
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 43
Title
Disruption of VEGFR Signaling Leading to Developmental Defects
Short name
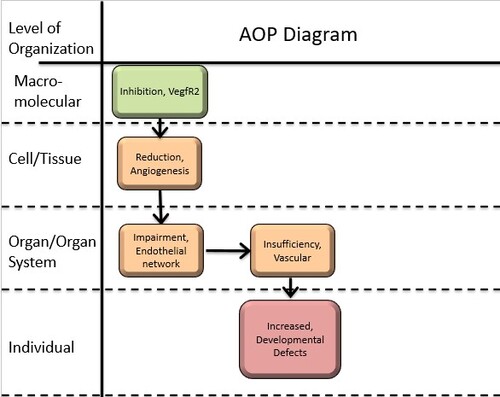
Graphical Representation
Point of Contact
Contributors
- Nancy Baker
- Cataia Ives
Coaches
- Dan Villeneuve
- Rex FitzGerald
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.6 | WPHA/WNT Endorsed |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition, VegfR2 | January 17, 2022 10:05 |
| Reduction, Angiogenesis | July 20, 2022 13:04 |
| Impairment, Endothelial network | January 18, 2022 13:21 |
| Insufficiency, Vascular | July 20, 2022 17:07 |
| Increased, Developmental Defects | August 31, 2023 12:37 |
| Inhibition, VegfR2 leads to Reduction, Angiogenesis | July 20, 2022 17:33 |
| Reduction, Angiogenesis leads to Impairment, Endothelial network | January 18, 2022 12:54 |
| Impairment, Endothelial network leads to Insufficiency, Vascular | January 18, 2022 13:05 |
| Insufficiency, Vascular leads to Increased, Developmental Defects | January 18, 2022 13:07 |
| Vatalanib | July 25, 2022 09:09 |
| Sunitinib malate Sunitinib (INN) | July 25, 2022 09:19 |
Abstract
BACKGROUND: The cardiovascular system is the first functional organ system to develop in the vertebrate embryo, reflecting its critical role during normal development and pregnancy. Elucidating an AOP for embryonic vascular disruption must consider the stepwise events underlying blood vessel patterning. Vascular development commences in the early embryo with in situ formation of nascent vessels from angioblasts, leading to a primary capillary plexus (vasculogenesis). After the onset of blood circulation, the primary vascular pattern is further expanded as new vessels sprout from pre-existing vessels (angiogenesis). Both processes, vasculogenesis and angiogenesis, are regulated by genetic signals and environmental factors dependent on anatomical region, physiological state, and developmental stage of the embryo. The developing vascular network is further shaped into a hierarchical system of arteries and veins, through progressive effects on blood vessel arborization, branching, and pruning (angioadaptation). These latter influences include hemodynamic forces, regional changes in blood flow, local metabolic demands and growth factor signals. Disruptions in embryonic vascular patterning-adaptation may result in adverse pregnancy outcomes, including birth defects, angiodysplasias and cardiovascular disease, intrauterine growth restriction or prenatal death. Some chemicals may act as potential vascular disrupting compounds (pVDCs) altering the expression, activity or function of molecular signals regulating blood vessel development and remodeling. Critical pathways involve receptor tyrosine kinases (e.g., growth factor-signaling), G-protein coupled receptors (e.g., chemokine signaling), and GPI-anchored receptors (e.g. uPAR system).
DESCRIPTION: This AOP focuses on the regulation and disruption of vasculogenesis-angiogenesis during embryonic development via disruption of the VEGF-signaling pathway. VEGFA binding to its cognate receptor (VEGFR2) triggers angiogenic sprouting, growth and fusion during early development, and in flow-sensing adaptation of vascular development during later development. VEGFR2 inhibition, the postulated molecular initiating event (MIE) for this AOP, may be invoked by effects on VEGFA production, mobility, or receptor binding, and by effects on VEGFR2 cellular expression, molecular function or post-receptor signal transduction pathways. Downstream key events (KE) include altered cell fate and behavior of 'endothelial tip cells' (exploratory behavior, cell migration) and endothelial 'stalk cells' (cell proliferation, apoptosis). KE relationships (KERs) leading to vascular insufficiency then involve local interactions with other cell types (stromal cells, macrophages), the extracellular matrix (ECM) and micro-physiology (hemodynamics, metabolism). Adverse outcomes (AO) would ultimately vary by anatomical region, organ system, gestational stage and state of the embryo, fetus or placenta when an MIE is invoked.
