
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 154
Title
Inhibition of Calcineurin Activity Leading to Impaired T-Cell Dependent Antibody Response
Short name
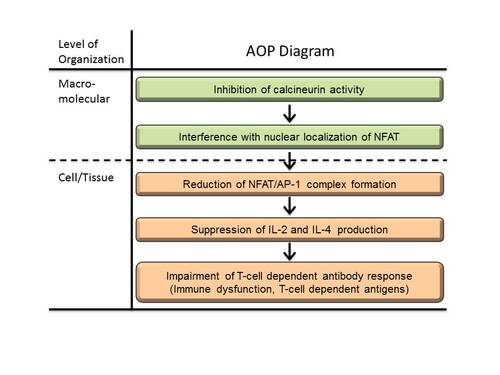
Graphical Representation
Point of Contact
Contributors
- Kiyoshi Kushima
- Takumi Ohishi
- Cataia Ives
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.38 | WPHA/WNT Endorsed |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Interference, nuclear localization of NFAT | July 02, 2021 04:16 |
| Reduction, NFAT/AP-1 complex formation | June 27, 2021 03:12 |
| Impairment, T-cell dependent antibody response | February 19, 2023 21:49 |
| Suppression, IL-2 and IL-4 production | June 27, 2021 03:28 |
| Inhibition, Calcineurin Activity | June 27, 2021 02:27 |
| Inhibition, Calcineurin Activity leads to Interference, nuclear localization of NFAT | June 27, 2021 04:47 |
| Interference, nuclear localization of NFAT leads to Reduction, NFAT/AP-1 complex formation | July 02, 2021 04:27 |
| Reduction, NFAT/AP-1 complex formation leads to Suppression, IL-2 and IL-4 production | June 27, 2021 08:06 |
| Suppression, IL-2 and IL-4 production leads to Impairment, T-cell dependent antibody response | June 27, 2021 08:08 |
| Tacrolimus (also FK506) | June 27, 2021 02:54 |
| Cyclosporin | May 18, 2017 08:31 |
| Pimecrolimus | March 20, 2020 02:29 |
| Gossypol | March 20, 2020 02:31 |
| Kaempferol | March 20, 2020 02:34 |
| Dodecylbenzene sulfonate | March 20, 2020 02:35 |
| Dibefurin | March 20, 2020 02:36 |
| Ascomycin | March 20, 2020 02:37 |
| 1,5-dibenzoyloxymethyl-norcantharidin | March 20, 2020 02:39 |
Abstract
Calcineurin (CN), a protein phosphatase, is known to impair immune function when its phosphatase activation is inhibited. The relationship between CN and immune functions is well understood, and immunosuppressants that work by inhibiting CN have been developed.
CN inhibitors (CNIs) inhibit CN phosphatase activity to suppress many kinds of immune functions and have been used to prevent hyper immune reactions such as rejection and graft versus host disease (GVHD), and treat autoimmune and allergic disorders such as psoriasis and atopic dermatitis. On the other hand, CNIs are reported to induce immunosuppression-derived adverse effects such as increased frequency and/or severity of infections and increased tumor incidences. CNIs might affect several T-cell derived immune functions to induce compromised host. Among the affected immune functions, T-cell dependent antibody response (TDAR) is an important factor to resist infections and thought to be the useful endpoint on evaluating immunotoxicity of chemicals; therefore, this AOP describes the linkage between the inhibition of CN activity and impairment of TDAR.
CN activity is inhibited when stressors bind to CN with their respective immunophilins, which interferes with the nuclear localization of nuclear factor of activated T cells (NFAT), a substrate of CN. As a result, the formation of functional NFAT complexes with activator protein-1 (AP-1) that bind at the site of IL-2, IL-4 and other T cell -derived cytokine promoters is reduced, thereby suppressing production of these cytokines. Among the affected cytokines from each of the helper T cell subsets, reduced production of IL-2 and IL-4 affects the proliferation and differentiation of B-cells to suppress TDAR.
We have identified a number of key events along this pathway and determined the key event relationships, based on which we have created an AOP for inhibition of CN activity leading to impaired TDAR.
Since CN is produced in a vast variety of species, this AOP might be applicable to many mammal species, including humans and rodents.
AOP Development Strategy
Context
Although there are other stressors that inhibit CN activity, this AOP is mainly based on an understanding of immunosuppression caused by the complex of FK506 and FKBP12 and cyclophilin and CsA, on which a significant body of scientific literature has been published.
We look forward to future amendments to this AOP with up-to-date information on other stressors, which will more clarify the linkage between inhibition of CN activity and impairment of TDAR.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 980 | Inhibition, Calcineurin Activity | Inhibition, Calcineurin Activity |
| KE | 979 | Interference, nuclear localization of NFAT | Interference, nuclear localization of NFAT |
| KE | 981 | Reduction, NFAT/AP-1 complex formation | Reduction, NFAT/AP-1 complex formation |
| KE | 1202 | Suppression, IL-2 and IL-4 production | Suppression, IL-2 and IL-4 production |
| AO | 984 | Impairment, T-cell dependent antibody response | Impairment, T-cell dependent antibody response |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | Moderate |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP
Inhibition of CN might induce suppression of cytokines production from all the T helper cell subsets as well as other immune functions of other immune cells. Suppression of cell-mediated immunity is involved in the pharmacology of preventing hyper immune reactions such as rejection and GVHD, and treatment of autoimmune and allergic disorders such as psoriasis and atopic dermatitis. On the other hand, CN inhibition might induce immunosuppression-derived adverse outcomes. One of the effects is increased frequency and/or severity of infections. Compromised host might be related with impairment of multiple immune functions; however, impaired TDAR deems to be usually related. Moreover, TDAR is the frequently used measurable endpoint in immunotoxicity testing according the ICH S8 or US EPA OPPTS 870.7800 immunotoxicity testing guideline. Therefore, the present AOP focus on CN inhibition-induced impairment of TDAR.
When stressors bind to calcineurin-A (CnA) with immunophilin, CN phosphatase activity is inhibited. Immunophilins are composed of a family of highly conserved proteins that have the ability to bind immunosuppressive drugs. This interfere with the nuclear localization of NFAT, the substrate for CN. As a result, the formation of functional NFAT/ AP-1 complexes that bind at the site of IL-2 , IL-4 and other cytokine promoters in each of the T helper cell subsets is reduced, thereby suppressing production of these cytokines. Among the affected cytokines TDAR is impaired mainly by the suppression of production of IL-2 and IL-4, which affect the proliferation and differentiation of B-cells to lower TDAR. We have identified a number of key events (KEs) along this pathway, and based on these key event relationships (KERs), created an AOP for inhibition of CN activity leading to impaired TDAR.
