
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 39
Title
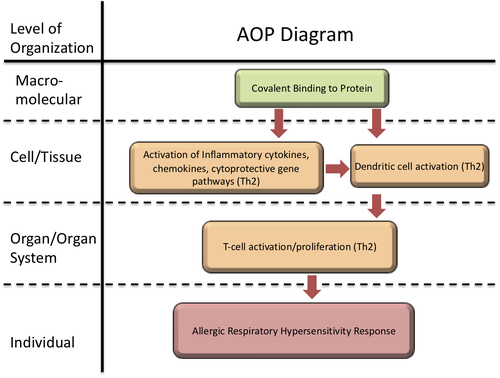
Covalent Binding, Protein, leading to Increase, Allergic Respiratory Hypersensitivity Response
Short name
Graphical Representation
Point of Contact
Contributors
- Kristie Sullivan
- Jessica Ponder
- Arthur Author
Coaches
- Jason M. O'Brien
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.20 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increase, Allergic Respiratory Hypersensitivity Response | February 07, 2023 13:07 |
| Activation, Dendritic Cells | December 03, 2020 10:15 |
| Activation/Proliferation, T-cells | November 05, 2020 19:14 |
| Covalent Binding, Protein | November 05, 2020 18:40 |
| Increased, secretion of proinflammatory mediators | May 17, 2023 15:18 |
| Covalent Binding, Protein leads to Increased proinflammatory mediators | September 08, 2022 05:24 |
| Covalent Binding, Protein leads to Increase, Allergic Respiratory Hypersensitivity Response | August 01, 2022 19:20 |
| Covalent Binding, Protein leads to Activation, Dendritic Cells | November 05, 2020 18:09 |
| Increased proinflammatory mediators leads to Activation, Dendritic Cells | August 01, 2022 14:06 |
| Activation, Dendritic Cells leads to Activation/Proliferation, T-cells | August 22, 2022 13:46 |
| Activation/Proliferation, T-cells leads to Increase, Allergic Respiratory Hypersensitivity Response | August 01, 2022 19:20 |
| Toluene diisocyanate | August 01, 2022 14:18 |
Abstract
The assessment of xenobiotics for potential to induce an allergenic response in the respiratory tract is of great regulatory and industrial interest. Ongoing work in this area has hypothesized some differences between the dermal and respiratory sensitisation pathways; however in some cases a lack of strong empirical evidence on a variety of chemistries to test these hypothesis. This AOP represents the currently available data with the aim of identifying knowledge gaps which may be filled with directed research. (Sullivan, et al., 2017)
Sensitization of the respiratory tract is an important occupational health challenge. Here we build on a previously published skin sensitization AOP (AOP 40), relying on literature evidence linked to low-molecular-weight organic chemicals and excluding other known respiratory sensitizers acting via different molecular initiating events. The established key events (KEs) are as follows: (1) covalent binding of chemicals to proteins, (2) activation of cellular danger signals (inflammatory cytokines and chemokines and cytoprotective gene pathways), (3) dendritic cell activation and migration, (4) activation, proliferation, and polarization of T cells, and (5) sensitization of the respiratory tract. There is some evidence that respiratory sensitizers bind preferentially to lysine moieties, whereas skin sensitizers bind to both cysteine and lysine, however this observation may be biased by the limited number of chemicals investigated. Furthermore, exposure to respiratory sensitizers seems to result in cell behavior for KEs 2 and 3, as well as the effector T cell response, in general skewing toward cytokine secretions predominantly associated with T helper 2 (Th2) response.
AOP Development Strategy
Context
Sensitization of the respiratory tract by chemicals is the first stage in the development of chemical respiratory allergy, an immune-mediated hypersensitivity reaction to an exogenous low-molecular-weight chemical, which can result in asthma and rhinitis on repeated exposure and is an important occupational health problem. (Mapp et al., 2005) Due to the severity and irreversibility of the adverse effect, identifying chemical respiratory allergens is of considerable regulatory, industrial, and socioeconomic importance. (Boverhof et al., 2008) Efforts to outline a framework for assessment of potential respiratory-sensitizing chemicals are underway. (North et al., 2016) Currently, however, there are no standardized, validated, and regulatory-accepted models for detecting these chemicals or discriminating them from skin sensitizers, potentially due to remaining gaps within the literature as to the exact mechanistic steps leading to respiratory allergy. (Kimber et al., 2011)
Another important issue in the development of predictive test methods is the route of exposure in the sensitization phase. Inhalation exposure is perhaps the most common exposure route of concern for many substances, but there is evidence that sensitization of the respiratory tract can be induced via skin exposure as well. (Tarlo and Malo, 2006, Heederik et al., 2012, Redlich and Herrick, 2008, Kimber and Dearman, 2002) This knowledge has implications for both the mechanistic understanding of the pathway and the potential test methods that may be used to detect respiratory sensitizers. Therefore, this AOP will include information from models using skin and lung exposure (in vivo) and with dermal and respiratory cells and tissues (in vitro/ex vivo).
