
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 440
Title
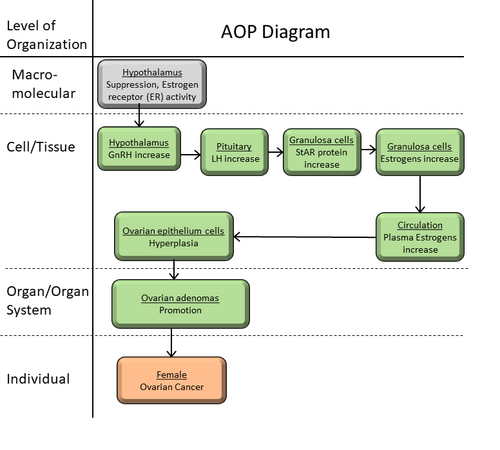
Hypothalamus estrogen receptors activity suppression leading to ovarian cancer via ovarian epithelial cell hyperplasia
Short name
Graphical Representation
Point of Contact
Contributors
- Kalyan Gayen
- Tridib Kumar Bhowmick
- Cataia Ives
Coaches
- Rex FitzGerald
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.104 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Suppression, Estrogen receptor (ER) activity | February 28, 2022 23:08 |
| Increased, secretion of GnRH from hypothalamus | September 16, 2017 10:17 |
| Increased, secretion of LH from anterior pituitary | December 03, 2016 16:37 |
| Increased, Steroidogenic acute regulatory protein (StAR) | March 01, 2022 07:00 |
| Increased, estrogens | March 01, 2022 06:58 |
| Increased, circulating estrogen levels | December 03, 2016 16:37 |
| Hyperplasia, ovarian epithelium | September 16, 2017 10:17 |
| Promotion, ovarian adenomas | December 03, 2016 16:37 |
| Promotion, Ovarian Cancer | March 11, 2023 01:37 |
| Suppression, Estrogen receptor (ER) activity leads to Increased, secretion of GnRH from hypothalamus | March 01, 2022 04:50 |
| Increased, secretion of GnRH from hypothalamus leads to Increased, secretion of LH from anterior pituitary | March 01, 2022 06:01 |
| Increased, circulating estrogen levels leads to Hyperplasia, ovarian epithelium | March 02, 2022 00:52 |
| Increased, secretion of LH from anterior pituitary leads to Increased, Steroidogenic acute regulatory protein (StAR) | March 01, 2022 06:43 |
| Hyperplasia, ovarian epithelium leads to Promotion, ovarian adenomas | December 03, 2016 16:38 |
| Increased, Steroidogenic acute regulatory protein (StAR) leads to Increased, estrogens | March 01, 2022 22:58 |
| Promotion, ovarian adenomas leads to Promotion, Ovarian Cancer | March 11, 2023 00:27 |
| Increased, estrogens leads to Increased, circulating estrogen levels | March 01, 2022 23:18 |
| Tamoxifen | November 29, 2016 18:42 |
| Raloxifene | November 29, 2016 18:42 |
| Clomiphene citrate (1:1) | February 26, 2022 23:24 |
Abstract
Malfunctioning of sex hormones (e.g., estradiol, estrone and progesterone) may result in ovarian cancer (Fooladi et al. 2020, Meehan and Sadar 2003). Exposure to endocrine-disrupting chemicals (EDCs) in the form of occupational usage of pesticides, fungicides, herbicides, plasticizers, cosmetics, etc. are the causes of ovarian cancer (Samtani, Sharma and Garg 2018). Some stressors molecules (e.g., clomiphene citrate, Tamoxifen, Toremifene) act on neuronal cell in the hypothalamus (molecular initiating event, MIE), where they inhibit hypothalamic Estrogen Receptors selectively and these chemicals increase the risk of ovarian cancer (McLemore et al. 2009). These stressors molecules stimulate the releasing of gonadotropin-releasing hormone (GnRH) from hypothalamic region of brain by the suppression of hypothalamic Estrogen Receptors. Subsequently, secretion of luteinizing hormone (LH) from pituitary becomes high(Cassidenti et al. 1992, Mungenast and Thalhammer 2014a, Tomao et al. 2014). This hormone regulates the synthesis of sex hormones (e.g., estrogens) at cellular level (Shoemaker et al. 2010a, Tomao et al. 2014). These sex hormones are primarily produced in the gonads through a series of enzyme-mediated reactions from cholesterol (precursor) and control through complex signalling pathway along hypothalamus – pituitary – gonadal (HPG) axis (Shoemaker et al. 2010a, Perkins et al. 2019). High estrogen level increases the risk of ovarian cancer via ovarian epithelial cell hyperplasia (McLemore et al. 2009, Tomao et al. 2014).