Since each KE involving MIE and AO is quantifiable, and shows similar dose responses with the CNIs in vitro, this AOP is useful for understanding immunosuppression due to inhibition of CN activity. In addition, each KER is based on sufficient scientific evidence and exhibits no contradiction with dose responses of adjacent KEs.
Since CN/NFAT system is conserved among vast variety of species and the function in immune system is common in at least human and mice, this AOP might be applicable to many mammalian species, including humans and rodents.
Domain of Applicability
The proposed AOP regarding inhibition of CN activity leading to impaired TDAR is not dependent on life stage, sex, or age. Since tacrolimus (FK506) ointment (Protopic) is approved for pediatric atopic dermatitis, the MOA for immunosuppression appears to be applicable to all life stages. The applicable state is considered supported by the draft FDA guidance for immunotoxicology that was recently issued (2020) indicating that “example of immunotoxicology testing could included TDAR assay” to address the concern of immunotoxicity in offspring in juvenile animal studies.
Since FK506 or CsA-induced outcomes in humans are mimicked by similar responses in a variety of animal models including non-human primates and rodents, immunosuppression induced by inhibition of CN activity is considered to occur across a variety of mammalian species.
In addition to the drugs, it is known that CN activity is suppressed by alkeylbenzene sulfonate (dodecylbenzene sulfonate) extracted from an acrylonitrile butadiene rubber (Ito et al. 2013)., suggesting that the proposed AOP would be applicable to non-pharmacological agents.
For the chemicals such as pesticide, the TDAR assay is also recommended in the US EPA OPPTS 870.7800 immunotoxicity testing guideline.
Essentiality of the Key Events
Essentiality is supported by several knockout animals as follows.
| Stage |
Essentiality |
Evidence |
Supported by literatures |
| MIE and later | CnA-KO mice | Strong |
The CN molecule consists of two regions, CnA and CnB, of which CnA exhibits phosphatase activity. In CnA-KO mice, T-cell proliferation in response to ovalbumin stimulation is lower than that for wild-type mice and is not complemented by normal antibody producing cells. In addition, when stimulated with ovalbumin, CnA-KO mice produce less IFN-γ, IL‑2, and IL‑4 than wild-type mice. However, primary antibody response in CnA-KO mice is normal in response to TNP-ovalbumin, which means that CnA deficiency affects only T cell-dependent antibody response (TDAR) (Zhang et al. 1996). |
| KE1 and later | NFAT-KO mice | Strong |
The following phenotypes are observed in NFAT-KO mice: moderate hyperproliferation with splenomegaly, moderately enhanced B- and T-cell responses, with bias towards Th2-cell response, decreased IFN-γ production in response to T-cell receptor (TCR) ligation, reduced proliferative responses by T cells, impaired repopulation of the thymus and lymphoid organs, impaired Th2- cell responses and IL-4 production, grossly impaired T-cell effector functions, profound defects in cytokine production and cytolytic activity, B-cell hyperactivity, impaired development of CD4 and CD8 single-positive cells, increased apoptosis of double-positive thymocytes, and mild hyperactivation of peripheral T cells. Therefore, the study of NFAT-KO mice shows that NFAT is involved in a wide range of immune responses, and some of these phenomenon are known to be regulated by CN. Suppression of T-cell-derived cytokines is noted both in CnA-knockout and NFAT-knockout mice, which indicates that the production of T-cell derived cytokines such as IL-2 and IL-4 is regulated by the CN-NFAT system (Macian, 2005). |
| Stressor | FKBP12-KO mice | Moderate | FK506 induces suppression of immune responses; however, there is no literature showing a relationship of a relationship between FKBP12 knockout and the immune system in the FKBP12-KO mouse model. Steric structure of FKBP12/FK506 complex is considered the key factor for inhibition of CN phosphatase activity, but not for the enzymatic activities of FKBP12. |
Evidence Assessment
Biological Plausibility
T-cell functions are mainly regulated by the CN-NFAT system and suppression of CN activity in T cells is known to induce multiple types of immunosuppression, including T cell-dependent antibody response (TDAR).
Experiments with T cells indicate that TCR stimulation brings about increases in intracellular concentrations of Ca2+ that trigger CN activity, thereby inducing nuclear localization of substrate NFAT per dephosphorylation. The localized NFAT forms complexes with activator protein 1 (AP-1) at the promoter sites of the T‑cell cytokine genes and induces production of the cytokines.
CN phosphatase activity is known to be inhibited by the formation of immunophilin-CN inhibitor (CNI) complexes, such as CsA/cyclophilin complexes or FK506/FK506-binding protein (FKBP) 12 complexes. Immunophilins are a general class of proteins that exhibit peptidyl-propyl isomerase (PPIase) activity, but there is no similarity between amino-acid sequences of the two classes of immunophilins. The three-dimensional structures of immunophilin complexes are essential to the inhibition of CN phosphatase activity, even though their enzymatic activities are not.
It is also known that one of the effects on immune function when CNI forms complexes with its respective immunophilin and inhibits CN activity is the suppression of IL-2 and other T-cell derived cytokine production. It is further known that inhibition of CN leads to suppression of TDAR because IL‑2 and IL‑4 mainly promote the proliferation, class switching, differentiation, and maturation of B-cells.
Furthermore, CN-NFAT also exists in B‑cells and it has been reported that CNIs do suppress production of certain cytokines from them. At the time of our review of the literature, however, we did not find any reports of a direct effect of CN inhibition on B‑cells, such as changes in proliferation, class switching, differentiation, or maturation of B‑cells.
Also, although CN-NFAT is known to exist in dendritic cells, natural killer T (NKT) cells, and other types of cells in which it regulates the expression of IL-2 receptors, there are no reports of effects on the production of T cell-dependent antibodies due to CNI-induced alteration in expression of IL-2 receptors in these cells.