The outlines of this pathway follow the already-published skin sensitization AOP 40. However, the divergent AOs of the two pathways reflect differences in the effector response (T helper 1 [Th1] vs. T helper 2 [Th2]) and other mechanistic details of at least some KEs;(Kimber et al., 2014) these differences are the focus of this effort. Therefore, the primary evidence relied on to build this AOP must relate directly to known low-molecular-weight organic chemicals to the exclusion of chemicals that act via other mechanisms and therefore require a separate AOP, for example, chloroplatinates.
In brief, the AOP can be summarized as beginning with covalent protein binding, potentially preferentially to lysine nucleophiles in the lung or skin after respiratory or dermal exposure to a low-molecular-weight organic chemical. This protein binding causes the activation of stress response pathways and cellular danger signals, including oxidative stress, cytokines, and chemokines released by epithelial and other cells, leading to dendritic cell (DC) maturation and migration to the draining lymph nodes (DLN). Haptens can also contribute to DC activation directly. Th2-skewed DCs in the DLN signal activation and maturation of T cells, which characterize the sensitization phase, resulting in chemical respiratory allergy. Consistent with regulatory practice, sensitization is considered the AO. (Vandebriel et al., 2011)
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 396 | Covalent Binding, Protein | Covalent Binding, Protein |
| KE | 1496 | Increased, secretion of proinflammatory mediators | Increased proinflammatory mediators |
| KE | 398 | Activation, Dendritic Cells | Activation, Dendritic Cells |
| KE | 272 | Activation/Proliferation, T-cells | Activation/Proliferation, T-cells |
| AO | 313 | Increase, Allergic Respiratory Hypersensitivity Response | Increase, Allergic Respiratory Hypersensitivity Response |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Covalent Binding, Protein leads to Increased proinflammatory mediators | adjacent | High | Not Specified |
| Covalent Binding, Protein leads to Activation, Dendritic Cells | adjacent | High | Not Specified |
| Increased proinflammatory mediators leads to Activation, Dendritic Cells | adjacent | Low | Low |
| Activation, Dendritic Cells leads to Activation/Proliferation, T-cells | adjacent | High | Not Specified |
| Activation/Proliferation, T-cells leads to Increase, Allergic Respiratory Hypersensitivity Response | adjacent | High | Not Specified |
Network View
Prototypical Stressors
| Name |
|---|
| Toluene diisocyanate |
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | Not Specified |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | Not Specified |
Overall Assessment of the AOP
1. Concordance of dose-response relationships
There is fairly sparse evidence wth more than a few respiratory sensitizers that can offer confidence in dose-response concordance. In BALB/c mice, toluene diisocyanate (TDI) applied to the skin led to TDI-haptenated protein (TDI-hp) (skin keratins and albumin) localization in the stratum corneum, hair follicles, and sebaceous glands within 3 hours, with intensity of staining following a dose–response relationship. (Nayak et al., 2014) provides a detailed dose–response analysis of TDI-induced protein binding, colocalization of immune messenger cells, and migration to Delphian lymph nodes.
2. Temporal concordance among the key events and adverse effect;
(Nayak et al., 2014) also provides a detailed temporal analysis of TDI-induced protein binding, colocalization of immune messenger cells, and migration to Delphian lymph nodes.
3. Strength, consistency, and specificity of association of adverse effect and initiating event
There are clear connections from chemicals with certain reactivity and binding profiles to cellular- and individual-level downstream Th2-related effects leading to respiratory sensitization. Though the number of chemicals studied is quite low, consistent patterns are identified. A better understanding of how differences in haptenation by these chemicals contribute to distinct cellular and immune-system-level responses, and how early DC gene changes contribute (or not) to the expression of maturation markers, will help to increase the specificity of the available test methods.
4. Biological plausibility, coherence, and consistency of the experimental evidence
Each of the hypothesized KERs is supported by evidence from studies with at least one, and sometimes a few, known respiratory sensitizers. The events fit with what is known in general for sensitization, and the basic KEs outlined here are consistent with established biological knowledge. However, further research is needed to understand, for a larger number of chemicals, the steps leading to a skewing of the effector response toward Th2 and sensitization of the respiratory tract; therefore, the WoE is considered to be ‘‘moderate.’’
