN, D., BEHRSING, H., BÉRUBÉ, K. A., BOLGER, M. B., CASEY, W., DELORME, M., GAÇA, M., GEHEN, S. C., GLOVER, K., HAYDEN, P., HINDERLITER, P., HOTCHKISS, J. A., ISKANDAR, A., KEYSER, B., LUETTICH, K., MA-HOCK, L., MAIONE, A. G., MAKENA, P., MELBOURNE, J., MILCHAK, L., NG, S. P., PAINI, A., PAGE, K., PATLEWICZ, G., PRIETO, P., RAABE, H., REINKE, E. N., ROPER, C., ROSE, J., SHARMA, M., SPOO, W., THORNE, P. S., WILSON, D. M. & JARABEK, A. M. 2018. Pathway-based predictive approaches for non-animal assessment of acute inhalation toxicity. Toxicol In Vitro, 52, 131-145.
DESHMUKH, H. S., SHAVER, C., CASE, L. M., DIETSCH, M., WESSELKAMPER, S. C., HARDIE, W. D., KORFHAGEN, T. R., CORRADI, M., NADEL, J. A., BORCHERS, M. T. & LEIKAUF, G. D. 2008. Acrolein-activated matrix metalloproteinase 9 contributes to persistent mucin production. Am J Respir Cell Mol Biol, 38, 446-54.
FENG, F., DU, J., MENG, Y., GUO, F. & FENG, C. 2019. Louqin Zhisou Decoction Inhibits Mucus Hypersecretion for Acute Exacerbation of Chronic Obstructive Pulmonary Disease Rats by Suppressing EGFR-PI3K-AKT Signaling Pathway and Restoring Th17/Treg Balance. Evid Based Complement Alternat Med, 2019, 6471815.
FRICKER, M., DEANE, A. & HANSBRO, P. M. 2014. Animal models of chronic obstructive pulmonary disease. Expert Opin Drug Discov, 9, 629-45.
GU, J., CHEN, L., SHATOS, M. A., RIOS, J. D., GULATI, A., HODGES, R. R. & DARTT, D. A. 2008. Presence of EGF growth factor ligands and their effects on cultured rat conjunctival goblet cell proliferation. Exp Eye Res, 86, 322-34.
HAO, Y., KUANG, Z., JING, J., MIAO, J., MEI, L. Y., LEE, R. J., KIM, S., CHOE, S., KRAUSE, D. C. & LAU, G. W. 2014. Mycoplasma pneumoniae modulates STAT3-STAT6/EGFR-FOXA2 signaling to induce overexpression of airway mucins. Infect Immun, 82, 5246-55.
HAO, Y., KUANG, Z., WALLING, B. E., BHATIA, S., SIVAGURU, M., CHEN, Y., GASKINS, H. R. & LAU, G. W. 2012. Pseudomonas aeruginosa pyocyanin causes airway goblet cell hyperplasia and metaplasia and mucus hypersecretion by inactivating the transcriptional factor FoxA2. Cell Microbiol, 14, 401-15.
HIROTA, N., RISSE, P. A., NOVALI, M., MCGOVERN, T., AL-ALWAN, L., MCCUAIG, S., PROUD, D., HAYDEN, P., HAMID, Q. & MARTIN, J. G. 2012. Histamine may induce airway remodeling through release of epidermal growth factor receptor ligands from bronchial epithelial cells. Faseb j, 26, 1704-16.
HUANG, L., PU, J., HE, F., LIAO, B., HAO, B., HONG, W., YE, X., CHEN, J., ZHAO, J., LIU, S., XU, J., LI, B. & RAN, P. 2017. Positive feedback of the amphiregulin-EGFR-ERK pathway mediates PM2.5 from wood smoke-induced MUC5AC expression in epithelial cells. Sci Rep, 7, 11084.
INNES, A. L., WOODRUFF, P. G., FERRANDO, R. E., DONNELLY, S., DOLGANOV, G. M., LAZARUS, S. C. & FAHY, J. V. 2006. Epithelial mucin stores are increased in the large airways of smokers with airflow obstruction. Chest, 130, 1102-8.
JIA, Z., BAO, K., WEI, P., YU, X., ZHANG, Y., WANG, X., WANG, X., YAO, L., LI, L., WU, P., YUAN, W., WANG, S., ZHENG, J., HUA, Y. & HONG, M. 2021. EGFR activation-induced decreases in claudin1 promote MUC5AC expression and exacerbate asthma in mice. Mucosal Immunol, 14, 125-134.