CN-NFAT system-mediated immunosuppression is well understood based on the pharmacology of some CNI drugs (mostly FK506 and Cs A); therefore, AOP of CN inhibition-induced suppression of TDAR is useful for prediction of CN-mediated immunotoxicity.
| KER | KEup-KEdown | Evidence | Rationales supported by literatures |
| KER1 | CN inhibition to interference, NFAT nuclear translocation | Moderate |
CN phosphatase activation through TCR stimulation dephosphorylates NFAT, thereby promoting nuclear localization of NFAT. CN phosphatase activity in T cells could be inhibited by CNI/immunophilin complexes, thus interfering with dephosphorylation and nuclear localization of NFAT. The known mechanisms for inhibition of CN phosphatase activity by FK506, CsA, or other CNIs are initiated by the formation of complexes with their respective immunophilin species. Immunophilins are general classes of proteins that exhibit PPlase activity, but modification of these functions is unrelated to inhibition of CN activity and thus thought to arise in the molecular structure of the complexes (Schreiber and Crabtree 1992, Liu et al. 1993, Bierer et al. 1993, Bram et al. 1993, Rao et al. 1997, Liu et al. 1991). |
| KER2 | Interference, nuclear localization NFAT to reduction, NFAT/AP-1 complex formation | Strong |
CN activity dephosphorylates NFAT, thereby promoting its nuclear translocation. Nuclear-located NFAT binds with AP-1 at the promoter regions of the cytokine genes to promote T-cell cytokine production. Inhibition of dephosphorylation of NFAT by CNIs prevents nuclear export of NFAT and resultant binding with AP-1 at the promoter region of the T cell cytokine genes. NFAT has NLS and NES domains among and adjacent to the N-terminal region rich in SP motifs, and once the SP region is dephosphorylated, the NLS domain is exposed whereas the NES domain is covered, which leads to translocation of NFAT into the nucleus (Matsuda and Koyasu 2000). CNIs interference with the nuclear localization of NFAT in T cells leads to a reduction in the formation of NFAT/AP-1 complexes, thereby suppressing transcription of IL-2, IL-4, and a number of other cytokines (Maguire et al. 2013, Jain et al. 1992, Jain et al. 1993). |
| KER3 | Reduction, NFAT/AP-1 complex formation to suppression of IL-2 and IL-4 production | Strong |
NFAT/AP-1 complexes bind to the promoter regions of the cytokine genes, which promotes production of cytokines in T cells. Of these cytokines, IL-2 and IL-4 have a major role in promoting proliferation, maturation and class-switching of B cells, and development of TDAR. Reduction of NFAT/AP-1 complex formation in the nucleus due to inhibition CN activity by CNIs suppresses production of T-cell derived cytokines, including IL-2 and IL-4. T-5224, a selective c-Fos/AP-1 inhibitor, inhibits the DNA-binding activity of AP-1 in primary murine T cells. T-5224 also inhibits CD25 (one of IL-2 receptors) up-regulation, IL-2 production, and c-Fos DNA-binding activity in mice (Yoshida et al. 2015). Dexamethasone represses the IL-2 mRNA induction. glucocorticoid-induced leucine zipper (GILZ) is one of the most prominent glucocorticoid-induced genes, and inhibited the induction of the NFAT reporter and interferes with the AP-1 component of the NFAT/AP-1 complex. GILZ also inhibits the IL-2 promoter (Mittelstadt et al. 2001). Ursolic acid suppressed activation of three immunoregulatory transcription factors NF-kB, NFAT and AP-1. Treatment of lymphocytes and CD4+ T cells with ursolic acid inhibited secretion of IL-2 and IL-4 cytokines. Treatment of CD4+ T cells with ursolic acid suppressed mRNA level of IL-2. Treatment of lymphocytes with ursolic acid inhibited the upregulation of CD25 expression on T cells (Checker et al. 2012). |
| KER4 | Suppression of IL-2 and IL-4 production to impaired TDAR | Strong |
T cell-derived cytokines play important roles in TDAR. Among them, IL-2 promotes proliferation of B cells, and IL-4 affects maturation and class switching of B cells as well as proliferation. Inhibition of CN activity by CNIs is known to suppress production of multiple cytokine species from T cells. Of these cytokines and receptors, suppression of IL-2 and IL-4 production mainly leads to impairment of TDAR. Suppressed production of other cytokines due to inhibition of CN activity exhibits only minor effects, if any, on TDAR. CsA is known to be one of the calcineurin inhibitiors. CsA-treatment is reported to suppress the productions of IL-2 and IL-4 and result in reduced productions of antigen-specific IgM and IgG in cynomolgus monkeys (Gaida K. 2015). Dupilumab is known as anti-IL-4/13 receptor (IL-4/13R) antibody. Dupilumab (Dupixent) reduces productions of immunoglobulin (Ig) E and antigen specific IgG1 in mice (Sanofi K.K. 2018). It suggests that the blocking of IL-4 signaling by anti-IL-4/13R antibody results in the decrease in T cell dependent antibody production. Th2 cells produce cytokines including IL-4. Suplatast tosilate (IPD) is known as an inhibitor of the production of IL-4 and IL-5 in Th2 cells and reduces the production of antigen specific IgE in human cell culture and mice (Taiho Pharmaceutical 2013). These findings suggests that the reduction of IL-4 production by the inhibitor of Th2 cell cytokines results in reduced production of IgE and/or IgG1 through inhibitions of maturation, proliferation and class switching of B cells. IL-2 binds to IL-2 receptor (IL-2R) and acts on T cells. CD25 is one the of IL-2R. Basiliximab (Simulect) is known as anti-CD25 antibody. Basiliximab binds to IL-2R and blocks IL-2 signaling. Clinical transplantation study of basiliximab reveals decreases in rejections. On the other hand, basiliximab inhibits the activation of antigen specific T cells (Novartis Pharma 2016). FK506 and CsA suppress mRNA expression levels of cytokines in T cells including IL-2 and IL-4 that stimulate proliferation of B cells as well as B cell activation and class switching (Heidt et al, 2010). |