AOP Development Strategy
Context
Development and progression of certain types of cancer disease (e.g. ovarian cancer, breast cancer, prostate cancer etc.) is related with the hormonal levels in human. Lack of proper diagnosis at early stage of the disease increase the mortality rate of the cancer. Among many types of cancer ovarian cancer has the high mortality rate (~50%) due to the lack of proper diagnosis at early stage of the disease progression. Circulating levels of the steroidal sex hormones in conjunction with the gene expression is related with the progression of this disease. Some important sex hormones which are related with many cancer diseases include oestrogen, progesterone and testosterone. Oestrogen hormone mainly involved in female sex organ development, controlling of menstruation cycle etc. Progesterone also involved in controlling menstrual cycle, maintaining pregnancy and spermatogenesis. Testosterone hormone regulates sexual development, bone mass development, red blood cell production in male.
Strategy
In females, sexual hormone balance protects the ovaries from the tumor development. A number of researches revealed that molecular level perturbation leading towards sex hormone imbalance plays important role in the development of the ovarian cancer. Specifically, high level of estrogens (estradiol, estrone and progesterone) enhances the risk of ovarian cancer via ovarian epithelial cell hyperplasia. Estrones are regulated in hypothalamus and activity of estrogen receptors plays crucial role. Research reports reveled that suppression of the activity of estrogen receptors, leads to high level of estrogens in blood circulation via hypothalamus – pituitary – gonadal (HPG) axis. Therefore, suppression of the activity of estrogen receptors is considered as molecular initiating event (MIE). This MIE regulates several Key Events (Kes) along HPG axis for high level of estrogens in circulating blood that increases the risk of ovarian cancer via ovarian epithelial cell hyperplasia.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1046 | Suppression, Estrogen receptor (ER) activity | Suppression, Estrogen receptor (ER) activity |
| KE | 1047 | Increased, secretion of GnRH from hypothalamus | Increased, secretion of GnRH from hypothalamus |
| KE | 1050 | Increased, secretion of LH from anterior pituitary | Increased, secretion of LH from anterior pituitary |
| KE | 1972 | Increased, Steroidogenic acute regulatory protein (StAR) | Increased, Steroidogenic acute regulatory protein (StAR) |
| KE | 1973 | Increased, estrogens | Increased, estrogens |
| KE | 1076 | Increased, circulating estrogen levels | Increased, circulating estrogen levels |
| KE | 1052 | Hyperplasia, ovarian epithelium | Hyperplasia, ovarian epithelium |
| AO | 1053 | Promotion, ovarian adenomas | Promotion, ovarian adenomas |
| AO | 2092 | Promotion, Ovarian Cancer | Promotion, Ovarian Cancer |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Suppression, Estrogen receptor (ER) activity leads to Increased, secretion of GnRH from hypothalamus | adjacent | High | Not Specified |
| Increased, secretion of GnRH from hypothalamus leads to Increased, secretion of LH from anterior pituitary | adjacent | High | Moderate |
| Increased, secretion of LH from anterior pituitary leads to Increased, Steroidogenic acute regulatory protein (StAR) | adjacent | High | Moderate |
| Increased, Steroidogenic acute regulatory protein (StAR) leads to Increased, estrogens | adjacent | High | Moderate |
| Increased, estrogens leads to Increased, circulating estrogen levels | adjacent | High | Moderate |
| Increased, circulating estrogen levels leads to Hyperplasia, ovarian epithelium | non-adjacent | High | Not Specified |
| Hyperplasia, ovarian epithelium leads to Promotion, ovarian adenomas | non-adjacent | High | Not Specified |
| Promotion, ovarian adenomas leads to Promotion, Ovarian Cancer | non-adjacent | Moderate | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult, reproductively mature | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | High |
Overall Assessment of the AOP
Suppression, Estrogen receptor (ER) activity [Evidence- Strong]: There are number of reports available related to suppression of Estrogen receptor activity (ER) (Baez-Jurado et al., 2018; Cosman, 2003; Haskell, 2003; Ng et al., 2009; Kang et al., 2001; Roy et al., 1999; Marques P, 2018; Mungenast and Thalhammer, 2014b; Ghasemnejad-Berenji et al., 2020; J. H. Liu, 2020; Oride et al., 2020; Zhang et al., 2020; John F. Kerin et al., 1985b; The Practice Committee of the American Society for Reproductive Medicine, 2013; Moskovic et al., 2012 ; Bryan J. Herzog, 2020). Stressors act on neuronal cell in the hypothalamus, where it inhibits hypothalamic Estrogen Receptors selectively. A number of compounds or molecules (e.g. Clomiphene citrate, Tamoxifen, Toremifene etc.) are detected which show the modulation activity of estrogen receptor in brain leading to high GnRH pulses (Haskell, 2003; Cosman, 2003).