RELEVANCE and APPLICATION: Angiogenesis and vascular disruption is a broad concept. The intended use of this AOP in a regulatory context is the predictive toxicology of developmental hazards, especially for integrating data from high-throughput screening (HTS) assays into cell agent-based models for predicting dysmorphogenesis. As part of an integrated assessment of toxicity, this AOP can identify useful information for assessing adverse outcomes relevant to risk assessment and efficient use of resources for validation through predictive models linking developmental toxicity to vascular disruption. AOP-based computer models that simulate vascular development can usher-in new virtual screening techniques to predict what might happen to a developing embryo when exposed to chemicals across different dose-time-stage scenarios, including the range of effects and how cellular injury propagates across development.
AOP Development Strategy
Context
 https://aopwiki.org/wiki/index.php/File:KleinstreuerKnudsenAOPVascularDisruption.jpg
https://aopwiki.org/wiki/index.php/File:KleinstreuerKnudsenAOPVascularDisruption.jpg
Functionalization of the ToxCast pVDC predictive signature
The ToxCast putative Vascular Disrupting Chemicals (pVDC) signature will be described here and parts will be incorporated into the relevant Key Events sections.
The sectors of the ToxPI are color-represented by features from ToxCast HTS assays indicated by the target of the assays, the characteristics as follows.
Vascular cell adhesion molecule 1 (VCAM1): the pVDC signature aggregates assays from the BioMAP Systems Predictive Toxicology panel [Houck et al., 2009, Kunkel et al., 2004] focusing here on chemical disruption of endothelial VCAM1 expression following stimulation by cytokines-growth factors. This assay endpoint is an in vitro surrogate for inflammatory cell recruitment per endothelial dysfunction and has been probed across five different cell systems: 4H (HUVECs stimulated with IL-4 + histamine); 3C (HUVECs stimulated with IL-1β + TNFα + IFNϒ); CASM3C (primary human coronary artery smooth muscle cells stimulated with IL-1β + TNFα + IFNϒ); LPS (HUVECs co-cultured with monocytes and stimulated with bacterial endotoxin); and hDFCGF (human dermal fibroblasts stimulated with IL-1β + TNFα + IFNϒ and EGF + bFGF + PDGF-BB)[Knudsen and Kleinstreuer, 2011, Kleinstreuer et al., 2014].
Angiogenic cytokines and chemokines: the pVDC signature aggregates features for LPS-induced TNFα protein expression (see BioMAP descriptor above), nuclear factor-kappa B (NFkB) mediated reporter gene activation (Attagene; cis- configuration), and caspase 8 enzymatic activity (NovaScreen; inhibition or activation). TNFα is a proinflammatory cytokine that can promote angiogenesis indirectly through NFkB-mediated expression of angiogenic growth factors, or inhibit angiogenesis by direct effects on endothelial proliferation and survival. The pVDC signature also aggregates features for signaling activity of the pro-angiogenic cytokines interleukin-1 alpha (IL1a, a macrophage-derived activator of TNFα) and interleukin 6 (IL6). These cytokines act through the G-protein coupled receptors (GPCRs) IL1R and IL6R, respectively. CXCL8 (chemokine (C-X-C motif) ligand 8), formerly known as interleukin 8 (IL8), is angiogenic through its cognate GPCRs (CXCR1, CXCR2). In contrast to CXCL8, the chemokines CXCL9 (alias MIG, monokine induced by IFNϒ) and CXCL10 (alias IP10, interferon-inducible cytokine IP-10) are considered anti-angiogenic through their cognate receptor, CXCR3.
Angiogenic growth factors: FGFs and VEGFs exert their effects on endothelial cell proliferation, migration, and differentiation via specific binding to receptor tyrosine kinases VEGFR and FGFR. The pVDC signature has features for liganding VEGFR1, VEGFR2, and VEGFR3 based on receptor kinase activity (RTK, inhibition or activation) from the NovaScreen biochemical profile [Sipes et al. 2013] and for down-regulation of VEGFR2 expression in the 4H BioMAP system (HUVECs stimulated with IL-4 + histamine, B). VEGFR1 is a non-signaling VEGF-A decoy receptor that can be cleaved from the cell surface; VEGFR2 is the most important VEGF-A receptor and a master switch for developmental angiogenesis; and VEGFR3 is a VEGF-C receptor up-regulated by Notch signals. The pVDC signature includes features for the basic helix-loop-helix transcription factors Aryl Hydrocarbon Receptor (AhR) and Hypoxia Inducible Factor-1 alpha (HIF1a) that are upstream regulators of VEGF gene expression during ischemia or hypoxia. HIF1a and AhR are measured in reporter assays (Attagene). In addition to HIF1a and AhR, the pVDC signature has features for the estrogen receptor alpha (ERa), also a trans-activator of VEGF expression. This included human ERa binding activity (NovaScreen), ERa reporter trans-activation (Attagene) and ERE (estrogen responsive element) reporter cis-activation (Attagene).