5. Alternative mechanisms
Currently, there are about 80 chemicals identified as respiratory allergens. Exposure occurs primarily in occupational settings. AOs are asthma and rhinitis. The biological mechanisms are often Th2 mediated leading to the production of IgE and eosinophilic inflammation. However, this may not always be the case. For example, human studies reveal PPD to be a respiratory sensitizer, (Helaskoski et al., 2014) but it does not cause a Th2 cytokine response in mice. (Rothe et al., 2011) Specific IgE is induced in some subjects, but not in others, particularly for diisocyanate sensitization. Thus, it is unclear whether IgE is mandatory or not.
Notably, it has to be mentioned that for protein-induced respiratory allergy, the clinical understanding of the disease has been changing dramatically during the last years. For many years, asthma has been considered as a single disease with a defined phenotype. It was assumed that the biology of sensitization is based on Th2-mediated IgE production, migration of mast cells, and subsequent eosinophilic infiltration. Nevertheless, clinical studies of cohort revealed that only about 50% of all patients show a Th2-driven eosinophilic inflammation of the airways. It also covers Th17-driven neutrophilic airway inflammation—an asthmatic phenotype that also can be observed with chemical allergens.
Nowadays, asthma is considered as an umbrella disease with multiple heterogeneous phenotypes, depending on the underlying immunology, pathology, symptoms, and the time of elicitation during lifetime. Furthermore, the concept takes other environmental and genetic influences into consideration. The development of animal models reflecting the heterogeneity of asthma phenotypes is still ongoing and shows in particular the (i) irritant properties of the allergen, (ii) the route of exposure during sensitization and elicitation, and (iii) the dose levels of allergen define whether a Th2 or Th17 phenotype develops.
For chemical allergens, less is known about the influence of atopy, viral infections, and indoor and outdoor environmental pollutants such as cigarette smoke. Of interest is the influence of an additional coexposure to irritant if the chemical allergen is present at low dose. Genetic susceptibility is also a variable of interest. (Yucesoy et al., 2012) and (Wisnewski et al., 2008) among others, have determined factors that may affect the potential for a person’s sensitization potential to diisocyanates, including genetic variants in antioxidant defense genes and PRRs.
A number of studies have looked into the sensitization of transition metal complexes, including one which outlines the evidence for these complexes initiating sensitization not through covalent bond formation, but rather through coordination complexes. (Chipinda et al., 2011) The authors provide evidence that these coordination complexes are not stable enough to survive the antigen processing that a covalent hapten undergoes. Instead an alternative MIE is outlined in which these complexes bind to cell surface proteins like MHC, bypassing the intracellular antigen process. This initiating event fits in with the observed cross-reactivity that appears to transcend the trends one would expect based on the periodic table (for example, complexes of Cr, a group 6 metal, cross sensitizing with complexes of Co, a group 9 metal). (Templeton, 2004) It is thought that the surface protein chelates the metal complex and presents it to T-cells directly, requiring a separate AOP from chemicals acting via covalent binding to proteins.
6. Uncertainties, inconsistencies and data gaps.
A better understanding of how differences in haptenation by these chemicals contribute to distinct cellular responses, and how early DC gene changes contribute (or not) to the expression of maturation markers, will help to increase the specificity of the available test methods. A better understanding of human response and population variability is also needed, along with a better quantitative understanding of the linkages between KEs. Additional studies using human cells and tissues are recommended.
Furthermore, as noted in the evaluation section, efforts to fully understand this pathway and develop toxicological test methods and strategies are hampered by a spare data portfolio, as well as a lack of a robust set of harmonized reference chemicals clearly identified as respiratory sensitizers. Previous authors have gathered preliminary chemical sets with supporting rationale, and collating this information and building a set of harmonized reference chemicals, which can be used to optimize and characterize potential test methods or strategies, are the clear next steps. (Enoch et al., 2010, Cochrane et al., 2015, Enoch et al., 2009)
Domain of Applicability
Sensitizers which do not fit into this AOP:
There have been a number of studies into the sensitisation (and toxicity) of transition metal complexes; key amongst these is a recent study outlining the evidence for these complexes initiating sensitisation via the formation of co-ordination complexes rather than covalent bond formation. (Chipinda et al., 2011) The authors of this study present the evidence that these co-ordination complexes are not stable enough to survive the antigen processing that a covalent hapten undergoes, thus cannot sensitise via this MIE. Instead an alternative MIE is outlined in which these complexes bind to cell surface proteins like MHC, bypassing the intracellular antigen process. This MIE fits in with the observed cross-reactivity that appears to transcend the trends one would expect based on the periodic table (for example, complexes of Cr, a group 6 metal, cross sensitising with complexes of Co, a group 9 metal). (Templeton, 2004) It is thought that the surface protein chelates the metal complex and presents it to T-cells directly. Therefore, transition metals would require a separate AOP from chemicals acting via covalent binding to proteins.