Empirical Support
| KER | KEup-KEdown | Evidence | Empirical support of KERs |
| KER1 | Inhibition, calcineurin activity leads to interference, nuclear localization of NFAT | Moderate |
CN phosphatase activity is inhibited with IC50 values of 0.5 nM (FK506) and 5nM (CsA) after 1 hours treatment (Fruman et al.1992). Concentration-dependent reduction of in vitro nuclear localization of NFAT was evident at concentrations from 0.1 nM (Jurkat T cells) or 10nM (human CD4+ T cells) and up to 1 μM (1000 nM) under the conditions of 2 hours treatment of tacrolimus (Maguire et al. 2013). Interference with translocation of NFAT to the nucleus is also detected using gel mobility shift assay to test nuclear extracts and cytoplasmic extracts, in which the examined concentration of FK506 was 10ng/mL (Flanagan et al. 1991). CN phosphatase activity and nuclear translocation of NFAT seems to be suppressed by CNIs at the similar ranges of doses and reaction times of 1 to 2 hours. |
| KER2 | Interference, nuclear localization of NFAT leads to reduction, NFAT/AP-1 complex formation | Strong |
Concentration-dependent reduction of in vitro nuclear localization of NFAT was evident at the concentration from 0.1 nM (Jurkat T cells) or 10nM (human CD4+ T cells) and up to 1 μM (1000 nM) under the conditions of 2 hours treatment (Maguire et al. 2013). Treatment of activated T cells with FK506 at 100ng/mL (124nM) or CsA at 500ng/mL (416nM) for 2 hours hinders the formation of functional NFAT/AP-1 in the nucleus (Flanagan et al. 1991). Gel mobility shift assays using Ar-5 human T cells stimulated with cross-linked anti-CD3 antibody showed that NFAT/AP-1 (cFos and Jun) complexes were found only in the nuclear extract with preexisting NFAT in the cytoplasm after T cell stimulation and that the NFAT/AP-1 complexes in the nucleus decreased after 2 hours treatment with CsA at 1µM (Jain et al. 1992). Decreased NFAT translocated to the nucleus, induced by FK506 at 100ng/mL (124nM) or CsA at 500ng/mL (416nM) after 2 hours treatment, hinders the formation of the functional NFAT/AP-1 complexes necessary to bind IL-2 promoters (Flanagan et al. 1991). NFAT/AP-1 complex formation was also reported to be inhibited by CNI (Rao et al. 1997). Quantitative data on NFAT/AP-1 complex formation in the nucleus is insufficient; however, inhibition of nuclear localization of NFAT and following NFAT/AP-1 complex formation in the nucleus are simultaneously detected by gel mobility shift assays at the concentration of FK506 within the range for inhibition of nuclear translocation of NFAT using imaging flowcytometry after 2 hours culture of T cells. |
| KER3 | Reduction, NFAT/AP-1 complex formation leads to suppression, IL-2 and IL-4 production | Moderate |
In NFATp- and NFAT4-deficient mice, cultured splenocytes bound anti-CD3 for 48 h indicates decreased production of Th1 cytokine including IL-2 (Ranger et al. 1998). In purified T cells from male C57BL/6J mice, T-5224 (a selective c-Fos/AP-1 inhibitor) inhibits the DNA-binding activity of AP-1 and CD25 (one of IL-2 receptors) up-regulation at 80 μg/mL, and IL-2 production in a dose-dependent manner from 40 to 80 μg/mL (Yoshida et al. 2015). In splenic lymphocytes stimulated with concanavalin A for 24 h in C57BL/6 mice, ursolic acid suppressed products of NF-kB, NFAT and AP-1 at 5 μM for 4 h. Secretion of IL-2 and IL-4 was inhibited in lymphocytes stimulated with concanavalin A for 24 h at concentrations of 0.5, 1 and 5 μM of ursolic acid, and lymphocytes and CD4+ T cells stimulated with anti-CD3/anti-CD28 mAb for 24 h at concentration of 5 μM of ursolic acid. In CD4+ T cells stimulated with anti-CD3/anti-CD28 mAb for 24 h, ursolic acid suppressed mRNA level of IL-2 at 5 μM for 4 h. In lymphocytes stimulated with concanavalin A for 24 h, ursolic acid inhibited CD25 expression at 5 μM for 4 h (Checker et al. 2012). Gel mobility shift assay revealed that treatment of activated T cells with FK506 at 100ng/mL (124nM) or CsA at 500ng/mL (416nM) for 2 hours hinders NFAT nuclear translocation and following formation of NFAT/AP-1 complexes in the nucleus (Flanagan et al. 1991). Preceding NFAT nuclear localization after T cell activation is suppressed with FK506 at the dose range of 0.01nM (Jarkat T cells) or 10nM (CD4+ T cells) to 1µM (Maguire et al. 2013), and NFAT nuclear localization and NFAT/AP-1 complex formation is shown to be strongly related (Jain et al. 1992, Jain et al. 1993). In CD3/PMA-activated human T cells, FK506 suppressed production of IL-2, IL-4, and IFN-γ at the concentrations of 1.2 to 12.5 nM after 22 to 24 hours culture as well as inhibited expression of IL-2, IL-4, and IFN-γ mRNA at 10 nM (Dumont et al. 1998). Treatment with CsA completely eliminated detectable IL-2 release from 3A9 T cells co-cultured with antigen-bearing Ch27 B cells with an IC25 and IC50 for IL-2 production of 1.19 nM and 1.99 nM. Treatment with other immunosuppressant compounds (dexamethasone, azathioprine, methotrexate, benzo(a)pyrene and urethane) also resulted in decreased IL-2 release from stimulated 3A9 T cells at non-cytotoxic concentrations. Urethane, a weakly immunosuppressive chemical, was least potent in the assay, with an IC25 and IC50 for IL-2 secretion of 4.24 mM and 13.26 mM (D.M. Lehmann. et al. 2018). In female B6C3F1 mice, 1,2:5,6-dibenzanthracene exposure reduced production of IL-2 in spleen cell culture supernatants after in vitro stimulation with Concanavalin A or lipopolysaccharide (Donna, C. et al. 2010). Treatment with CsA at 50 mg/kg BID via oral gavage or 2C1.1 (a fully human anti-ORAI1 monoclonal antibody) at 25 mg/kg single IV resulted in reduction of IL-2, IL-4, IL-5, and IL-17 cytokine production from PMA/ionomycin stimulation of whole blood in the cynomolgus monkey (Kevin, G. et al. 2014). In male CD-1 mice, chronic psychosocial stress (types of social outcome occurred: residents becoming subordinates) reduced IL-2 release in response to keyhole limpet hemocyanine (Alessandro, B. et al. 2003). Therefore, concentration of CNI needed for inhibition of NFAT/AP-1 complex formation in the nucleus is higher than that for inhibition of IL-2 and IL-4 production. A time lag is found between the two KEs; 2 hours for KE2 and 22 to 48 hours for KE3. |