Increased, secretion of GnRH from hypothalamus[Evidence- Strong]: A number of evidencesare found by the researchesthat the increased secretion of gonadotropin-releasing hormone (GnRH)(Shander and Goldman, 1978; Tsourdi et al., 2009). Studies had shown that of inhibition of Estrogen receptor activity (ER) enhances the secretion of GnRH in human (Adashi et al., 1980; Bussenot et al., 1990; JOHN F KERIN et al., 1985a; Tan et al., 1996), rat and mice (Bharti et al., 2013; Kumar and Pakrasi, 1995; Zoeller and Young, 1988). Studies on human patient had shown the application of clomiphene is able to promote response of GnRH secretion (Goerzen et al., 1985; Tan et al., 1996).
Increased, secretion of LH from anterior pituitary [Evidence- Strong]: Good evidence may be acquired from different published articles for the increased secretion of LH increases from anterior pituitary (Plouffe and Siddhanti, 2001; Wright et al., 2012; Shoemaker et al., 2010b). It is also reported that increased secretion of the GnRH in hypothalamus leads to high levelofLH in human (John F Kerin et al., 1985a; Adashi et al., 1980; Bussenot et al., 1990), mice/rat.(Bharti et al., 2013; Kumar and Pakrasi, 1995; Botte et al., 1999) and cow (Fields et al., 2009).
Increased, Steroidogenic acute regulatory protein (StAR) [Evidence- Strong]: Steroidogenic acute regulatory protein (StAR) plays critical role in luteal steroidogenesis by controlling the transport of cholesterol from the outer to inner mitochondrial membrane(Wu et al., 2003; Shoemaker et al., 2010b).It had been reported that increase in LH level leads to increase StAR protein concentration in human(Tsang et al., 1980; Johnson and Bridgham, 2001; Murayama et al., 2012; Rekawiecki et al., 2005), rat(T. Liu et al., 2007; Martinat et al., 2005) and mice(Eacker et al., 2008; Tsuchiya et al., 2003).
Increased, estrogens [Evidence- Strong]: Aromatase is a key enzyme for estrogen formation in human tissues. In female, one of the important sites of estrogen enzyme synthesis is ovarian granulose cells(Holesh et al., 2017; Shoemaker et al., 2010b). Although ovarian aromatase enzyme expression in postmenopausal female is very low, high estrogen level is maintained in the blood through aromatase expression in other tissues. A number of researches had shown increased synthesis of StAR Protein increases the estrogen in ovarian granulosa cellsin human (Kiriakidou et al., 1996; Fang et al., 2016; Men et al., 2017), rat (Ronen-Fuhrmann et al., 1998; Nimrod, 1981) and fish (Kusakabe et al., 2002).
Increased, circulating estrogen levels [Evidence- Strong]: Researches had shown increased synthesis of estrogen in ovarian granulosa cells leads to maintain the high circulating estrogen levels in blood (Holesh et al., 2017; Shoemaker et al., 2010b).
Hyperplasia, ovarian epithelium [Evidence- High]: Ovarian surface is covered by the epithelium cells often called as ovarian mesothelium tissue. High evidence is available which supports that hyperplasia of the stromal cells might lead towards the hyperplasia of the ovarian epithelium tissue(Nyboe Andersen et al., 2008; Kang et al., 2001).
Promotion, ovarian adenomas[Evidence- Moderate]: Ovarian adenoma or cystadenoma is classified as benign tumor in the epithelial tissue. Evidence on the promotion of ovarian adenoma due to the hyperplasia in the ovarian epithelial tissue is available.
Promotion, ovarian cancer [Evidence- Strong]: Promotion of ovarian adenomas leads to the phenotype outcome of ovarian cancer at individual level (Johansson et al., 2022, Christine Stewart et al., 2019).