Angiogenic sprouting: the ephrins (EFNA1 and EFNB2 in particular) couple VEGF signaling to angiogenic sprouting during early development of the embryonic vasculature (vasculogenesis, angiogenesis). The ToxCast pVDC signature included features for EPH-receptor tyrosine kinase biochemical activity (increased or decreased) for receptors EPHA1, EPHA2, EPHB1 and EPHB2 via their cognate cell membrane-anchored ligands (EFNAs). In contrast to the ephrin system, a number of chemicals had activity on diverse assays for urokinase-type plasminogen activator (uPA). That system, consisting of uPA (4 features) and its GPI-anchored receptor, uPAR (8 features) - both assayed in the BioMAP System [Kleinstreuer et al. 2014], functions in VEGFR2-induced changes to focal adhesion and extracellular matrix (ECM) degradation at the leading edge of endothelial cells during angiogenic sprouting. Binding of uPA to uPAR results in serine-protease conversion of plasminogen to plasmin that initiates a proteolytic cascade leading to degradation of the basement membrane and angiogenic sprouting. The uPA proteolytic cascade is suppressed by the serine protease inhibitor, endothelial plasminogen activator inhibitor type 1 (PAI1). The PAI1/uPA/uPAR assays report chemical effects on the system (up or down) across diverse cellular platforms: 4H, 3C, CASM3C, and hDFCGF noted above; BE3C (human bronchial epithelial cells stimulated with IL-1β + TNFα + IFNϒ); and KF3T (human keratinocytes + fibroblasts stimulated with IL-1β + TNFα + IFNϒ + TGF-β). The pVDC signature has features for thrombomodulin (THBD) and the thromboxane A2 (TBXA2) receptor that participate in the regulation of endothelial migration during angiogenic sprouting. THBD is a type I transmembrane glycoprotein that mediates regulator of uPA/uPAR and TBXA2 is an angiogenic eicosanoid generated by endothelial cyclooxygenase-2 (COX-2) following VEGF- or bFGF stimulation. THBD protein expression was monitored in the 3C and CASM3C BioMAP systems (up, down) and TBXA2 was assayed for ligand binding in the NovaScreen platform.
Endothelial cell migration and proliferation: the pVDC signature includes assays for human primary vascular cultures (endothelial and vascular smooth muscle cells). Assays for nuclear localization of beta-catenin (CTNB) are based on the principle that nuclear translocation activates pathways important for endothelial cell migration, proliferation and survival during capillary network formation in HUVEC cells [Muller et al. 2002; Masckauchan et al. 2005].
Vascular stabilization: The signature has features for transforming growth factor-beta 1 (TGF-b), which regulates vascular morphogenesis and integrity, and for Tie2 - a receptor tyrosine kinase activated by the angiopoietins (ANG1, ANG2) that function stabilize nascent vasculature. The pVDC signature has features for the anti-angiogenic phosphatases PTEN (phosphatase and tensin homolog), PTPN11 (tyrosine-protein phosphatase non-receptor type 11) and PTPN12, and endothelial-specific receptor tyrosine protein phosphatase beta (PTPRB). Matrix metalloproteinases (MMPs) 1/2/9 aggregate features on biochemical activity and cellular function of zinc-dependent endopeptidases MMP1, MMP2 and MMP9 that facilitate angiogenesis through ECM degradation by activated endothelial cells.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 305 | Inhibition, VegfR2 | Inhibition, VegfR2 |
| KE | 28 | Reduction, Angiogenesis | Reduction, Angiogenesis |
| KE | 110 | Impairment, Endothelial network | Impairment, Endothelial network |
| KE | 298 | Insufficiency, Vascular | Insufficiency, Vascular |
| AO | 1001 | Increased, Developmental Defects | Increased, Developmental Defects |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Inhibition, VegfR2 leads to Reduction, Angiogenesis | adjacent | High | High |
| Reduction, Angiogenesis leads to Impairment, Endothelial network | adjacent | High | Moderate |
| Impairment, Endothelial network leads to Insufficiency, Vascular | non-adjacent | Moderate | Low |
| Insufficiency, Vascular leads to Increased, Developmental Defects | non-adjacent | High | Moderate |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Conception to < Fetal | High |
| Pregnancy | High |
Taxonomic Applicability
Sex Applicability
Overall Assessment of the AOP
Domain of Applicability
