
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 289
Title
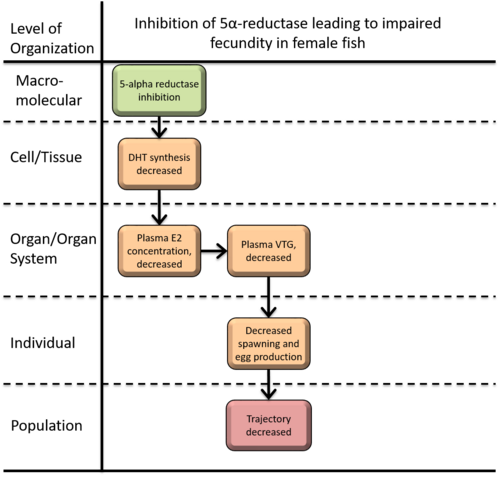
Inhibition of 5α-reductase leading to impaired fecundity in female fish
Short name
Graphical Representation
Point of Contact
Contributors
- Young Jun Kim
- Chang-Beom Park
- Cataia Ives
Coaches
- Dan Villeneuve
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.81 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition, 5α-reductase | April 05, 2024 08:23 |
| Decrease, dihydrotestosterone (DHT) level | April 05, 2024 08:10 |
| Reduction, Plasma 17beta-estradiol concentrations | September 26, 2017 11:30 |
| Reduction, Plasma vitellogenin concentrations | September 16, 2017 10:14 |
| Reduction, Cumulative fecundity and spawning | March 20, 2017 17:52 |
| Decrease, Population growth rate | January 03, 2023 09:09 |
| Inhibition, 5α-reductase leads to Decrease, DHT level | April 05, 2024 08:40 |
| Decrease, DHT level leads to Reduction, Plasma 17beta-estradiol concentrations | April 18, 2019 19:55 |
| Reduction, Plasma 17beta-estradiol concentrations leads to Reduction, Plasma vitellogenin concentrations | October 18, 2018 11:02 |
| Reduction, Plasma vitellogenin concentrations leads to Reduction, Cumulative fecundity and spawning | September 18, 2018 20:55 |
| Reduction, Cumulative fecundity and spawning leads to Decrease, Population growth rate | March 20, 2017 13:49 |
| finasteride, dutasteride, epristeride | April 18, 2019 19:56 |
Abstract
This AOP is designed to detect changes in cumulative fecundity and spawning resulted from the inhibition of 5α-reductases by 5α-reductase inhibitors. 5α-reductase catalyzes the conversion of a 3-oxo-5α-steroid to a 3-oxo-Δ4-steroid. Major reaction is the conversion of testosterone to 5α-dihydrotestosterone (DHT) which is a strong endogenous androgen receptor agonist. Inhibition of 5α-reductase can be caused by chemical inhibitors such as finasteride, dutasteride, epristeride etc. 5α-reductase inhibition (KE 790), the MIE for this AOP, results in decreasing levels of DHT and possibly 3β-androstanediol (3β-diol, the agonist of estrogen receptor β), metabolite of a DHT. Whereas inhibition of 5α-reductase leads to decrease in the level of 17β-estradiol (E2) (KE 219) in a female by the unknown mechanism, which corresponds to decreased egg production and spawning.
AOP Development Strategy
Context
The possible KE is the inhibition of 5α-reductase affects the level of the other endogenous substrate steroids such as androstenedione, progesterone, cortisol, and aldosterone. The physiological responses of the reduction of these steroids and the inhibition of 5α-reductase are not fully understood (Azzouni et al. 2012).
Acknowledgements: This research was supported by the National Research Council of Science & Technology(NST) grant by the Korea government (MSIP) (No. CAP-17-01-KIST Europe)
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1617 | Inhibition, 5α-reductase | Inhibition, 5α-reductase |
| KE | 1613 | Decrease, dihydrotestosterone (DHT) level | Decrease, DHT level |
| KE | 219 | Reduction, Plasma 17beta-estradiol concentrations | Reduction, Plasma 17beta-estradiol concentrations |
| KE | 221 | Reduction, Plasma vitellogenin concentrations | Reduction, Plasma vitellogenin concentrations |
| KE | 78 | Reduction, Cumulative fecundity and spawning | Reduction, Cumulative fecundity and spawning |
| AO | 360 | Decrease, Population growth rate | Decrease, Population growth rate |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| 3 to < 6 months | Moderate |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| fish | fish | Moderate | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | Moderate |
Overall Assessment of the AOP
Domain of Applicability
Chemical: Compounds which can inhibit 5α reductase in vitro, such as Flutamide, Dutasteride, and Finasteride, are relevant for this AOP
Sex: The AOP applies to female only.