Essentiality of the Key Events
|
Support for Essentiality of KEs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
Direct evidence from experimental studies illustrating essentiality for at least one of the important KEs. |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE. |
No or contradictory experimental evidence of the essentiality of any of the KEs. |
|
|
MIE (KE1): Covalent Binding, Protein |
Moderate |
There is extensive evidence in the literature for haptenation being the MIE for respiratory sensitization. In general, haptenation can be divided into five types of chemistry, so-called mechanistic domains. These being acylation, aliphatic nucleophilic substitution (SN1/SN2), aromatic nucleophilic substitution (SNAr), Michael addition, and Schiff base formation. (Enoch et al., 2011, Aptula et al., 2005, Aptula and Roberts, 2006, Lalko et al., 2011, Landsteiner and Jacobs, 1935, Landsteiner and Jacobs, 1936, Hopkins et al., 2005) |
||
|
KE2: Activation of Inflammatory Signaling |
High |
Strong evidence exists for the essential nature of cellular danger signals in respiratory sensitization. (Silva et al., 2014) found that HDI increased ROS by inhibiting superoxide dismutase (SOD1) in THP-1 cells. This inhibition may further encourage a redox environment via matrix metalloproteinase (MMP reduction). Increased ROS also led to extracellular signal-related kinase (ERK) signaling pathway phosphorylation and the transcription of cytoprotective and maturation pathways (HMOX1 and CD83). Coincubation with the antioxidant n-acetyl cysteine and SOD decreased ERK phosphorylation. |
||
|
KE3: Dendritic cells activation |
High |
Some evidence indicates that IL-10, upregulated by TMA, may block the migration of LC for a short period of time to allow a Th2 phenotype to develop. Increased IL-4 and IL-10 were detected in the draining lymph nodes of mice after TMA exposure, and DC migration to the DLN was confirmed. Anti-IL-10 antibody ameliorated this response to TMA. (Holden et al., 2008, Cumberbatch et al. 2005) |
||
|
KE4: T-cells, activation and proliferation |
High |
In humans, support for the Th2-skewing being associated with sensitization of the respiratory tract rather than the skin comes from studying the responses of individuals who already have an immune response skewed in one direction or the other. (Holden et al., 2008, Newell et al., 2013, Ouyang et al., 2013) |
||
Evidence Assessment
|
Support for Biological Plausibility of KERs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
Extensive understanding of the KER based on previous documentation and broad acceptance. |
KER is plausible based on analogy to,accepted biological relationships, but scientific understanding is incomplete. |
Empirical support for association between KEs, but the structural or functional relationship between them is not understood. |
|
|
MIE => KE2: Covalent Binding, Protein leads to Activation of Inflammatory Signaling |
High |
It has been demonstrated with lung cell lines that exposure to haptenated human serum albumin increases reactive oxygen species. (Hur et al., 2009) It is well accepted and experimentally proved in lung cell lines, 3D human airway epithelial cell cultures, and human lung slices that exposure to haptens induces proinflammatory cytokine and chemokine (e.g. IL-1α, TNF-α, IL-6, IL-8, CCL2, CXCL1, CCL5, etc.) release. (Huang et al., 2013, Lauenstein et al., 2014, Verstraelen et al., 2009) |
||
|
MIE => KE3: Covalent Binding, Protein leads to Dendritic Cells Activation |
High |
It is well-accepted and experimentally proven that dendritic cells represent the most important antigen-presenting cells in the lung. Immature DCs are distributed above and beneath the basal membrane of the lung epithelium and sample antigens by extending dendrites into the airway lumen. Immature cells mature after encountering antigen, an essential event in the activation of immune response. (Lambrecht and Hammad, 2010, Lambrecht and Hammad, 2003, Lambrecht and Hammad, 2009, Holt et al., 1994) |
||
|
KE2 => KE3: Activation of Inflammatory Signaling leads to Dendritic Cells Activation |
Low |
DCs express receptors for, and respond to, constitutive and inflammatory chemokines and other chemoattractants, such as platelet-activating factor and formyl peptides. Much investigation has gone into assessing the specific mechanistic events involved in skin sensitizer-caused DC migration. Ex vivo studies with intact human skin, epidermal sheets, and MUTZ-3-derived Langerhans cells (LC) show that fibroblasts mediate migration of cytokine-matured LC via chemokines, including CXCL12, CXCR4, and dermis-derived CCL2 and CCL5. (Ouwehand, et al., 2011) The relevance of these studies for respiratory sensitization is not known. |
||
|
KE3 => KE4: Dendritic Cells Activation leads to T-cells, activation and proliferation |
High |
It is well-accepted and experimentally proven that a Th2-type T cell polarization is associated with respiratory sensitization. (Hopkins et al., 2005, Huang et al., 2013) |
||
|