The cardiovascular system is the first organ system to function in the vertebrate embryo, reflecting its critical role during organogenesis [Chan et al. 2002; Jin et al. 2005; Walls et al. 2008]. Blood vessel development commences in the early (sexually undifferentiated) embryo with de novo assembly of angioblasts into a primary capillary plexus (vasculogenesis). With the onset of blood circulation, the primary vascular pattern is further expanded as new vessels sprout from pre-existing vessels (angiogenesis). Both processes, vasculogenesis and angiogenesis, are developmentally regulated by genetic signals and environmental factors dependent on anatomical region, physiological state, and gestational age of the embryo-fetus [Shalaby et al. 1995; Patan, 2000; Jin et al. 2005; Knudsen and Kleinstreuer, 2011; Eberlein et al. 2021]. Disruption of embryonic vascular development is a potential framework for adverse outcome pathways (AOPs) in developmental toxicity [Knudsen and Kleinstreuer, 2011; Kleinstreuer et al. 2013; Saili et al. 2019; Zurlinden et al. 2020]. Developmental angiogenesis is supported by evidence in genetic models of abnormal vascularization leading to severe developmental phenotypes [Fong et al. 1995; Shalaby et al. 1995; Carmeliet et al. 1996; Maltepe et al. 1997; Abbott and Buckalew, 2000; Chan et al. 2002; Coultas et al. 2005; van den Akker et al. 2007; Eberlein et al. 2021]. This may include cell signals and responses driving formation of the primitive capillary network in the early embryo and extraembryonic membranes (vasculogenesis), the subsequent expansion and patterning of the embryonic and placental vasculature (angiogenesis), and its further stabilization, specialization, and remodeling during growth, organogenesis and differentiation. Additional evidence comes from dysmorphogenesis induced with known anti-angiogenic compounds across multiple vertebrate species (e.g., zebrafish, frog, chick, mouse, rat) [Therapontos et al. 2009; Jang et al. 2009; Rutland et al. 2009; Tal et al. 2014; Vargesson, 2015; Beedie et al. 2016; Ellis-Hutchings et al. 2017; Kotini et al. 2020] and human studies of malformations correlated with genetic and/or environmental factors that target vascular development [Husain et al. 2008; Gold et al. 2011; Vargesson and Hootnick, 2017]. An analysis of pharma compounds to which women of reproductive age were exposed identified vascular disruption as one of six potential mechanisms of teratogenesis [van Gelder et al. 2010]. This AOP is focused on disruption of ‘developmental angiogenesis’ from the perspective of dysmorphogenesis leading to severe developmental defects. Although uterine-decidual vascularization is critically important for healthy pregnancy outcomes, the emphasis here is the direct role on anatomical development of the embryo proper.
Essentiality of the Key Events
|
Event |
Direct Evidence |
Indirect Evidence |
No experimental evidence |
Contradictory experimental evidence |
|
MIE: Inhibition, VegfR2 |
***** |
|||
|
KE1: Reduction, Angiogenesis |
***** |
|||
|
KE2: Impairment, Endothelial network |
**** |
* |
||
|
KE3: Insufficiency, Vascular |
** |
*** |
||
|
AO: Increased, Developmental Defects |
*** |
** |
The Vascular Endothelial Growth Factor (VEGF) pathway is a critical regulatory system for assembly of embryonic blood vessels [Fong et al. 1995; Shalaby et al. 1995; Carmeliet et al. 1996; Ferrara, et al. 1996; Argraves et al. 2002; Hogan et al. 2004; Roberts et al. 2004; Chung and Ferrara, 2011; Shibuya, 2013; Chapell et al. 2016; Jin et al. 2017; Queisser et al. 2021]. The VEGF growth factors belong to the platelet-derived growth factor supergene family. VEGF-A, the major regulator for angiogenesis, binds receptor tyrosine kinases VEGFR-1 (Flt-1) and VEGFR2 (KDR/Flk-1) to regulate endothelial cell proliferation, survival, migration, tubular morphogenesis, and sprouting [Hogan et al. 2004; Douglas et al. 2009; Shibuya, 2013]. This pro-angiogenic effect is locally fashioned as VEGF gradients where the soluble VEGFR1 (sFlt-1) is released from the cell surface as an endogenous VEGF inhibitor that sets up VEGF-A corridors in the developing embryo [Roberts et al. 2004; Chappell et al. 2016]. Genetic studies have shown that perturbing the VEGF signaling system can invoke varying degrees of adverse consequences, ranging from congenital angiodysplasia to fetal malformations and embryolethality [Fong et al. 1995; Ferrara et al. 1996; Eshkar-Oren et al. 2015; Jin et al. 2017]. Filopodial sprouting of the endothelial cell tip (EC-tip) is the critical VEGFR2 responsive cell type specifically in this AOP [Belair et al. 2016a and 2016b]; however, other relevant cell types include: angioblasts (AB) as direct precursors to primary endothelial cells; endothelial ‘stalk’ cells (EC-stalk) proliferate