Life stages: The relevant life stages for this AOP are reproductively mature adults.
Taxonomic: Teleost fish species.
Essentiality of the Key Events
The level of E2 is highly correlated with the synthesis of vitellogenin (VTG), supporting evidence for relationship 1386. Reduced VTG (KE 221) in fish has been used as an endpoint for adverse effects on fecundity and reproduction (Toxicol Sci, 2013. 132(2):284-297; Environ Toxicol Chem, 2016. 35(8): 2117-2224; Environ Toxicol, 2017. 32(7):1869-1877; Aquat Toxicol, 2018. 200:206-216).
Evidence Assessment
For the description of a non-adjacent KER linking 5a-reductase inhibition to decreased E2, There have been a few studies on the evaluation of the inhibition of 5α-reductase in fish (L.Mariotta-Calsaluci et al., 2013 Aquatic Toxicol.;García-García et al. 2017 J Steroid Biochem Mol Biol) and these studies did not clarify the mechanism of the inhibition of 5α-reductase to decrease 17β-estradiol (E2) in female fish. This information could be placed in the description of a non-adjacent KER linking 5a-reductase inhibition to decreased E2. Ornostay et al. (2016) reported DHT increased the level of E2 and steroidogenesis gene expression in fathead minnow ovary. Cumulative fecundity is the major endpoint for the evaluation of reproductive toxicity caused by endocrine disruption with the exposure to endocrine-disrupting chemicals (Ecotoxicol Environ Saf, 2018. 162:438-445; Environ Pollut, 2018. 240:403-411; J Appl Toxicol, 2018.38(4):544-551).
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
This AOP documents the scientifically credible links between inhibition of the enzyme 5areductase and reductions in cumulative fecundity in reproductively mature, female, teleost fish. Alteration of fecundity and spawning in fish is the critical apical endpoint for reproductive toxicity caused by endocrine disruption (e.g., OECD Test No. 229: Fish Short Term Reproduction Assay). This endpoint is essential and useful for screening of the potential endocrine disrupting chemicals and/or risk assessment sites contaminated by these chemicals. This AOP establishes that an in vitro assay that screens chemicals for their ability to inhibit 5a reductase is useful for identifying chemicals that may inhibit fish reproduction. It outlines several in vivo endpoints (e.g., reduction in circulating estrogen concentrations, reduction in circulating vitellogenin), that can be used to verify the probable reproductive outcomes without doing a completed OECD 229.
References
Ankley, G. T., M. D. Kahl, K. M. Jensen, M. W. Hornung, J. J. Korte, E. A. Makynen and R. L. Leino (2002). "Evaluation of the aromatase inhibitor fadrozole in a short-term reproduction assay with the fathead minnow (Pimephales promelas)." Toxicological sciences : an official journal of the Society of Toxicology 67(1): 121-130.
Baumann, L., H. Holbech, S. Keiter, K. L. Kinnberg, S. Knörr, T. Nagel and T. Braunbeck (2013). "The maturity index as a tool to facilitate the interpretation of changes in vitellogenin production and sex ratio in the Fish Sexual Development Test." Aquatic toxicology (Amsterdam, Netherlands) 128-129: 34-42.
Bowman, C. J., K. J. Kroll, M. J. Hemmer, L. C. Folmar and N. D. Denslow (2000). "Estrogen-induced vitellogenin mRNA and protein in sheepshead minnow (Cyprinodon variegatus)." General and comparative endocrinology 120(3): 300-313.
Diotel, N., J.-L. Do Rego, I. Anglade, C. Vaillant, E. Pellegrini, H. Vaudry and O. Kah (2011). "The brain of teleost fish, a source, and a target of sexual steroids." Frontiers in neuroscience 5: 137-137.