KATO, T., ASAKURA, T., EDWARDS, C. E., DANG, H., MIKAMI, Y., OKUDA, K., CHEN, G., SUN, L., GILMORE, R. C., HAWKINS, P., DE LA CRUZ, G., COOLEY, M. R., BAILEY, A. B., HEWITT, S. M., CHERTOW, D. S., BORCZUK, A. C., SALVATORE, S., MARTINEZ, F. J., THORNE, L. B., ASKIN, F. B., EHRE, C., RANDELL, S. H., O'NEAL, W. K., BARIC, R. S. & BOUCHER, R. C. 2022. Prevalence and Mechanisms of Mucus Accumulation in COVID-19 Lung Disease. Am J Respir Crit Care Med.
KIM, V. & CRINER, G. J. 2015. The chronic bronchitis phenotype in chronic obstructive pulmonary disease: features and implications. Current Opinions in Pulmonary Medicine, 21, 133-141.
LAI, H. Y. & ROGERS, D. F. 2010. Mucus hypersecretion in asthma: intracellular signalling pathways as targets for pharmacotherapy. Curr Opin Allergy Clin Immunol, 10, 67-76.
LEE, H. M., TAKEYAMA, K., DABBAGH, K., LAUSIER, J. A., UEKI, I. F. & NADEL, J. A. 2000. Agarose plug instillation causes goblet cell metaplasia by activating EGF receptors in rat airways. Am J Physiol Lung Cell Mol Physiol, 278, L185-92.
LEE, Y. C., OSLUND, K. L., THAI, P., VELICHKO, S., FUJISAWA, T., DUONG, T., DENISON, M. S. & WU, R. 2011. 2,3,7,8-Tetrachlorodibenzo-p-dioxin-induced MUC5AC expression: aryl hydrocarbon receptor-independent/EGFR/ERK/p38-dependent SP1-based transcription. Am J Respir Cell Mol Biol, 45, 270-6.
LIANG, Y., LIU, K. W. K., YEUNG, S. C., LI, X., IP, M. S. M. & MAK, J. C. W. 2017. (-)-Epigallocatechin-3-gallate Reduces Cigarette Smoke-Induced Airway Neutrophilic Inflammation and Mucin Hypersecretion in Rats. Front Pharmacol, 8, 618.
LIN, X. G., LI, W., XIANG, S. Y., WU, S. B., ZHANG, X. F., JIANG, C. W., LIU, Z. B. & CHEN, Y. N. 2021. [Electroacupuncture improves lung function by suppressing mucin-5AC mediated EGFR-p38MAPK signaling and inflammation reaction in chronic obstructive pulmonary disease rats]. Zhen Ci Yan Jiu, 46, 180-6.
LIU, Z., TIAN, F., FENG, X., HE, Y., JIANG, P., LI, J., GUO, F., ZHAO, X., CHANG, H. & WANG, S. 2013. LPS increases MUC5AC by TACE/TGF-α/EGFR pathway in human intrahepatic biliary epithelial cell. Biomed Res Int, 2013, 165715.
MA, R., WANG, Y., CHENG, G., ZHANG, H. Z., WAN, H. Y. & HUANG, S. G. 2005. MUC5AC expression up-regulation goblet cell hyperplasia in the airway of patients with chronic obstructive pulmonary disease. Chin Med Sci J, 20, 181-4.
MEMON, T. A., NGUYEN, N. D., BURRELL, K. L., SCOTT, A. F., ALMESTICA-ROBERTS, M., RAPP, E., DEERING-RICE, C. E. & REILLY, C. A. 2020. Wood Smoke Particles Stimulate MUC5AC Overproduction by Human Bronchial Epithelial Cells Through TRPA1 and EGFR Signaling. Toxicol Sci, 174, 278-290.
NIKULA, K. J. & GREEN, F. H. 2000. Animal models of chronic bronchitis and their relevance to studies of particle-induced disease. Inhal Toxicol, 12 Suppl 4, 123-53.
PANIZZA, J. A., JAMES, A. L., RYAN, G., DE KLERK, N. & FINUCANE, K. E. 2006. Mortality and airflow obstruction in asthma: a 17-year follow-up study. Intern Med J, 36, 773-80.