in the wake of an angiogenic sprout; macrophage/microglial cells (MCs) release cytokines, chemokines, and growth factors; and stromal cells (SCs) are recruited to the nascent vascular wall for vessel stabilization. As such, the VEGF gradient/response system influences a multicellular dimension determined by cellular patterns of VEGF expression and processing (eg, MCs, SCs) and biochemical corridors set up by the extracellular matrix and the VEGFR1 decoy receptor (eg, EC-stalk). Evidence supporting an AOP for chemical disruption is available for thalidomide, estrogens, endothelins, dioxin, retinoids, cigarette smoke, and metals among other compounds [Kleinstreuer et al. 2011; Knudsen and Kleinstreuer, 2011; Shirinifard et al. 2013; Tal et al. 2014 and 2017; McCollum et al. 2017; Toimela et al. 2017; Mahony et al. 2018; Saili et al. 2019; Zurlinden et al. 2020]. Although not all compounds with developmental toxicity show an in vitro vascular bioactivity signature, many 'putative vascular disruptor compounds' (pVDCs) invoke adverse developmental consequences [Kleinstreuer et al. 2011 and 2013]. The molecular and cellular biology of human vascular development, stabilization and remodeling is amenable to in vitro assays with human cells [Bishop et al. 1999; Sarkanen et al. 2010; Kleinstreuer et al. 2014; Belair et al. 2016a and 2016b; Nguyen et al. 2017; Toimela et al. 2017; Pauty et al. 2018; van Duinen et al. 2019a and 2019b; Zurlinden et al. 2020], pluripotent stem cells induced to endothelial differentiation [Belair et al. 2015; Sinha and Santoro, 2018; Li et al. 2018; Galaris et al. 2021], and endothelial-specific reporter zebrafish [Tran et al. 2007; Shirinifard et al. 2013; Tal et al. 2014 and 2017; Beedie et al. 2017; McCollum et al. 2017]. An integrated portfolio of assays is thus available to cover many aspects of the angiogenic cycle and its ramifications during neurovascular development [Bautch and James, 2009; Eichman and Thomas, 2013; Saili et al. 2017; Uwamori et al. 2017; van Duinen et al. 2019; and Zurlinden et al. 2020]. Evidence is also available to support the essentiality of this AOP outside the embryo proper, such as uterine angiogenesis [Douglas et al. 2009; Araujo et al. 2021], placentation [Abbott and Bucklew, 2000; Chen and Zheng, 2014], and human pregnancies complicated by preeclampsia and small-for-gestational age infants [Andraweera et al. 2012].
Evidence Assessment
Weight of evidence for the MIE and AO are strong; the intermediate KEs have in some cases strong evidence but in other cases weaker evidence, due to the lack of quantitative information. the KERs are biologically plausible. Several manuscripts have been published recently that bolster weight of evidence [Belair et al. 2016; Nguyen et al. 2017; Tal et al. 2017; McCollum et al. 2017; Ellis-Hutchings et al. 2017; Saili et al. 2019; Zurlinden et al. 2020].
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Abbott, B. D. and A. R. Buckalew (2000). Placental defects in ARNT-knockout conceptus correlate with localized decreases in VEGF-R2, Ang-1, and Tie-2. Developmental dynamics : an official publication of the American Association of Anatomists 219(4): 526-5384. PMID: 11084652.
Andraweera, P. H., G. A. Dekker, S. D. Thompson and C. T. Roberts (2012). Single-nucleotide polymorphisms in the KDR gene in pregnancies complicated by gestational hypertensive disorders and small-for-gestational-age infants. Reproductive sciences (Thousand Oaks, Calif.) 19(5): 547-5545. PMID: 22344734.
Araujo Júnior, E., A. C. Zamarian, A. C. Caetano, et al. (2021). Physiopathology of late-onset fetal growth restriction. Minerva obstetrics and gynecology 73(4): 392-4084. PMID: 33876907.
Argraves, W. S., A. C. Larue, P. A. Fleming and C. J. Drake (2002). VEGF signaling is required for the assembly but not the maintenance of embryonic blood vessels. Developmental dynamics : an official publication of the American Association of Anatomists 225(3): 298-3043. PMID: 12412012.
Bautch, V. L. and J. M. James (2009). Neurovascular development: The beginning of a beautiful friendship. Cell adhesion & migration 3(2): 199-2042. PMID: 19363295.
Beedie, S. L., A. J. Diamond, L. R. Fraga, et al. (2017). Vertebrate embryos as tools for anti-angiogenic drug screening and function. Reproductive toxicology (Elmsford, N.Y.) 70: 49-59. PMID: 27888069.
Beedie, S. L., C. Mahony, H. M. Walker, et al. (2016). Shared mechanism of teratogenicity of anti-angiogenic drugs identified in the chicken embryo model. Scientific reports 6: 30038-30038. PMID: 27443489.