| KER4 | Suppression, IL-2 and IL-4 production leads to Impairment, T-cell dependent antibody response | Strong |
Cynomolgus monkeys treated wth CsA at 50 mg/kg BID for 24 days suppression of IL-2, IL-4 and sheep red blood cell (SRBC)-specific IgM and IgG (Gaida K. 2015). In the allergen-induced pneumonia model in mice, dupilumab (anti-IL-4/13R antibody) reduced productions of IgE and antigen specific IgG1 at 25 mg/kg of twice weekly subcutaneous administration for 4weeks (Sanofi K.K. 2018). In mice immunized with dinitrophenyl antigen by i.p. injection, suplatast tosilate (an inhibitor of the production of cytokines such as IL-4 and IL-5 on Th2 cell) reduced productions of antigen specific IgE at 10, 20, 50 and 100 mg/kg of oral administration for 5 days (Taiho Pharmaceutical 2013). In human cell culture immunized with Japanese cedar antigen, suplatast tosilate reduced productions of antigen specific IgE at the concentration of 10 μg/mL for 10 days (Taiho Pharmaceutical 2013). 1,2:5,6-dibenzanthracene single adoministration suppressed production of IL-2 and total IgG antibody in mice at the dose levels of 3 and 30 mg/kg(Donna, C. et al. 2010). In male CD-1 mice, chronic psychosocial stress (types of social outcome occurred: residents becoming subordinates) for 21 days reduced IL-2 release in response to KLH (keyhole limpet hemocyanine) and decrease in anti-KLH IgG(Alessandro, B. et al. 2003). FK506 or CsA suppressed production of IL-2 in mouse mixed lymphocyte reaction (MLR) at 0.1 to 10 nM of FK506 and 10 to 100 nM of CsA as well as in human MLR at 0.1 to 10 nM of FK506 and 10 to 100 nM of CsA (Kino et al. 1987a). In CD3/phorbol 12-myristate-13-acetate-activated human T cells, FK506 suppressed production of IL-2, IL-4 and Interferon (IFN)-γ at the concentrations of 1.2 to 12.5 nM as well as inhibited expression of IL-2, IL-4 and IFN-γ mRNA at the concentrations of 10 nM. (Dumont et al. 1998). Rats were treated with FK506 for over four weeks and immunized with keyhole limpet hemocyanine (KLH), after which serum concentration of anti-KLH IgM and IgG reduced at the dose levels of 3 mg/kg/day (Ulrich et al. 2004). Mice were treated with FK506 or CsA for 4 days, and immunized with sheep red blood cells (SRBC), after which antigen-specific plaque-forming splenocytes were reduced at dose levels of 3.2, 10, 32 and 100 mg/kg of FK506 or 32 and 100 mg/kg of CsA (Kino et al. 1987b). After 9-day culture of B cells and non-pre-activated T cell stimulationwith FK506 or CsA, the levels of IgM and IgG in the culture supernatant were reduced at 0.3 and 1.0 ng/mL (0.37 and 1.24 nM) of FK506 or 50 and 100 ng/mL (41 and 83nM) of CsA (Heidt et al, 2010). After 4-day culture of SKW6.4 cells (IL-6-dependent IgM-secreting human B-cell line) and anti-CD3/CD28 stimulated PBMC culture supernatant with FK506 or CsA, the level of IgM in the culture supernatant was reduced at the concentrations of 0.01 to 100 ng/mL (0.01 to 124 nM) of FK506 or 0.1 to 1000 ng/mL (0.08 to 832 nM) of CsA (Sakuma et al, 2001). In vitro suppression of T-cell–derived cytokines and T-cell–dependent antibody production or antibody production after polyclonal T-cell stimulation showed similar dose responses to CNIs. Time gaps were found, however, between these two events, which showed earlier onset of cytokine production and delayed onset of antibody production. |
Based on these findings of empirical support, each KE involving MIE and AO except for KE2 shows similar dose responses to the CNIs in vitro; however, culture time lag is noted, in that, 1 hour for MIE, 2 hours for KE1 and KE2, 22 to 24 hours for KE3 and more than days for AO.
Known Modulating Factors
Quantitative Understanding
KER1:
No literature is available showing a clear quantitative relationship between the inhibition of CN phosphatase activity and nuclear translocation of NFAT; however, the dose responses of CN phosphatase activity and nuclear translocation of NFAT to CNI deem to be the same.
KER2:
Gel mobility shift assay of activated T cells showed that NFAT/AP-1 complexes are only found in nuclear extract, which indicates a strong relationship between the nuclear translocation of NFAT and simultaneous complex formation with AP-1 in the nucleus. CNI treatment clearly suppresses the complex formation of nuclear located NFAT and AP-1 in the nucleus, which also shows the solid relationship between these adjacent two KEs although quantitative data on suppressed NFAT/AP-1 complex formation is insufficient (Flaganan W.M. et al. 1991).
KER3:
The quantitative relationship between the decreased formation of NFAT/AP-1 complexes and the production of IL2/IL-4 formation induced by CNIs has not been reported.
However, as mentioned in the empirical support, nuclear localization of NFAT is strongly related to NFAT/AP-1 complex formation in the nucleus based on the fact that these two events are detected simultaneously by gel mobility shift assay, and the dose responses of IL2/IL-4 production and nuclear translocation of NFAT inhibited by CNI are similar; therefore, dose ranges of CNI in the inhibitions of IL2/IL-4 production and NFAT/AP-1 complex formation in the nucleus might also be the same.
In addition, T-5224 and ursolic acid inhibit AP-1 DNA binding activity or production of NF-κB, NFAT and AP-1, respectively, and both suppress the IL-2 and/or IL-4 production with dose dependent manner including the doses of inhibiting NFAT-AP-1 system (Yoshida et al. 2015, Checker et al. 2012).
KER4:
Cynomolgus monkeys treated wth CsA at 50 mg/kg BID for 24 days suppression of IL-2, IL-4 and sheep red blood cell (SRBC)-specific IgM and IgG (Gaida K. 2015).