KE4 => AO: T-cells, activation and proliferation leads to Sensitisation of the Respiratory Tract |
High |
It is well-demonstrated that clonal expansion of Th2 cells leads to the production of Th2 cytokines that induce Ig class-switching, with clonal expansion of B cells producing antigen-specific IgE. (Dearman et al., 2003) |
||
|
Empirical Support for KERs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? Does KEup occur at lower doses, earlier time points, and higher in incidence than KEdown ? Inconsistencies? |
Multiple studies showing dependent change in both events following exposure to a wide range of specific stressors. No or few critical data gaps or conflicting data.. |
Demonstrated dependent change in both events following exposure to a small number of stressors. Some inconsistencies with expected pattern that can be explained by various factors. |
Limited or no studies reporting dependent change in both events following exposure to a specific stressor; and/or significant inconsistencies in empirical support across taxa and species |
|
|
MIE => KE2: Covalent Binding, Protein leads to Activation of Inflammatory Signaling |
Moderate |
Haptenated peptides generated in vitro can be quantified after 15 minutes. (Hettick, et al., 2009) Most in vitro cellular assay protocols quantify inflammatory readouts after 24 – 48 hours of exposure. TMA induced increased production of IL-10 when incubated with precision cut lung slices (PCLS) for 24 hours. (Lauenstein et al., 2014) | ||
|
MIE => KE3: Covalent Binding, Protein leads to Dendritic Cells Activation |
Moderate |
In BALB/c mice, TDI applied to the skin led to TDI-haptenated protein (TDI-hp) (skin keratins and albumin) localization in the stratum corneum, hair follicles, and sebaceous glands within 3 hours, with intensity of staining following a dose–response relationship (Nayak et al. 2014). Subsequently, CD11b+, Langerin (CD207)-expressing DCs, and CD103+ cells migrated to regions of TDI-hp staining. These cells are involved in antigen uptake and stimulation of effector T cells. |
||
|
KE2 => KE3: Activation of Inflammatory Signaling leads to Dendritic Cells Activation |
Low |
(Silva et al., 2014) found that HDI increased ROS by inhibiting superoxide dismutase (SOD1) in THP-1 cells. Increased ROS also led to extracellular signal-related kinase (ERK) signaling pathway phosphorylation and the transcription of cytoprotective and maturation pathways (HMOX1 and CD83). |
||
|
KE3 => KE4: Dendritic Cells Activation leads to T-cells, activation and proliferation |
Low |
There is little known about many aspects of antigen processing, such as uptake pathway, peptide generation, and MHC peptide complex stability and density, in chemical sensitization of the respiratory tract. Differences may exist in how skin and respiratory sensitizers are processed that may provide key insight into how to distinguish such chemicals. (Hopkins et al, 2005) found increased expression of type 2 cytokines n mouse lymph node cells after topical exposure to TMA and FITC. | ||
|
KE4 => AO: T-cells, activation and proliferation leads to Sensitisation of the Respiratory Tract |
Low |
T-cells are typically affected by protein-hapten complexes presented by dendritic cells on MHC molecules. The T-cell will be then activated to form a memory T-cell, which subsequently proliferates (Vocanson et al., 2009) |
||
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Frequency of exposures to toluene diisocyanate exceeding 3 ppb in the time-weighted average (8 hrs, TWA-8) without respiratory protection were found to be associated with incidence. In this study, TWA-8 values above 3 ppb were indicative of peak exposure events, i.e. spills. (Plehiers et al., 2020a and 2020b) This is consistent with a prior report by (Collins et al., 2017) which found a significant link between peak exposure and asthma incidence.
Considerations for Potential Applications of the AOP (optional)
The construction of an AOP for this endpoint would allow the following: (1) organization of available information to identify remaining uncertainties and prioritize further research, (2) highlighting of differences and similarities between skin and respiratory sensitization pathways, and (3) improvement of existing or identification of novel predictive models that, alone or in an integrated approach, could be used to identify respiratory sensitizers.
Given the available (WoE) outlined above, we propose that the AOP for sensitization of the respiratory tract outlined here allows the identification of gaps in knowledge, research needs, and potential test methods that may be developed further using a larger set of respiratory sensitizers.
References
APTULA, A. O., PATLEWICZ, G. & ROBERTS, D. W. 2005. Skin sensitization: reaction mechanistic applicability domains for structure-activity relationships. Chem Res Toxicol, 18, 1420-6.
APTULA, A. O. & ROBERTS, D. W. 2006. Mechanistic applicability domains for nonanimal-based prediction of toxicological end points: general principles and application to reactive toxicity. Chem Res Toxicol, 19, 1097-105.
