
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 279
Title
Microtubule interacting drugs lead to peripheral neuropathy
Short name
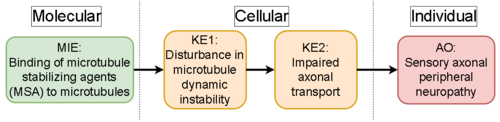
Graphical Representation
Point of Contact
Contributors
- Marvin Martens
- Arthur Author
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Binding of microtubule stabilizing agents (MSA) to microtubules | January 29, 2019 10:01 |
| Disturbance in microtubule dynamic instability | January 29, 2019 10:06 |
| Impaired axonial transport | January 29, 2019 10:07 |
| Sensory axonal peripheral neuropathy | January 29, 2019 10:08 |
| Binding of MSAs to microtubules leads to Disturbance in microtubule dynamic instability | January 29, 2019 10:10 |
| Disturbance in microtubule dynamic instability leads to Impaired axonial transport | January 29, 2019 10:11 |
| Impaired axonial transport leads to Sensory axonal peripheral neuropathy | January 29, 2019 10:12 |
Abstract
Peripheral neuropathy is regularly observed as a side-effect in microtubule-targeted chemotherapies. The length of their axons, together with the absence of protection by a blood brain barrier, renders peripheral neurons particularly vulnerable to disturbances in microtubule dynamics.
The present AOP was developed to summarize mechanistic data that support the link between the interaction of “microtubule stabilizing agents” with microtubules (MIE) and sensory axonal peripheral neuropathy (AO), via disturbances in microtubule dynamic instability (KE1) and an impaired axonal transport (KE2).
The present AOP allowed the identification of the following knowledge gaps in the literature: (i) experimental support for the mechanistic link of microtubule interacting drugs with the onset of sensory axonal peripheral neuropathy is generally limited by laborious and circumstantial access to peripheral neurons in vivo and by limited availability of human in vitro models of peripheral neurons; (ii) it is necessary to distinguish the biological consequences of microtubule-stabilizing drugs on the one side, and microtubule-destabilizing compounds on the other; (iii) the link of clinical symptoms to the action of microtubule stabilizers is complicated by incomplete toxicokinetic data, by the time offset between drug treatment and clinical symptoms, and by partial reversibility of the AO following washout of the microtubule-interacting drugs.
The present AOP was developed to contribute to cross systems testing as well as to experimental studies covering the testing of National Toxicology Program compounds.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1580 | Binding of microtubule stabilizing agents (MSA) to microtubules | Binding of MSAs to microtubules |
| KE | 1581 | Disturbance in microtubule dynamic instability | Disturbance in microtubule dynamic instability |
| KE | 1582 | Impaired axonial transport | Impaired axonial transport |
| AO | 1583 | Sensory axonal peripheral neuropathy | Sensory axonal peripheral neuropathy |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Binding of MSAs to microtubules leads to Disturbance in microtubule dynamic instability | adjacent | Not Specified | Not Specified |
| Disturbance in microtubule dynamic instability leads to Impaired axonial transport | adjacent | Not Specified | Not Specified |
| Impaired axonial transport leads to Sensory axonal peripheral neuropathy | adjacent | Not Specified | Not Specified |