Domain of Applicability
Sex: This particular AOP is mainly applicable for the females. Sex hormone regulation in female is more complex compare to the male. Development and growth of the ovaries depend on the hormonal balance in the body. This hormonal balance in female changes often observed during the menstrual cycle and pregnancy. Imbalance in the hormonal levels leads to the abnormal function of the ovaries.Predominant form of estrogen (estradiol) hormone also found in male and plays critical role in sexual behavior and spermatogenesis. However, males more likely experiences imbalance in testosterone hormonelevels.
Life stage: This AOP is closer to the adult female. In particular the females (at the age of 45-55) going through the menopause are having greater chance of developing ovarian cancer compared to the young adult female. Young female undergoing through the hormonal therapy (usually estrogen) also having high risk of developing ovarian cancer. Risk factor of ovarian cancer is high in case of adult females who are taking ovulation stimulating drugs to increase fertility.
Taxonomic: For this AOP taxonomic domain is applicable to the different species like mice, rat, guinea pig and human.
Essentiality of the Key Events
In this AOP the essentiality of the proposed events are supported by a number of scientific works.
Kettel et al., had shown the treatment of seventeen females with clomiphene citrate with 150mg/day dose for 5 days enhance the estrogen levels. Analysis of the other hormones (follicle-stimulating hormone, luteinizing hormone, gonadotropin-releasing hormone) levels suggest the clomiphene citrate involved in the modulation in hormonal secretion at the hypothalamic site (Kettel et al., 1993).
Koch et al, had shown female rat injected with the clomiphene citrate (1-100 ng/kg) for 20 days increase the gonadotropin-releasing hormone (GnRH) release in the hypothalamus region (Koch et al., 1971).
Research by Kurosawa et al., on 293T cells (transfectable derivative of human embryonic kidney 293 cells, revealed that effect of clomiphene citrate depend on the concentration of the molecule. Clomiphene citrate at higher concentration (10-10 - 10-12 M) showed the estrogenic activity. However at higher concentration (10-6 - 10-12 M) no estrogenic activity was observed. Results of the study also suggest that clomiphene citrate either act as agonist or as an antagonist depends on the presence of 17β-estradiol (E2) receptor(Kurosawa et al., 2010).
Evidence Assessment
Overall assessment of the biological plausibility, empirical support and quantitative understanding of the KEs and KERs associated with this AOP shows that molecular mechanism or signaling pathway of tumor development in the female ovaries due to the suppression of estrogen receptors activities in the hypothalamus is still unclear.
Empirical evidence is available which shows the release of gonadotropin-releasing hormone (GnRH) depends on the concentration of the Selective Estrogen Receptors Modulator (SERM) compound (e.g. clomiphene citrate). However, molecular mechanism for the enhancement of GnRH by suppression of Estrogen receptor activity is poorly known.A number of researches had shown secretion of luteinizing hormone (LH) from anterior pituitary depends on the GnRH concentration or dose. Scientific reports have shown the both stimulatory and inhibitory effects on the GnRH secretion exhibited by the estradiol depending on the concentration of stressor (clomiphene) molecules and presence of types of receptors. The requirement of the GnRH dose for the secretary release of the LH in the different species varies widely.
A number of articles had shown that release of LH from the anterior pituitary regulates the steroidogenic function of cells by controlling the cholesterol transportation to the mitochondria. Biological plausibility of this event is very high as a number of studies have shown the similar results using different biological models (e.g. granulosa cells of adult female, bovine luteal cells, leydig cells of mice and rat etc.) in their study. Estradiol synthesis during menstrual cycle is governed via expression of StAR protein synthesis. Quantitative estimation of the event has been performed through indirect measurement (e.g. Northern blot analysis of mRNA collected from ovarian follicle granulosa cells). Therefore in many studies finding results are inconsistent. Circulating estrogen levels increases due to the increased estradiol synthesis and concentration controlled by the negative feedback loop of the other steroidal hormone synthesis.Biological evidence of tumor formation in the ovarian granulose cells due to the high circulating estrogen levels in the plasma is pretty high. High circulating estrogen drives the endometrial hyperplasia towards the progression of endometrial cancer.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Quantitative understanding in many KEs and KERs are available. However, exploitation of different biological models and use of different assay techniques provide incoherent results. Inconsistent results also have been mentioned in many KEs and KERs. A few assay techniques such as radioimmunoassay, radioreceptor assay, estrogen receptor binding assay etc. are sensitive enough to measure the concentration of a molecule at pictogram level. Some other techniques such as quantitative real time PCR (qRT-PCR), northern blot analysis of RNA also have been used for quantitative estimation of molecules at low concentration. Some indirect methods such as immunohistochemistry also have been employed for identification and quantitative estimation of biological molecule.