Belair, D. G., M. J. Miller, S. Wang, et al. (2016). Differential regulation of angiogenesis using degradable VEGF-binding microspheres. Biomaterials 93: 27-37. PMID: 27061268.
Belair, D. G., M. P. Schwartz, T. Knudsen and W. L. Murphy (2016). Human iPSC-derived endothelial cell sprouting assay in synthetic hydrogel arrays. Acta biomaterialia 39: 44554-44554. PMID: 27181878.
Belair, D. G., J. A. Whisler, J. Valdez, et al. (2015). Human vascular tissue models formed from human induced pluripotent stem cell derived endothelial cells. Stem cell reviews and reports 11(3): 511-5253. PMID: 25190668.
Bishop, E. T., G. T. Bell, S. Bloor, et al. (1999). An in vitro model of angiogenesis: basic features. Angiogenesis 3(4): 335-3444. PMID: 14517413.
Carmeliet, P., V. Ferreira, G. Breier, et al. (1996). Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 380(6573): 435-4396573. PMID: 8602241.
Chan, J., P. E. Bayliss, J. M. Wood and T. M. Roberts (2002). Dissection of angiogenic signaling in zebrafish using a chemical genetic approach. Cancer cell 1(3): 257-2673. PMID: 12086862.
Chappell, J. C., J. G. Cluceru, J. E. Nesmith, et al. (2016). Flt-1 (VEGFR-1) coordinates discrete stages of blood vessel formation. Cardiovascular research 111(1): 84-931. PMID: 27142980.
Chen, D. B. and J. Zheng (2014). Regulation of placental angiogenesis. Microcirculation (New York, N.Y. : 1994) 21(1): 15-251. PMID: 23981199.
Chung, A. S. and N. Ferrara (2011). Developmental and pathological angiogenesis. Annual review of cell and developmental biology 27: 563-584. PMID: 21756109.
Coultas, L., K. Chawengsaksophak and J. Rossant (2005). Endothelial cells and VEGF in vascular development. Nature 438(7070): 937-9457070. PMID: 16355211.
Douglas, N. C., H. Tang, R. Gomez, et al. (2009). Vascular endothelial growth factor receptor 2 (VEGFR-2) functions to promote uterine decidual angiogenesis during early pregnancy in the mouse. Endocrinology 150(8): 3845-38548. PMID: 19406950.
Eberlein, J., L. Herdt, J. Malchow, et al. (2021). Molecular and Cellular Mechanisms of Vascular Development in Zebrafish. Life (Basel, Switzerland) 11(10)10. PMID: 34685459.
Eichmann, A. and J. L. Thomas (2013). Molecular parallels between neural and vascular development. Cold Spring Harbor perspectives in medicine 3(1): a006551-a0065511. PMID: 23024177.
Ellis-Hutchings, R. G., R. S. Settivari, A. T. McCoy, et al. (2017). Embryonic vascular disruption adverse outcomes: Linking high throughput signaling signatures with functional consequences. Reproductive toxicology (Elmsford, N.Y.) 70: 82-96. PMID: 28527947.
Eshkar-Oren, I., S. Krief, N. Ferrara, et al. (2015). Vascular patterning regulates interdigital cell death by a ROS-mediated mechanism. Development (Cambridge, England) 142(4): 672-6804. PMID: 25617432.
Ferrara, N., K. Carver-Moore, H. Chen, et al. (1996). Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 380(6573): 439-4426573. PMID: 8602242.
Fong, G. H., J. Rossant, M. Gertsenstein and M. L. Breitman (1995). Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 376(6535): 66-706535. PMID: 7596436.
Galaris, G., J. H. Thalgott, E. Teston and F. P. G. Lebrin (2021). In vitro Three-Dimensional Sprouting Assay of Angiogenesis using Mouse Embryonic Stem Cells for Vascular Disease Modeling and Drug Testing. Journal of visualized experiments : JoVE(171)171. PMID: 34057453.
Gold, N. B., M. N. Westgate and L. B. Holmes (2011). Anatomic and etiological classification of congenital limb deficiencies. American journal of medical genetics. Part A 155A(6): 1225-12356. PMID: 21557466.
Hogan, K. A., C. A. Ambler, D. L. Chapman and V. L. Bautch (2004). The neural tube patterns vessels developmentally using the VEGF signaling pathway. Development (Cambridge, England) 131(7): 1503-15137. PMID: 14998923.
Husain, T., P. H. Langlois, L. E. Sever and M. J. Gambello (2008). Descriptive epidemiologic features shared by birth defects thought to be related to vascular disruption in Texas, 1996-2002. Birth defects research. Part A, Clinical and molecular teratology 82(6): 435-4406. PMID: 18383510.