BOVERHOF, D. R., BILLINGTON, R., GOLLAPUDI, B. B., HOTCHKISS, J. A., KRIEGER, S. M., POOLE, A., WIESCINSKI, C. M. & WOOLHISER, M. R. 2008. Respiratory sensitization and allergy: current research approaches and needs. Toxicol Appl Pharmacol, 226, 1-13.
CHIPINDA, I., HETTICK, J. M. & SIEGEL, P. D. 2011. Haptenation: chemical reactivity and protein binding. J Allergy (Cairo), 2011, 839682.
COCHRANE, S. A., ARTS, J. H. E., EHNES, C., HINDLE, S., HOLLNAGEL, H. M., POOLE, A., SUTO, H. & KIMBER, I. 2015. Thresholds in chemical respiratory sensitisation. Toxicology, 333, 179-194.
COLLINS, J. J., ANTEAU, S., CONNER, P. R., CASSIDY, L. D., DONEY, B., WANG, M. L., KURTH, L., CARSON, M., MOLENAAR, D., REDLICH, C. A. & STOREY, E. 2017. Incidence of Occupational Asthma and Exposure to Toluene Diisocyanate in the United States Toluene Diisocyanate Production Industry. Journal of occupational and environmental medicine, 59 Suppl 12, S22-S27.
ENOCH, S. J., ELLISON, C. M., SCHULTZ, T. W. & CRONIN, M. T. 2011. A review of the electrophilic reaction chemistry involved in covalent protein binding relevant to toxicity. Crit Rev Toxicol, 41, 783-802.
ENOCH, S. J., ROBERTS, D. W. & CRONIN, M. T. 2009. Electrophilic reaction chemistry of low molecular weight respiratory sensitizers. Chem Res Toxicol, 22, 1447-53.
ENOCH, S. J., ROBERTS, D. W. & CRONIN, M. T. 2010. Mechanistic category formation for the prediction of respiratory sensitization. Chem Res Toxicol, 23, 1547-55.
HEEDERIK, D., HENNEBERGER, P. K. & REDLICH, C. A. 2012. Primary prevention: exposure reduction, skin exposure and respiratory protection. Eur Respir Rev, 21, 112-24.
HELASKOSKI, E., SUOJALEHTO, H., VIRTANEN, H., AIRAKSINEN, L., KUULIALA, O., AALTO-KORTE, K. & PESONEN, M. 2014. Occupational asthma, rhinitis, and contact urticaria caused by oxidative hair dyes in hairdressers. Ann Allergy Asthma Immunol, 112, 46-52.
HETTICK, J.M., RUWONA, T.B. & SIEGEL, P.D. 2009. Structural elucidation of isocyanate-peptide adducts using tandem mass spectrometry. J Am Soc Mass Spectrom 20, 1567–1575.
HOLDEN, N. J., BEDFORD, P. A., MCCARTHY, N. E., MARKS, N. A., IND, P. W., JOWSEY, I. R., BASKETTER, D. A. & KNIGHT, S. C. 2008. Dendritic cells from control but not atopic donors respond to contact and respiratory sensitizer treatment in vitro with differential cytokine production and altered stimulatory capacity. Clin Exp Allergy, 38, 1148-59.
HOLT, P. G., HAINING, S., NELSON, D. J. & SEDGWICK, J. D. 1994. Origin and steady-state turnover of class II MHC-bearing dendritic cells in the epithelium of the conducting airways. J Immunol, 153, 256-61.
HUANG, S., WISZNIEWSKI, L., CONSTANT, S. & ROGGEN, E. 2013. Potential of in vitro reconstituted 3D human airway epithelia (MucilAir™) to assess respiratory sensitizers. Toxicol In Vitro, 27, 1151-6.
HUR, G. Y., KIM, S. H., PARK, S. M., YE, Y. M., KIM, C. W., JANG, A. S., PARK, C. S., HONG, C. S. & PARK, H. S. 2009. Tissue transglutaminase can be involved in airway inflammation of toluene diisocyanate-induced occupational asthma. J Clin Immunol, 29, 786-94.
KAROL, M. H. & STOLIKER, D. 1999. Immunotoxicology: past, present, and future. Inhal Toxicol, 11, 523-34.
KIMBER, I., BASKETTER, D. A., GERBERICK, G. F., RYAN, C. A. & DEARMAN, R. J. 2011. Chemical allergy: translating biology into hazard characterization. Toxicol Sci, 120 Suppl 1, S238-68.
KIMBER, I. & DEARMAN, R. J. 2002. Chemical respiratory allergy: role of IgE antibody and relevance of route of exposure. Toxicology, 181-182, 311-5.
KIMBER, I., DEARMAN, R. J., BASKETTER, D. A. & BOVERHOF, D. R. 2014. Chemical respiratory allergy: reverse engineering an adverse outcome pathway. Toxicology, 318, 32-9.