Inhibition of IL-4 production in mice treated with oral administration of suplatast tosilate suppresses antigen-specific IgE production in a dose-dependent manner (Taiho Pharmaceutical 2013). In the inhibition of IL-4 production in human cell culture by suplatast tosilate at the concentration of 10 μg/mL for 10 days, antigen specific IgE production was suppressed from 56 to 72% and IL-4 production was suppressed from 58 to 76% (Taiho Pharmaceutical 2013).
As for IL-2 and antibody production, in vitro T-cell-induced polyclonal B cell activation to produce antibody was inhibited with anti-IL-2 and anti-IL-2R antibodies. T (Owens T, 1991).
In the human T-B cell co-culture stimulated with anti-CD3 monoclonal antibody, CNIs of FK506 and CsA lowered the levels of T-cell cytokines including IL-2 and IL-4 and inhibited IgM and IgG productions with a dose-dependent manner (Heidt S. 2010).
These results show the quantitative relationships between the inhibition of IL-4 or IL-2 by specific antibodies or CNI and suppression of antibody production.
Considerations for Potential Applications of the AOP (optional)
The ICH S8 guideline, which covers immunosuppression of small molecule drugs, determines the need for immunotoxicity studies by comprehensively evaluating the findings of pharmacology, changes in the immune system in repeated-dose toxicity studies, and other factors using a Weight of Evidence approach. If there is concern about immunotoxicity, the presence or absence of immunotoxicity should be determined using an in vivo test system capable of assessing the functional changes of predicted immunotoxic target cells. If immunotoxicity is observed, additional studies including in vitro assays or clinical evaluation should be considered to assess the risk of immunotoxicity in humans. Because TDAR involves many immune cell populations, including T cells, B cells, and antigen-presenting cells, evaluation of TDAR is recommended when there is concern about immunotoxicity but the immunotoxic target cells are unclear. The S8 guidelines list KLH, SRBC, and tetanus toxin as antigens for TDAR.
The draft FDA immunotoxicity testing guidance (2020) covers immunosuppressive and immunostimulatory drugs and biologics; evaluating immunosuppressive drugs in the draft FDA guidance is similar to that in the S8 guideline, with in vivo TDAR assays recommended when toxic target cells are unknown. The draft guidance states that TDAR assays using KLH as an antigen have been established in mice, rats, dogs, minipigs, and cynomolgus monkeys, but the use of SRBC and tetanus toxin as antigens is also acceptable.
For the assessment for pesticides, US EPA OPPTS 870.7800 immunotoxicity testing guideline recommends TDAR using SRBC. The REACH guideline does not provide for immunotoxicity testing, but it provides triggers for conducting immunotoxicity testing.
The WHO/IPSS Immunotoxicity Risk assessment Guidance (2012) describes a strategy for assessing five categories of immunotoxicity risks, including immunosuppression. For risk assessment of immunosuppression, it calls for identification of immunosuppression risks, prediction of pathogenesis that may occur, and consideration of safety margins based on the WoE approach from human findings, infection resistance tests, immune function tests, general immune system assays, histopathological findings and organ weights in general toxicity studies, and hematological data.
The evaluation of immunotoxicity in F1 animals in the OECD Guidelines for Extended First Generation Reproductive and Developmental Toxicity Studies (TG443) requires that PFC and ELSA assays to measure primary IgM antibody production by TDAR using T-cell dependent antigens (SRBC, KLH, etc.) be performed. Furthermore, if changes are observed, the significance of the changes should be examined by comprehensively evaluating other data.
The outcomes of immunosuppression are susceptibility to infection and tumorigenesis, and the FDA guidance requires that immunosuppressive drugs be evaluated for carcinogenic risk using WoE approach based on the results of carcinogenicity and immunotoxicity studies. Meanwhile, the ICH S1B(R1) Draft Step 2 Guidelines for Carcinogenicity Testing calls for evaluation of carcinogenicity by WoE approach instead of rat carcinogenicity testing, because rodent carcinogenicity test models are less capable of detecting carcinogenicity. On the other hand, it is difficult to define susceptibility to infection as a measurable AO with a clear mechanism, because immune responses vary among pathogens. In fact, many immunotoxicity guidelines require that the risk of immunotoxicity be identified and assessed by evaluating immune functions.
In AOP154, it was difficult to define susceptibility to infection as an AO for the AOP154, so TDAR, which is recommended as an indicator of immunosuppresoin in many guidelines, was used as an AO. It is expected that several AOPs with TDARs as AOs will be developed, and based on these AOPs, it may be possible to develop an IATA to assess the risk of immunotoxicity characterized by TDARs.
References
- Alessiani, M., Kusne, S., Martin, M., Jain, A., Abu-Elmagd, K., Moser, J., Todo, S., Fung, J. and Starzl, T. (1991). Transplantation proceedings 23 (1 Pt 2): 1501-3.
- Alessandro B, Paola S, Alberto E. Paneraic, Tiziana P,Paola Palanzaa and Stefano P(2003). Journal of Neuroimmunology 141: 58–64
- Antiga, E., Volpi, W., Torchia, D., Fabbri, P. and Caproni, M. (2011). Clinical and experimental dermatology 36 (3): 235-41.
- Beals, C.R., Clipstone, N.A., Ho, S.N. and Crabtree, G.R. (1997). Genes & development 11 (7): 824-34.
- Bhattacharyya, S., Deb, J., Patra, A.K., Thuy Pham, D.A., Chen, W., Vaeth, M., Berberich-Siebelt, F., Klein-Hessling, S., Lamperti, E.D., Reifenberg, K., Jellusova, J., Schweizer, A., Nitschke, L., Leich, E., Rosenwald, A., Brunner, C., Engelmann, S., Bommhardt, U., Avots, A., Müller, M.R., Kondo, E. and Serfling, E. (2011). The Journal of experimental medicine 208 (4): 823-39.
- Bierer, B.E., Holländer, G., Fruman, D. and Burakoff, S.J. (1993). Current opinion in immunology 5 (5): 763-73.
- Boussiotis, V.A., Nadler, L.M., Strominger, J.L. and Goldfeld, A.E. (1994).Proceedings of the National Academy of Sciences of the United States of America 91 (15): 7007-11.
- Bram, R.J., Hung, D.T., Martin, P.K., Schreiber, S.L. and Crabtree, G.R. (1993). Molecular and cellular biology 13 (8): 4760-9.