Considerations for Potential Applications of the AOP (optional)
This AOP provides the valuable informations regarding chemical messengers and different glands of endocrine system that are related for the risk and promotion of ovarian cancer. Linkage of qualitative and quantitative informations of different chemical messengers for the promotion of ovarian cancer would be beneficial for the cancer therapy and cancer drug development. Further, this AOP would be helpful to evaluate the hazardous long-term effects of the endocrine-disrupting chemicals and drugs which may lead towards the development of the ovarian cancer. This AOP would also help to regulate the uses of these stressor molecules which have inhibitory effects on the hypothalamic Estrogen Receptors. Understanding of the molecular events related with this AOP would help to screen these molecules and provide guideline to access the risk associated with these stressors.
References
Adashi, E., A. Hsueh & S. Yen (1980) Alterations induced by clomiphene in the concentrations of oestrogen receptors in the uterus, pituitary gland and hypothalamus of female rats. Journal of Endocrinology, 87, 383-392.
Baez-Jurado, E., M. A. Rincon-Benavides, O. Hidalgo-Lanussa, G. Guio-Vega, G. M. Ashraf, A. Sahebkar, V. Echeverria, L. M. Garcia-Segura & G. E. Barreto (2018) Molecular mechanisms involved in the protective actions of Selective Estrogen Receptor Modulators in brain cells. Front Neuroendocrinol, 52, 44-64.
Bharti, S., M. Misro & U. Rai (2013) Clomiphene citrate potentiates the adverse effects of estrogen on rat testis and down-regulates the expression of steroidogenic enzyme genes. Fertility and sterility, 99, 140-148. e5.
Botte, M., Y. Lerrant, A. Lozach, A. Berault, R. Counis & M. Kottler (1999) LH down-regulates gonadotropin-releasing hormone (GnRH) receptor, but not GnRH, mRNA levels in the rat testis. Journal of Endocrinology, 162, 409-415.
Bryan J. Herzog, H. M. T. N., Ayman Soubra, and Wayne J.G. Hellstrom (2020) Clomiphene Citrate for Male Hypogonadism and Infertility: An Updated Review. Androgens: Clinical Research and Therapeutics, 1, 62-69.
Bussenot, I., J. Parinaud, C. Clamagirand, G. Vieitez & G. Pontonnier (1990) Effect of clomiphene cirate on oestrogen secretion by human granulosa cells in culture. Human Reproduction, 5, 533-536.
Cassidenti, D. L., R. J. Paulson, R. A. Lobo & M. V. Sauer (1992) The synergistic effects of clomiphene citrate and human menopausal gonadotrophin in the folliculogenesis of stimulated cycles as assessed by the gonadotrophin-releasing hormone antagonist Nal-Glu. Hum Reprod, 7, 344-8.
Christine Stewart, Christine Ralyea & S. Lockwood (2019) Ovarian Cancer: An Integrated Review. Seminars in Oncology Nursing, 35, 151-156.
Cosman, F. (2003) Selective estrogen-receptor modulators. Clin Geriatr Med, 19, 371-9.
Eacker, S. M., N. Agrawal, K. Qian, H. L. Dichek, E. Y. Gong, K. Lee & R. E. Braun (2008) Hormonal regulation of testicular steroid and cholesterol homeostasis. Mol Endocrinol, 22, 623-35.
Fang, L., Y. Yu, R. Zhang, J. He & Y. P. Sun (2016) Amphiregulin mediates hCG-induced StAR expression and progesterone production in human granulosa cells. Sci Rep, 6, 24917.
Fields, S. D., B. L. Perry & G. A. Perry (2009) Effects of GnRH treatment on initiation of pulses of LH, LH release, and subsequent concentrations of progesterone. Domest Anim Endocrinol, 37, 189-95.
Fooladi, S., H. Akbari, M. Abolhassani, E. Sadeghi & H. Fallah (2020) Estradiol, des-acylated, and total ghrelin levels might be associated with epithelial ovarian cancer in postmenopausal women. medRxiv.