Jang, G. H., I. S. Park, S. H. Lee, et al. (2009). Malachite green induces cardiovascular defects in developing zebrafish (Danio rerio) embryos by blocking VEGFR-2 signaling. Biochemical and biophysical research communications 382(3): 486-4913. PMID: 19364469.
Jin, D., D. Zhu, Y. Fang, et al. (2017). Vegfa signaling regulates diverse artery/vein formation in vertebrate vasculatures. Journal of genetics and genomics = Yi chuan xue bao 44(10): 483-49210. PMID: 29037991.
Jin, S. W., D. Beis, T. Mitchell, et al. (2005). Cellular and molecular analyses of vascular tube and lumen formation in zebrafish. Development (Cambridge, England) 132(23): 5199-520923. PMID: 16251212.
Kleinstreuer, N., D. Dix, M. Rountree, et al. (2013). A computational model predicting disruption of blood vessel development. PLoS computational biology 9(4): e1002996-e10029964. PMID: 23592958.
Kleinstreuer, N. C., R. S. Judson, D. M. Reif, et al. (2011). Environmental impact on vascular development predicted by high-throughput screening. Environmental health perspectives 119(11): 1596-160311. PMID: 21788198.
Kleinstreuer, N. C., J. Yang, E. L. Berg, et al. (2014). Phenotypic screening of the ToxCast chemical library to classify toxic and therapeutic mechanisms. Nature biotechnology 32(6): 583-5916. PMID: 24837663.
Knudsen, T. B. and N. C. Kleinstreuer (2011). Disruption of embryonic vascular development in predictive toxicology. Birth defects research. Part C, Embryo today : reviews 93(4): 312-3234. PMID: 22271680.
Kotini, M. P., F. Bachmann, J. Spickermann, et al. (2020). Probing the Effects of the FGFR-Inhibitor Derazantinib on Vascular Development in Zebrafish Embryos. Pharmaceuticals (Basel, Switzerland) 14(1)1. PMID: 33396726.
Li, S., C. W. Hsu, S. Sakamuru, et al. (2018). Identification of Angiogenesis Inhibitors Using a Co-culture Cell Model in a High-Content and High-Throughput Screening Platform. SLAS technology 23(3): 217-2253. PMID: 28922619.
Mahony, C., S. McMenemy, A. J. Rafipay, et al. (2018). CPS49-induced neurotoxicity does not cause limb patterning anomalies in developing chicken embryos. Journal of anatomy 232(4): 568-5744. PMID: 29023763.
Maltepe, E., J. V. Schmidt, D. Baunoch, et al. (1997). Abnormal angiogenesis and responses to glucose and oxygen deprivation in mice lacking the protein ARNT. Nature 386(6623): 403-4076623. PMID: 9121557.
McCollum, C. W., J. Conde-Vancells, C. Hans, et al. (2017). Identification of vascular disruptor compounds by analysis in zebrafish embryos and mouse embryonic endothelial cells. Reproductive toxicology (Elmsford, N.Y.) 70: 60-69. PMID: 27838387.
Nguyen, E. H., W. T. Daly, N. N. T. Le, et al. (2017). Versatile synthetic alternatives to Matrigel for vascular toxicity screening and stem cell expansion. Nature biomedical engineering 1. PMID: 29104816.
Patan, S. (2000). Vasculogenesis and angiogenesis as mechanisms of vascular network formation, growth and remodeling. Journal of neuro-oncology 50(44198): 44211-4421144198. PMID: 11245270.
Pauty, J., R. Usuba, I. G. Cheng, et al. (2018). A Vascular Endothelial Growth Factor-Dependent Sprouting Angiogenesis Assay Based on an In Vitro Human Blood Vessel Model for the Study of Anti-Angiogenic Drugs. EBioMedicine 27: 225-236. PMID: 29289530.
Queisser, A., E. Seront, L. M. Boon and M. Vikkula (2021). Genetic Basis and Therapies for Vascular Anomalies. Circulation research 129(1): 155-1731. PMID: 34166070.
Roberts, D. M., J. B. Kearney, J. H. Johnson, et al. (2004). The vascular endothelial growth factor (VEGF) receptor Flt-1 (VEGFR-1) modulates Flk-1 (VEGFR-2) signaling during blood vessel formation. The American journal of pathology 164(5): 1531-15355. PMID: 15111299.
Rutland, C. S., K. Jiang, G. A. Soff and C. A. Mitchell (2009). Maternal administration of anti-angiogenic agents, TNP-470 and Angiostatin4.5, induces fetal microphthalmia. Molecular vision 15: 1260-1269. PMID: 19572040.
Saili, K. S., J. A. Franzosa, N. C. Baker, et al. (2019). Systems Modeling of Developmental Vascular Toxicity. Current opinion in toxicology 15(1): 55-631. PMID: 32030360.