PARKER, J. C., DOUGLAS, I., BELL, J., COMER, D., BAILIE, K., SKIBINSKI, G., HEANEY, L. G. & SHIELDS, M. D. 2015. Epidermal Growth Factor Removal or Tyrphostin AG1478 Treatment Reduces Goblet Cells & Mucus Secretion of Epithelial Cells from Asthmatic Children Using the Air-Liquid Interface Model. PLoS One, 10, e0129546.
PERRAIS, M., PIGNY, P., COPIN, M. C., AUBERT, J. P. & VAN SEUNINGEN, I. 2002. Induction of MUC2 and MUC5AC mucins by factors of the epidermal growth factor (EGF) family is mediated by EGF receptor/Ras/Raf/extracellular signal-regulated kinase cascade and Sp1. J Biol Chem, 277, 32258-67.
PISTELLI, R., LANGE, P. & MILLER, D. L. 2003. Determinants of prognosis of COPD in the elderly: mucus hypersecretion, infections, cardiovascular comorbidity. Eur Respir J Suppl, 40, 10s-14s.
RAJU, S. V., KIM, H., BYZEK, S. A., TANG, L. P., TROMBLEY, J. E., JACKSON, P., RASMUSSEN, L., WELLS, J. M., LIBBY, E. F., DOHM, E., WINTER, L., SAMUEL, S. L., ZINN, K. R., BLALOCK, J. E., SCHOEB, T. R., DRANSFIELD, M. T. & ROWE, S. M. 2016. A ferret model of COPD-related chronic bronchitis. JCI Insight, 1, e87536.
RAMOS, F. L., KRAHNKE, J. S. & KIM, V. 2014. Clinical issues of mucus accumulation in COPD. Int J Chron Obstruct Pulmon Dis, 9, 139-50.
ROGERS, D. F. 2003. Pulmonary mucus: Pediatric perspective. Pediatr Pulmonol, 36, 178-88.
ROSE, M. C. & VOYNOW, J. A. 2006. Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol Rev, 86, 245-78.
SHATOS, M. A., GU, J., HODGES, R. R., LASHKARI, K. & DARTT, D. A. 2008. ERK/p44p42 mitogen-activated protein kinase mediates EGF-stimulated proliferation of conjunctival goblet cells in culture. Invest Ophthalmol Vis Sci, 49, 3351-9.
SHIM, J. J., DABBAGH, K., UEKI, I. F., DAO-PICK, T., BURGEL, P. R., TAKEYAMA, K., TAM, D. C. & NADEL, J. A. 2001. IL-13 induces mucin production by stimulating epidermal growth factor receptors and by activating neutrophils. Am J Physiol Lung Cell Mol Physiol, 280, L134-40.
SINGANAYAGAM, A., FOOTITT, J., MARCZYNSKI, M., RADICIONI, G., CROSS, M. T., FINNEY, L. J., TRUJILLO-TORRALBO, M. B., CALDERAZZO, M., ZHU, J., ANISCENKO, J., CLARKE, T. B., MOLYNEAUX, P. L., BARTLETT, N. W., MOFFATT, M. F., COOKSON, W. O., WEDZICHA, J., EVANS, C. M., BOUCHER, R. C., KESIMER, M., LIELEG, O., MALLIA, P. & JOHNSTON, S. L. 2022. Airway mucins promote immunopathology in virus-exacerbated chronic obstructive pulmonary disease. J Clin Invest, 132.
SONG, L., TANG, H., LIU, D., SONG, J., WU, Y., QU, S. & LI, Y. 2016. The Chronic and Short-Term Effects of Gefinitib on Airway Remodeling and Inflammation in a Mouse Model of Asthma. Cell Physiol Biochem, 38, 194-206.
TAKEYAMA, K., DABBAGH, K., LEE, H. M., AGUSTÍ, C., LAUSIER, J. A., UEKI, I. F., GRATTAN, K. M. & NADEL, J. A. 1999. Epidermal growth factor system regulates mucin production in airways. Proc Natl Acad Sci U S A, 96, 3081-6.
TAKEYAMA, K., JUNG, B., SHIM, J. J., BURGEL, P. R., DAO-PICK, T., UEKI, I. F., PROTIN, U., KROSCHEL, P. & NADEL, J. A. 2001. Activation of epidermal growth factor receptors is responsible for mucin synthesis induced by cigarette smoke. Am J Physiol Lung Cell Mol Physiol, 280, L165-72.