- Cameron, A.M., Nucifora, F.C. Jr., Fung, E.T., Livingston, D.J., Aldape, R.A., Ross, C.A. and Snyder, S.H. (1997). The Journal of biological chemistry 272 (44): 27582-8.
- Checker, R., Sandur, SK., Sharma, D., Patwardhan, RS., Jayakumar, S., Kohli, V., Sethi, G., Aggarwal, BB. and Sainis, KB. (2012). PLoS One. 7(2): e31318.
- Chung, B.H., Kim, K.W., Yu, J.H., Kim, B.M., Choi, B.S., Park, C.W., Kim, Y.S., Cho, M.L. and Yang, C.W. (2014). Transplant immunology 30 (4): 159-67.
- Cohan, V.L., Undem, B.J., Fox, C.C., Adkinson, N.F. Jr., Lichtenstein, L.M. and Schleimer, R.P. (1989). The American review of respiratory disease 140 (4): 951-4.
- Conboy, I.M., Manoli, D., Mhaiskar, V., and Jones, P.P. (1999). Proceedings of the National Academy of Sciences of the United States of America 96 (11):6324-9.
- Donna C. S, Matthew J. S and Kimber L. W Jr. (2010). Journal of Immunotoxicology, 7:3, 219-231
- Dumont, F.J., Staruch, M.J., Fischer, P., DaSilva, C. and Camacho, R. (1998). Journal of immunology 160 (6): 2579-89.
- Ekberg, H., Tedesco-Silva, H., Demirbas, A., Vítko, S., Nashan, B., Gürkan, A., Margreiter, R., Hugo, C., Grinyó, J.M., Frei, U., Vanrenterghem, Y., Daloze, P. and Halloran, P.F.; ELITE-Symphony Study. (2007). The New England journal of medicine 357 (25): 2562-75.
- Ekberg, H., Bernasconi, C., Tedesco-Silva, H., Vítko, S., Hugo, C., Demirbas, A., Acevedo, R.R., Grinyó, J., Frei, U., Vanrenterghem, Y., Daloze, P. and Halloran, P. (2009). American journal of transplantation 9 (8): 1876-85.
- Flanagan, W.M., Corthésy, B., Bram, R.J. and Crabtree, G.R. (1991). Nature 352 (6338): 803-7.
- Foletta, V.C., Segal, D.H. and Cohen, D.R. (1998). Journal of leukocyte biology 63 (2): 139-52.
- Fruman, D. A., Klee, C. B., Bierer, B. E. and Burakoff, S. J. (1992). Proceedings of the National Academy of Sciences of the United States of America. 89(9):3686-90.
- Fruman, D.A., Bierer, B.E., Benes, J.E., Burakoff, S.J., Austen, K.F. and Katz, H.R. (1995). Journal of immunology 154 (4): 1846-51.
- Fung, J., Abu-Elmagd, K., Jain, A., Gordon, R., Tzakis, A., Todo, S., Takaya, S., Alessiani, M., Demetris, A., Bronster, O., Martin, M., Mieles, L., Selby, R., Reyes, J., Doyle, H., Stieber, A., Casavilla, A. and Starzl, T. (1991). Transplantation proceedings 23 (6): 2977-83.
- Gaida K., Salimi-Moosavi H., Subramanian R., Almon V., Knize A., Zhang M., Lin F.F., Nguyen H.Q., Zhou L., Sullivan J.K., Wong M., McBride H.J. (2015). J Immunotoxicol 12:164-173.
- Glynne, R., Akkaraju, S., Healy, J.I., Rayner, J., Goodnow, C.C. and Mack, D.H. (2000). Nature 403 (6770): 672-6.
- Goldfeld, A.E., Flemington, E.K., Boussiotis, V.A., Theodos, C.M., Titus, R.G., Strominger, J.L. and Speck, S.H. (1992). Proceedings of the National Academy of Sciences of the United States of America 89 (24): 12198-201.
- Goldfeld, A. E., Tsai, E., Kincaid, R., Belshaw, P. J., Schrieber, S. L., Strominger, J. L. and Rao, A. (1994). Journal of experimental medicine. 180(2): 763-768.
- Heidt, S., Roelen, D. L., Eijsink, C., Eikmans, M., van Kooten, C., Claas, F. H. and Mulder, A. (2010). Clinical and experimental immunology. 159(2): 199-207.
- Hiroi, J., Sengoku, T., Morita, K., Kishi, S., Sato, S., Ogawa, T., Tsudzuki, M., Matsuda, H., Wada, A. and Esaki, K. (1998). Japanese journal of pharmacology. 76(2): 175-183.
- Hultsch, T., Albers, M. W., Schreiber, S.L. and Hohman, R. J. (1991). Proceedings of the national academic science of the United States of America. 14: 6229-6233.
- Imai, A., Sahara, H., Tamura, Y., Jimbow, K., Saito, T., Ezoe, K., Yotsuyanagi, T. and Sato, N. (2007). European journal of immunology. 37(7): 1730-1738.
- Ito N., Shibuguchi N., Ishikawa R., Tanaka S., Tokita Y., Nakajima-Shimada J., Hosaka K. (2013). Biosci Biotechnol Biochem. 77(5): 954-960
- Jain, J., McCaffrey, P. G., Valge-Archer, V. E. and Rao, A. (1992). Nature. 356(6372): 801-804.
- Jain, J., Miner, Z. and Rao, A. (1993). Journal of immunology. 151(2): 837-848.
- Jennings, C., Kusler, B. and Jones, P. P. (2009). Innate immunity. 15(2): 109-120.
- Kang, Y. J., Kusler, B., Otsuka, M., Hughes, M., Suzuki, N., Suzuki, S., Yeh, W. C., Akira, S., Han, J. and Jones, P. P. (2007). Journal of immunology. 179(7): 4598-4607.
- Kang, C. B., Hong, Y., Dhe-Paganon, S. and Yoon, H. S. (2008). Neurosignals. 16: 318-325.
- Kevin G, Hossein S, Raju S, Valerie A, Anna K, Ming Z, Fen-Fen L, Hung Q. N, Lei Z, John K. S, Min W and Helen J. M(2015). Journal of Immunotoxicology, 12:2, 164-173,
- Kim, T., Kim, N. and Kang, H. J. (2010). Journal of leukocyte biology. 88:1089-1097.