Ghasemnejad-Berenji, M., S. Pashapour & H. Ghasemnejad-Berenji (2020) Therapeutic potential for clomiphene, a selective estrogen receptor modulator, in the treatment of COVID-19. Medical Hypotheses, 145.
Goerzen, J., B. Corenblum & P. J. Taylor (1985) Potentiation of GnRH response by clomiphene citrate. J Reprod Med, 30, 749-52.
Haskell, S. G. (2003) Selective estrogen receptor modulators. South Med J, 96, 469-76.
Holesh, J. E., A. N. Bass & M. Lord (2017) Physiology, Ovulation.
Janson, P. O., L. Hamberger, J. E. Damber, B. Dennefors & F. Knutson (1980) Steroid production in vitro of a hilus cell tumor of the human ovary. Obstet Gynecol, 55, 662-5.
Johansson, Å., D. Schmitz, J. Höglund, F. Hadizadeh, T. Karlsson & W. E. Ek (2022) Investigating the Effect of Estradiol Levels on the Risk of Breast, Endometrial, and Ovarian Cancer. J Endocr Soc, 6, bvac100.
Johnson, A. L. & J. T. Bridgham (2001) Regulation of steroidogenic acute regulatory protein and luteinizing hormone receptor messenger ribonucleic acid in hen granulosa cells. Endocrinology, 142, 3116-24.
Kang, S. K., K. C. Choi, C. J. Tai, N. Auersperg & P. C. Leung (2001) Estradiol regulates gonadotropin-releasing hormone (GnRH) and its receptor gene expression and antagonizes the growth inhibitory effects of GnRH in human ovarian surface epithelial and ovarian cancer cells. Endocrinology. 2001 Feb;142(2):580-8. doi: 10.1210/endo.142.2.7982.
Kerin, J. F., J. H. Liu, G. Phillipou & S. Yen (1985a) Evidence for a hypothalamic site of action of clomiphene citrate in women. The Journal of Clinical Endocrinology & Metabolism, 61, 265-268.
Kerin, J. F., J. H. Liu, G. Phillipou & S. S. C. Yen (1985b) Evidence for a Hypothalamic Site of Action of Clomiphene Citrate in Women. The Journal of Clinical Endocrinology & Metabolism, 61, 265-268.
Kettel, L. M., S. J. Roseff, S. L. Berga, J. F. Mortola & S. S. Yen (1993) Hypothalamic-pituitary-ovarian response to clomiphene citrate in women with polycystic ovary syndrome. Fertil Steril. , 59, 532-38.
Kiriakidou, M., J. M. Mcallister, T. Sugawara & J. Strauss 3rd (1996) Expression of steroidogenic acute regulatory protein (StAR) in the human ovary. The Journal of Clinical Endocrinology & Metabolism, 81, 4122-4128.
Koch, Y., S. Dikstein, E. Superstine & F. G. Sulman (1971) THE EFFECT OF PROMETHAZINE AND CLOMIPHENE ON GONADOTROPHIN SECRETION IN THE RAT. Journal of Endocrinology, 49, 13-17.
Kumar, A. & P. L. Pakrasi (1995) Estrogenic and antiestrogenic properties of clomiphene citrate in laboratory mice. Journal of Biosciences, 20, 665-673.
Kurosawa, T., H. Hiroi, M. Momoeda, S. Inoue & Y. Taketani (2010) Clomiphene citrate elicits estrogen agonistic/antagonistic effects differentially via estrogen receptors alpha and beta. Endocr J, 57, 517-21.
Kusakabe, M., T. Todo, H. J. McQuillan, F. W. Goetz & G. Young (2002) Characterization and expression of steroidogenic acute regulatory protein and MLN64 cDNAs in trout. Endocrinology, 143, 2062-70.
Liu, J. H. (2020) Selective estrogen receptor modulators (SERMS): keys to understanding their function. Menopause-the Journal of the North American Menopause Society, 27, 1171-1176.
Liu, T., J. Wimalasena, R. L. Bowen & C. S. Atwood (2007) Luteinizing hormone receptor mediates neuronal pregnenolone production via up-regulation of steroidogenic acute regulatory protein expression. J Neurochem. , 100, 1329-39.
Marques P, S. K., George JT, et al. (2018) Physiology of GNRH and Gonadotropin Secretion. [Updated 2018 Jun 19]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279070/.
Martinat, N., P. Crepieux, E. Reiter & F. Guillou (2005) Extracellular signal-regulated kinases (ERK) 1, 2 are required for luteinizing hormone (LH)-induced steroidogenesis in primary Leydig cells and control steroidogenic acute regulatory (StAR) expression. Reprod Nutr Dev, 45, 101-8.