LALKO, J. F., KIMBER, I., DEARMAN, R. J., API, A. M. & GERBERICK, G. F. 2013. The selective peptide reactivity of chemical respiratory allergens under competitive and non-competitive conditions. J Immunotoxicol, 10, 292-301.
LALKO, J. F., KIMBER, I., DEARMAN, R. J., GERBERICK, G. F., SARLO, K. & API, A. M. 2011. Chemical reactivity measurements: potential for characterization of respiratory chemical allergens. Toxicol In Vitro, 25, 433-45.
LAMBRECHT, B. N. & HAMMAD, H. 2003. Taking our breath away: dendritic cells in the pathogenesis of asthma. Nature Reviews Immunology, 3, 994-1003.
LAMBRECHT, B. N. & HAMMAD, H. 2009. Biology of Lung Dendritic Cells at the Origin of Asthma. Immunity, 31, 412-424.
LAMBRECHT, B. N. & HAMMAD, H. 2010. The role of dendritic and epithelial cells as master regulators of allergic airway inflammation. Lancet, 376, 835-43.
LANDSTEINER, K. & JACOBS, J. 1935. STUDIES ON THE SENSITIZATION OF ANIMALS WITH SIMPLE CHEMICAL COMPOUNDS. J Exp Med, 61, 643-56.
LANDSTEINER, K. & JACOBS, J. 1936. STUDIES ON THE SENSITIZATION OF ANIMALS WITH SIMPLE CHEMICAL COMPOUNDS. II. J Exp Med, 64, 625-39.
LANGE, R. W., DAY, B. W., LEMUS, R., TYURIN, V. A., KAGAN, V. E. & KAROL, M. H. 1999. Intracellular S-glutathionyl adducts in murine lung and human bronchoepithelial cells after exposure to diisocyanatotoluene. Chem Res Toxicol, 12, 931-6.
LANTZ, R. C., LEMUS, R., LANGE, R. W. & KAROL, M. H. 2001. Rapid reduction of intracellular glutathione in human bronchial epithelial cells exposed to occupational levels of toluene diisocyanate. Toxicol Sci, 60, 348-55.
LAUENSTEIN, L., SWITALLA, S., PRENZLER, F., SEEHASE, S., PFENNIG, O., FÖRSTER, C., FIEGUTH, H., BRAUN, A. & SEWALD, K. 2014. Assessment of immunotoxicity induced by chemicals in human precision-cut lung slices (PCLS). Toxicol In Vitro, 28, 588-99.
MAPP, C. E., BOSCHETTO, P., MAESTRELLI, P. & FABBRI, L. M. 2005. Occupational asthma. Am J Respir Crit Care Med, 172, 280-305.
NATSCH, A., RYAN, C. A., FOERTSCH, L., EMTER, R., JAWORSKA, J., GERBERICK, F. & KERN, P. 2013. A dataset on 145 chemicals tested in alternative assays for skin sensitization undergoing prevalidation. J Appl Toxicol, 33, 1337-52.
NAYAK, A. P., HETTICK, J. M., SIEGEL, P. D., ANDERSON, S. E., LONG, C. M., GREEN, B. J. & BEEZHOLD, D. H. 2014. Toluene diisocyanate (TDI) disposition and co-localization of immune cells in hair follicles. Toxicol Sci, 140, 327-37.
NEWELL, L., POLAK, M. E., PERERA, J., OWEN, C., BOYD, P., PICKARD, C., HOWARTH, P. H., HEALY, E., HOLLOWAY, J. W., FRIEDMANN, P. S. & ARDERN-JONES, M. R. 2013. Sensitization via healthy skin programs Th2 responses in individuals with atopic dermatitis. J Invest Dermatol, 133, 2372-2380.
NORTH, C. M., EZENDAM, J., HOTCHKISS, J. A., MAIER, C., AOYAMA, K., ENOCH, S., GOETZ, A., GRAHAM, C., KIMBER, I., KARJALAINEN, A., PAULUHN, J., ROGGEN, E. L., SELGRADE, M., TARLO, S. M. & CHEN, C. L. 2016. Developing a framework for assessing chemical respiratory sensitization: A workshop report. Regul Toxicol Pharmacol, 80, 295-309.
OUYANG, B., BERNSTEIN, D. I., LUMMUS, Z. L., YING, J., BOULET, L. P., CARTIER, A., GAUTRIN, D. & HO, S. M. 2013. Interferon-γ promoter is hypermethylated in blood DNA from workers with confirmed diisocyanate asthma. Toxicol Sci, 133, 218-24.
OUWEHAND K, SPIEKSTRA SW, WAAJIMAN T, SCHEPER RJ, DE GRUJIL TD, GIBBS S. 2011. Technical advance: Langerhans cells derived from a human cell line in a full-thickness skin equivalent undergo allergen-induced maturation and migration. J Leukoc Biol. 290(5):1027-33.