TAKEYAMA, K., TAMAOKI, J., KONDO, M., ISONO, K. & NAGAI, A. 2008. Role of epidermal growth factor receptor in maintaining airway goblet cell hyperplasia in rats sensitized to allergen. Clin Exp Allergy, 38, 857-65.
TAKEZAWA, K., OGAWA, T., SHIMIZU, S. & SHIMIZU, T. 2016. Epidermal growth factor receptor inhibitor AG1478 inhibits mucus hypersecretion in airway epithelium. Am J Rhinol Allergy, 30, 1-6.
TYNER, J. W., KIM, E. Y., IDE, K., PELLETIER, M. R., ROSWIT, W. T., MORTON, J. D., BATTAILE, J. T., PATEL, A. C., PATTERSON, G. A., CASTRO, M. & SPOOR, M. S. 2006a. Blocking airway mucous cell metaplasia by inhibiting EGFR antiapoptosis and IL-13 transdifferentiation signals. Journal of Clinical Investigation, 116, 309-321.
TYNER, J. W., KIM, E. Y., IDE, K., PELLETIER, M. R., ROSWIT, W. T., MORTON, J. D., BATTAILE, J. T., PATEL, A. C., PATTERSON, G. A., CASTRO, M., SPOOR, M. S., YOU, Y., BRODY, S. L. & HOLTZMAN, M. J. 2006b. Blocking airway mucous cell metaplasia by inhibiting EGFR antiapoptosis and IL-13 transdifferentiation signals. J Clin Invest, 116, 309-21.
VAL, S., BELADE, E., GEORGE, I., BOCZKOWSKI, J. & BAEZA-SQUIBAN, A. 2012. Fine PM induce airway MUC5AC expression through the autocrine effect of amphiregulin. Arch Toxicol, 86, 1851-9.
VALLATH, S., HYNDS, R. E., SUCCONY, L., JANES, S. M. & GIANGRECO, A. 2014. Targeting EGFR signalling in chronic lung disease: therapeutic challenges and opportunities. Eur Respir J, 44, 513-22.
VESTBO, J., PRESCOTT, E. & LANGE, P. 1996. Association of chronic mucus hypersecretion with FEV1 decline and chronic obstructive pulmonary disease morbidity. Copenhagen City Heart Study Group. Am J Respir Crit Care Med, 153, 1530-5.
VESTBO, J. & RASMUSSEN, F. V. 1989. Respiratory symptoms and FEV1 as predictors of hospitalization and medication in the following 12 years due to respiratory disease. Eur Respir J, 2, 710-5.
VIEGI, G., PISTELLI, F., SHERRILL, D. L., MAIO, S., BALDACCI, S. & CARROZZI, L. 2007. Definition, epidemiology and natural history of COPD. European Respiratory Journal, 30, 993-1013.
WANG, J., ZHU, M., WANG, L., CHEN, C. & SONG, Y. 2019. Amphiregulin potentiates airway inflammation and mucus hypersecretion induced by urban particulate matter via the EGFR-PI3Kα-AKT/ERK pathway. Cell Signal, 53, 122-131.
WHITSETT, J. A. 2018. Airway Epithelial Differentiation and Mucociliary Clearance. Ann Am Thorac Soc, 15, S143-s148.
WHO 2008. World health statistics World Health Organization.
YOSHIDA, T. & TUDER, R. M. 2007. Pathobiology of cigarette smoke-induced chronic obstructive pulmonary disease. Physiological Reviews 87, 1047-1082.
YU, H., LI, Q., KOLOSOV, V. P., PERELMAN, J. M. & ZHOU, X. 2012a. Regulation of cigarette smoke-mediated mucin expression by hypoxia-inducible factor-1α via epidermal growth factor receptor-mediated signaling pathways. J Appl Toxicol, 32, 282-92.
YU, H., ZHOU, X., WEN, S. & XIAO, Q. 2012b. Flagellin/TLR5 responses induce mucus hypersecretion by activating EGFR via an epithelial cell signaling cascades. Exp Cell Res, 318, 723-31.
ZUHDI ALIMAM, M., PIAZZA, F. M., SELBY, D. M., LETWIN, N., HUANG, L. & ROSE, M. C. 2000. Muc-5/5ac mucin messenger RNA and protein expression is a marker of goblet cell metaplasia in murine airways. Am J Respir Cell Mol Biol, 22, 253-60.