McLemore, M. R., C. Miaskowski, B. E. Aouizerat, L. M. Chen & M. J. Dodd (2009) Epidemiological and genetic factors associated with ovarian cancer. Cancer Nurs, 32, 281-8; quiz 289-90.
Meehan, K. L. & M. D. Sadar (2003) Androgens and androgen receptor in prostate and ovarian malignancies. Front Biosci, 8, d780-800.
Men, Y., Y. Fan, Y. Shen, L. Lu & A. N. Kallen (2017) The Steroidogenic Acute Regulatory Protein (StAR) Is Regulated by the H19/let-7 Axis. Endocrinology, 158, 402-409.
Moskovic, D. J., D. J. Katz, A. Akhavan, K. Park & J. P. Mulhall (2012 ) Clomiphene citrate is safe and effective for long-term management of hypogonadism. BJU International, 100, 1524 - 28.
Mungenast, F. & T. Thalhammer (2014a) Estrogen biosynthesis and action in ovarian cancer. Front Endocrinol (Lausanne), 5, 192.
--- (2014b) Estrogen biosynthesis and action in ovarian cancer. Front Endocrinol (Lausanne). 2014 Nov 12;5:192. doi: 10.3389/fendo.2014.00192. eCollection 2014.
Murayama, C., H. Miyazaki, A. Miyamoto & T. Shimizu (2012) Luteinizing hormone (LH) regulates production of androstenedione and progesterone via control of histone acetylation of StAR and CYP17 promoters in ovarian theca cells. Mol Cell Endocrinol, 350, 1-9.
Ng, Y., A. Wolfe, H. J. Novaira & S. Radovick (2009) Estrogen regulation of gene expression in GnRH neurons. Mol Cell Endocrinol. 2009 May 6;303(1-2):25-33. doi: 10.1016/j.mce.2009.01.016. Epub 2009 Feb 2.
Nimrod, A. (1981) On the synergistic action of androgen and FSH on progestin secretion by cultured rat granulosa cells: cellular and mitochondrial cholesterol metabolism. Molecular and cellular endocrinology, 21, 51-62.
Nyboe Andersen, A., A. Balen, P. Platteau, P. Devroey, L. Helmgaard & J. C. Arce (2008) Predicting the FSH threshold dose in women with WHO Group II anovulatory infertility failing to ovulate or conceive on clomiphene citrate. Hum Reprod. 2008 Jun;23(6):1424-30. doi: 10.1093/humrep/den089. Epub 2008 Mar 26.
Oride, A., H. Kanasaki, T. Tumurbaatar, T. Zolzaya, H. Okada, T. Hara & S. Kyo (2020) Effects of the Fertility Drugs Clomiphene Citrate and Letrozole on Kiss-1 Expression in Hypothalamic Kiss-1-Expressing Cell Models. Reproductive Sciences, 27, 806-814.
Perkins, E. J., K. Gayen, J. E. Shoemaker, P. Antczak, L. Burgoon, F. Falciani, S. Gutsell, G. Hodges, A. Kienzler, D. Knapen, M. McBride, C. Willett, F. J. Doyle & N. Garcia-Reyero (2019) Chemical hazard prediction and hypothesis testing using quantitative adverse outcome pathways. ALTEX, 36, 91-102.
Plouffe, L., Jr. & S. Siddhanti (2001) The effect of selective estrogen receptor modulators on parameters of the hypothalamic-pituitary-gonadal axis. Ann N Y Acad Sci, 949, 251-8.
Rekawiecki, R., M. Nowik & J. Kotwica (2005) Stimulatory effect of LH, PGE2 and progesterone on StAR protein, cytochrome P450 cholesterol side chain cleavage and 3beta hydroxysteroid dehydrogenase gene expression in bovine luteal cells. Prostaglandins Other Lipid Mediat, 78, 169-84.
Ronen-Fuhrmann, T., R. Timberg, S. R. King, K. H. Hales, D. B. Hales, D. M. Stocco & J. Orly (1998) Spatio-temporal expression patterns of steroidogenic acute regulatory protein (StAR) during follicular development in the rat ovary. Endocrinology, 139, 303-15.