PLEHIERS, P. M., CHAPPELLE, A. H. & SPENCE, M. W. 2020a. Practical learnings from an epidemiology study on TDI-related occupational asthma: Part I-Cumulative exposure is not a good indicator of risk. Toxicol Ind Health, 36, 876-884. PLEHIERS, P. M., CHAPPELLE, A. H. & SPENCE, M. W. 2020b. Practical learnings from an epidemiology study on TDI-related occupational asthma: Part II-Exposure without respiratory protection to TWA-8 values indicative of peak events is a good indicator of risk. Toxicol Ind Health, 36, 885-891.
REDLICH, C. A. & HERRICK, C. A. 2008. Lung/skin connections in occupational lung disease. Curr Opin Allergy Clin Immunol, 8, 115-9.
REMY, S., VERSTRAELEN, S., VAN DEN HEUVEL, R., NELISSEN, I., LAMBRECHTS, N., HOOYBERGHS, J. & SCHOETERS, G. 2014. Gene expressions changes in bronchial epithelial cells: markers for respiratory sensitizers and exploration of the NRF2 pathway. Toxicol In Vitro, 28, 209-17.
ROTHE, H., SARLO, K., SCHEFFLER, H. & GOEBEL, C. 2011. The hair dyes PPD and PTD fail to induce a T(H)2 immune response following repeated topical application in BALB/c mice. J Immunotoxicol, 8, 46-55.
SILVA, A., NUNES, C., MARTINS, J., DINIS, T. C., LOPES, C., NEVES, B. & CRUZ, T. 2014. Respiratory sensitizer hexamethylene diisocyanate inhibits SOD 1 and induces ERK-dependent detoxifying and maturation pathways in dendritic-like cells. Free Radic Biol Med, 72, 238-46.
SULLIVAN, K.M., ENOCH, S.J., EZENDAM, J., SEWALD, K., ROGGEN, E.L., COCHRANE, S. 2017. An Adverse Outcome Pathway for Sensitization of the Respiratory Tract by Low-Molecular-Weight Chemicals: Building Evidence to Support the Utility of In Vitro and In Silico Methods in a Regulatory Context. Appl In Vitro Tox, 3:3, 213-226
TARLO, S. M. & MALO, J. L. 2006. An ATS/ERS report: 100 key questions and needs in occupational asthma. Eur Respir J, 27, 607-14.
TEMPLETON, D. 2004. Mechanisms of immunosensitization to metals (IUPAC Technical Report). Pure and Applied Chemistry - PURE APPL CHEM, 76, 1255-1268.
VANDEBRIEL, R., CALLANT CRANSVELD, C., CROMMELIN, D., DIAMANT, Z., GLAZENBURG, B., JOOS, G., KUPER, F., NATSCH, A., NIJKAMP, F., NOTEBORN, H., PIETERS, R., ROBERTS, D., ROGGEN, E., RORIJE, E., SEED, M., SEWALD, K., VAN DEN HEUVEL, R., VAN ENGELEN, J., VERSTRAELEN, S. & VAN LOVEREN, H. 2011. Respiratory sensitization: advances in assessing the risk of respiratory inflammation and irritation. Toxicol In Vitro, 25, 1251-8.
VERSTRAELEN, S., NELISSEN, I., HOOYBERGHS, J., WITTERS, H., SCHOETERS, G., VAN CAUWENBERGE, P. & VAN DEN HEUVEL, R. 2009. Gene profiles of a human alveolar epithelial cell line after in vitro exposure to respiratory (non-)sensitizing chemicals: identification of discriminating genetic markers and pathway analysis. Toxicol Lett, 185, 16-22.
VOCANSON M, HENNINO A, ROZIERES A, POYET G, NICOLAS JF. 2009. Effector and regulatory mechanisms in allergic contact dermatitis. Allergy. 64(12), 1699-714.
WISNEWSKI, A. V., LIU, Q., LIU, J. & REDLICH, C. A. 2008. Human innate immune responses to hexamethylene diisocyanate (HDI) and HDI-albumin conjugates. Clin Exp Allergy, 38, 957-67.
YUCESOY, B., JOHNSON, V. J., LUMMUS, Z. L., KISSLING, G. E., FLUHARTY, K., GAUTRIN, D., MALO, J. L., CARTIER, A., BOULET, L. P., SASTRE, J., QUIRCE, S., GERMOLEC, D. R., TARLO, S. M., CRUZ, M. J., MUNOZ, X., LUSTER, M. I. & BERNSTEIN, D. I. 2012. Genetic variants in antioxidant genes are associated with diisocyanate-induced asthma. Toxicol Sci, 129, 166-73.