Saili, K. S., T. J. Zurlinden, A. J. Schwab, et al. (2017). Blood-brain barrier development: Systems modeling and predictive toxicology. Birth defects research 109(20): 1680-171020. PMID: 29251840.
Sarkanen, J. R., M. Mannerström, H. Vuorenpää, et al. (2010). Intra-Laboratory Pre-Validation of a Human Cell Based in vitro Angiogenesis Assay for Testing Angiogenesis Modulators. Frontiers in pharmacology 1: 147-147. PMID: 21779245.
Shalaby, F., J. Rossant, T. P. Yamaguchi, et al. (1995). Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 376(6535): 62-666535. PMID: 7596435.
Shibuya, M. (2013). Vascular endothelial growth factor and its receptor system: physiological functions in angiogenesis and pathological roles in various diseases. Journal of biochemistry 153(1): 13-191. PMID: 23172303.
Shirinifard, A., C. W. McCollum, M. B. Bolin, et al. (2013). 3D quantitative analyses of angiogenic sprout growth dynamics. Developmental dynamics : an official publication of the American Association of Anatomists 242(5): 518-5265. PMID: 23417958.
Sinha, S. and M. M. Santoro (2018). New models to study vascular mural cell embryonic origin: implications in vascular diseases. Cardiovascular research 114(4): 481-4914. PMID: 29385541.
Tal, T., C. Kilty, A. Smith, et al. (2017). Screening for angiogenic inhibitors in zebrafish to evaluate a predictive model for developmental vascular toxicity. Reproductive toxicology (Elmsford, N.Y.) 70: 70-81. PMID: 28007540.
Tal, T. L., C. W. McCollum, P. S. Harris, et al. (2014). Immediate and long-term consequences of vascular toxicity during zebrafish development. Reproductive toxicology (Elmsford, N.Y.) 48: 51-61. PMID: 24907688.
Therapontos, C., L. Erskine, E. R. Gardner, et al. (2009). Thalidomide induces limb defects by preventing angiogenic outgrowth during early limb formation. Proceedings of the National Academy of Sciences of the United States of America 106(21): 8573-857821. PMID: 19433787.
Toimela, T., O. Huttala, E. Sabell, et al. (2017). Intra-laboratory validated human cell-based in vitro vasculogenesis/angiogenesis test with serum-free medium. Reproductive toxicology (Elmsford, N.Y.) 70: 116-125. PMID: 27915012.
Tran, T. C., B. Sneed, J. Haider, et al. (2007). Automated, quantitative screening assay for antiangiogenic compounds using transgenic zebrafish. Cancer research 67(23): 11386-1139223. PMID: 18056466.
Uwamori, H., T. Higuchi, K. Arai and R. Sudo (2017). Integration of neurogenesis and angiogenesis models for constructing a neurovascular tissue. Scientific reports 7(1): 17349-173491. PMID: 29229920.
van den Akker, N. M., D. G. Molin, P. P. Peters, et al. (2007). Tetralogy of fallot and alterations in vascular endothelial growth factor-A signaling and notch signaling in mouse embryos solely expressing the VEGF120 isoform. Circulation research 100(6): 842-8496. PMID: 17332426.
van Duinen, V., W. Stam, V. Borgdorff, et al. (2019). Standardized and Scalable Assay to Study Perfused 3D Angiogenic Sprouting of iPSC-derived Endothelial Cells In Vitro. Journal of visualized experiments : JoVE(153)153. PMID: 31762444.
van Duinen, V., D. Zhu, C. Ramakers, et al. (2019). Perfused 3D angiogenic sprouting in a high-throughput in vitro platform. Angiogenesis 22(1): 157-1651. PMID: 30171498.
van Gelder, M. M., I. A. van Rooij, R. K. Miller, et al. (2010). Teratogenic mechanisms of medical drugs. Human reproduction update 16(4): 378-3944. PMID: 20061329.
Vargesson, N. (2015). Thalidomide-induced teratogenesis: history and mechanisms. Birth defects research. Part C, Embryo today : reviews 105(2): 140-1562. PMID: 26043938.
Vargesson, N. and D. R. Hootnick (2017). Arterial dysgenesis and limb defects: Clinical and experimental examples. Reproductive toxicology (Elmsford, N.Y.) 70: 21-29. PMID: 27773741.
Walls, J. R., L. Coultas, J. Rossant and R. M. Henkelman (2008). Three-dimensional analysis of vascular development in the mouse embryo. PloS one 3(8): e2853-e28538. PMID: 18682734.
Zurlinden, T. J., K. S. Saili, N. C. Baker, et al. (2020). A cross-platform approach to characterize and screen potential neurovascular unit toxicants. Reproductive toxicology (Elmsford, N.Y.) 96: 300-315. PMID: 32590145.