Roy, D., N. L. Angelini & D. D. Belsham (1999) Estrogen Directly Represses Gonadotropin-Releasing Hormone (GnRH) Gene Expression in Estrogen Receptor-α (ERα)- and ERβ-Expressing GT1–7 GnRH Neurons1. Endocrinology, 140, 5045-5053.
Samtani, R., N. Sharma & D. Garg (2018) Effects of Endocrine-Disrupting Chemicals and Epigenetic Modifications in Ovarian Cancer: A Review. Reprod Sci, 25, 7-18.
Scirpa, P., D. Mango, A. Montemurro, F. Battaglia & L. Cantafio (1984) Androstenedione, 17 beta-estradiol and progesterone plasma levels in gonadotropins induction of ovulation. J Endocrinol Invest, 7, 357-62.
Shander, D. & B. Goldman (1978) Ovarian steroid modulation of gonadotropin secretion and pituitary responsiveness to luteinizing hormone-releasing hormone in the female hamster. Endocrinology, 103, 1383-93.
Shoemaker, J. E., K. Gayen, N. Garcia-Reyero, E. J. Perkins, D. L. Villeneuve, L. Liu & F. J. Doyle (2010a) Fathead minnow steroidogenesis: in silico analyses reveals tradeoffs between nominal target efficacy and robustness to cross-talk. BMC Systems Biology, 4, 89.
Shoemaker, J. E., K. Gayen, Natà l. Garcia-Reyero, E. J. Perkins, D. L. Villeneuve, L. Liu & F. J. Doyle (2010b) Fathead minnow steroidogenesis: in silico analyses reveals tradeoffs between nominal target efficacy and robustness to cross-talk. BMC Systems Biology, 4, 89.
Tan, S. L., J. Farhi, R. Homburg & H. S. Jacobs (1996) Induction of ovulation in clomiphene-resistant polycystic ovary syndrome with pulsatile GnRH. Obstet Gynecol, 88, 221-6.
The Practice Committee of the American Society for Reproductive Medicine (2013) Use of clomiphene citrate in infertile women: a committee opinion. Fertility and Sterility, 100, 341-348.
Tomao, F., G. Lo Russo, G. P. Spinelli, V. Stati, A. A. Prete, N. Prinzi, M. Sinjari, P. Vici, A. Papa, M. S. Chiotti, P. Benedetti Panici & S. Tomao (2014) Fertility drugs, reproductive strategies and ovarian cancer risk. J Ovarian Res, 7, 51.
Tsang, B. K., D. T. Armstrong & J. F. Whitfield (1980) Steroid biosynthesis by isolated human ovarian follicular cells in vitro. J Clin Endocrinol Metab. , 51, 1407-11.
Tsourdi, E., A. Kourtis, D. Farmakiotis, I. Katsikis, M. Salmas & D. Panidis (2009) The effect of selective estrogen receptor modulator administration on the hypothalamic-pituitary-testicular axis in men with idiopathic oligozoospermia. Fertil Steril, 91, 1427-30.
Tsuchiya, M., K. Inoue, H. Matsuda, K. Nakamura, T. Mizutani, K. Miyamoto & T. Minegishi (2003) Expression of steroidogenic acute regulatory protein (StAR) and LH receptor in MA-10 cells. Life Sciences, 73, 2855-2863.
Wright, D. J., J. N. Earnhardt, R. Perry, S. Bailey, B. Komm, D. R. Minck & M. A. Cukierski (2012) Carcinogenicity and hormone studies with the tissue-selective estrogen receptor modulator bazadoxifene. J Cell Physiol, 228, 724-33.
Wu, Q., S. Sucheta, S. Azhar & K. M. Menon (2003) Lipoprotein enhancement of ovarian theca-interstitial cell steroidogenesis: relative contribution of scavenger receptor class B (type I) and adenosine 5'-triphosphate- binding cassette (type A1) transporter in high-density lipoprotein-cholesterol transport and androgen synthesis. Endocrinology, 144, 2437-45.
Zhang, Z., J. W. Bartsch, J. Benzel, T. Lei, C. Nimsky & B. Voellger (2020) Selective estrogen receptor modulators decrease invasiveness in pituitary adenoma cell lines AtT-20 and TtT/GF by affecting expression of MMP-14 and ADAM12. Febs Open Bio, 10, 2489-2498.
Zoeller, R. T. & W. S. Young, 3rd (1988) Changes in cellular levels of messenger ribonucleic acid encoding gonadotropin-releasing hormone in the anterior hypothalamus of female rats during the estrous cycle. Endocrinology, 123, 1688-9.