García-García, M., M. Sánchez-Hernández, M. P. García-Hernández, A. García-Ayala and E. Chaves-Pozo (2017). "Role of 5α-dihydrotestosterone in testicular development of gilthead seabream following finasteride administration." The Journal of steroid biochemistry and molecular biology 174: 48-55.
Jensen, K. M., M. D. Kahl, E. A. Makynen, J. J. Korte, R. L. Leino, B. C. Butterworth and G. T. Ankley (2004). "Characterization of responses to the antiandrogen flutamide in a short-term reproduction assay with the fathead minnow." Aquatic toxicology (Amsterdam, Netherlands) 70(2): 99-110.
Kim, B.-H., A. Takemura, S. J. Kim and Y.-D. Lee (2003). "Vitellogenin synthesis via androgens in primary cultures of tilapia hepatocytes." General and comparative endocrinology 132(2): 248-255.
Kwon, H. C., S. H. Choi, Y. U. Kim, S. O. Son and J. Y. Kwon (2005). "Androgen action on hepatic vitellogenin synthesis in the eel, Anguilla japonica is suppressed by an androgen receptor antagonist." The Journal of steroid biochemistry and molecular biology 96(2): 175-178.
Lee, M. R., J. R. Loux-Turner and K. Oliveira (2015). "Evaluation of the 5α-reductase inhibitor finasteride on reproduction and gonadal development in medaka, Oryzias latipes." General and comparative endocrinology 216: 64-76.
Margiotta-Casaluci, L., R. E. Hannah and J. P. Sumpter (2013). "Mode of action of human pharmaceuticals in fish: the effects of the 5-alpha-reductase inhibitor, dutasteride, on reproduction as a case study." Aquatic toxicology (Amsterdam, Netherlands) 128-129: 113-123.
Marlatt, V. L., B. P. Lo, A. Ornostay, N. S. Hogan, C. J. Kennedy, J. R. Elphick and C. J. Martyniuk (2013). "The effects of the urea-based herbicide linuron on reproductive endpoints in the fathead minnow (Pimephales promelas)." Comparative biochemistry and physiology. Toxicology & pharmacology : CBP 157(1): 24-32.
Martyniuk, C. J., S. Bissegger and V. S. Langlois (2013). "Current perspectives on the androgen 5 alpha-dihydrotestosterone (DHT) and 5 alpha-reductases in teleost fishes and amphibians." General and comparative endocrinology 194: 264-274.
Panter, G. H., T. H. Hutchinson, K. S. Hurd, A. Sherren, R. D. Stanley and C. R. Tyler (2004). "Successful detection of (anti-)androgenic and aromatase inhibitors in pre-spawning adult fathead minnows (Pimephales promelas) using easily measured endpoints of sexual development." Aquatic toxicology (Amsterdam, Netherlands) 70(1): 11-21.
Rajakumar, A., R. Singh, S. Chakrabarty, R. Murugananthkumar, C. Laldinsangi, Y. Prathibha, C. C. Sudhakumari, A. Dutta-Gupta and B. Senthilkumaran (2012). "Endosulfan and flutamide impair testicular development in the juvenile Asian catfish, Clarias batrachus." Aquatic toxicology (Amsterdam, Netherlands) 110-111: 123-132.
Sullivan, C. V. and O. Yilmaz (2018). Vitellogenesis and Yolk Proteins, Fish. Encyclopedia of Reproduction (Second Edition). M. K. Skinner. Oxford, Academic Press: 266-277.
Villeneuve, D. L., N. D. Mueller, D. Martinović, E. A. Makynen, M. D. Kahl, K. M. Jensen, E. J. Durhan, J. E. Cavallin, D. Bencic and G. T. Ankley (2009). "Direct effects, compensation, and recovery in female fathead minnows exposed to a model aromatase inhibitor." Environmental health perspectives 117(4): 624-631.
Dahye Kim, Hyunki Cho, Ruth Eggers, Sang Kyum Kim, Chang Seon Ryu and Young Jun Kim (2020)"Development of a Liquid Chromatography/Mass Spectrometry-Based Inhibition Assay for the Screening of Steroid 5-α Reductase in Human and Fish Cell Lines" Molecules 2021, 26, 893