- Kino, T., Hatanaka, H., Miyata, S., Inamura, N., Nishiyama, M., Yajima, T., Goto, T., Okuhara, M., Kohsaka, M. and Aoki, H. (1987a). Journal of antibiotics. 40(9): 1256-1265.
- Kino, T., Hatanaka, H., Hashimoto, M., Nishiyama, M., Goto, T., Okuhara, M., Kohsaka, M., Aoki, H. and Imanaka, H. (1987b). Journal of antibiotics. 40(9): 1249-1255.
- Klee, C. B., Draetta, G. F. and Hubbard, M. J. (1988). Advances in enzymology and related areas of molecular biology. 61:149-200.
- Lee, Y. R., Yang, I. H., Lee, Y. H., Im, S. A., Song, S., Li, H., Han, K., Kim, K., Eo, S. K. and Lee, C. K. (2005). Blood. 105(10): 3951-3955.
- Lehmann, D.M., Williams, W.C. (2018). Toxicol In Vitro.53: 20–28.
- Liu, J., Farmer, J. D. Jr., Lane, W. S., Friedman, J., Weissman, I., and Schreiber, S. L. (1991). Cell. 66(4): 807-815.
- Liu, J., Albers, M. W., Wandless, T. J., Luan, S., Alberg, D. G., Belshaw, P. J., Cohen, P., MacKintosh, C., Klee, C. B. and Schreiber, S.L.. (1992). Biochemistry. 31(16):3896-901.
- Liu, J. (1993). Immunology today. 14(6): 290-305.
- Luster, M.I., and Rosenthal, G.J. (1993). Environmental Health Perspectives. 100: 219-36.
- Macian, F. (2005). Nature reviews. Immunology. 5(6): 472-84.
- Magari, K., Miyata, S., Ohkubo, Y., Mutoh, S. and Goto, T. (2003). British journal of pharmacology. 139: 927-934.
- Maguire O, Tornatore KM, O'Loughlin KL, Venuto RC and Minderman H. (2013). Cytometry A. 83(12):1096-104.
- Matsuda, S., Koyasu, S. (2000). Tanpakushitsu kakusan koso. 45(11): 1823-1831.
- Meingassner, J.G. and Stütz, A. (1992). Journal of investigative dermatology 98(6): 851-5
- Mittelstadt, PR. and Ashwell, JD. (2001). J Biol Chem. 276(31):29603-10.
- Nalesnik, MA., Todo, S., Murase, N., Gryzan, S., Lee, PH., Makowka, L., and Starzl, TE. (1987). Transplantation Proceedings 19(5 Suppl 6): 89-92.
- Panhans-Gross, A., Novak, N., Kraft, S., and Bieber, T. (2001). Journal of Allergy and Clinical Immunology 107(2): 345-52.
- Pirsch, JD., Miller, J., Deierhoi, MH., Vincenti, F., and Filo, RS. (1997). Transplantation 63(7): 977-83.
- Ranger, AM., Oukka, M., Rengarajan, J. and Glimcher, LH. (1998). Immunity. 9(5):627-35.
- Rao, A., Luo, C., and Hogan, PG. (1997). Annual Review of Immunology 15: 707-47.
- Roman D., Ulrich P., Paul G., Court P., Vit P., Kehren., Mahl A., (2004). Toxicology Letters 149: 133-140.
- Sakuma, S., Kato, Y., Nishigaki, F., Sasakawa, T., Magari, K., Miyata, S., Ohkubo, Y., and Goto, T. (2000). British Journal of Pharmacology 130(7): 1655-63.
- Sakuma, S., Higashi, Y., Sato, N., Sasakawa, T., Sengoku, T., Ohkubo, Y., Amaya, T., and Goto, T. (2001a). International Immunopharmacology 1(6): 1219-26.
- Sakuma, S., Kato, Y., Nishigaki, F., Magari, K., Miyata, S., Ohkubo, Y., and Goto, T. (2001b). International Immunopharmacology 1(4): 749-57.
- Sasakawa, Y., Sakuma, S., Higashi, Y., Sasakawa, T., Amaya, T., and Goto, T. (2000). European Journal of Pharmacology 403(3): 281-8.
- Sasaki, T., Nakamura, W., Inokuma, S., and Matsubara, E. (2015). Journal of Clinical Rheumatology Feb 3.
- Schreiber, SL., and Crabtree, GR. (1992). Immunology Today 13(4): 136-42.
- Serfling, E., Berberich-Siebelt, F., Chuvpilo, S., Jankevics, E., Klein-Hessling, S., Twardzik, T., and Avots, A., (2000). Biochimica et Biophysica Act 1498 (1): 1-18.
- Sieber M., Baumgrass R., (2009). Cell Commun Signal Oct 27;7:25
- Siekierka, JJ., Hung, SH., Poe, M., Lin, CS., and Sigal, NH. (1989a). Nature 341(6244): 755-57.
- Siekierka, JJ., Staruch, MJ., Hung, SH., and Sigal, NH. (1989b). Journal of immunology 143(5): 1580-3.
- Siekierka, JJ., Wiederrecht, G., Greulich, H., Boulton, D., Hung, SH., Cryan, J., Hodges, PJ., and Sigal, NH. (1990). Journal of Biological Chemistry 265(34): 21011-5.
- Sonoda, T., Takahara, S., Takahashi, K., Uchida, K., Ohshima, S., Toma, H., Tanabe, K., Yoshimura, N.; Japanese Tacrolimus Study Group. (2003). Transplantation 75(2): 199-204.
- Standaert, RF., Galat, A., Verdine, GL., and Schreiber, SL. (1990). Nature 346(6285): 671-4.
- Tamura, F., Masuhara, A., Sakaida, I., Fukumoto, E., Nakamura, T., and Okita, K. (1998). Journal of Gastroenterology and Hepatology 13(7): 703-8.
- Ulrich, P., Paul, G., Perentes, E., Mahl, A., and Roman D. (2004). Toxicology Letters 149(1-3): 123-31.
- Vacher-Coponat, H., Brunet, C., Moal, V., Loundou, A., Bonnet, E., Lyonnet, L., Ravet, S., Sampol-Manos, E., Sampol, J., Berland, Y., George, FD., and Paul, P. (2006). Transplantation 82(4): 558-66.
- Vandewalle, A., Tourneur, E., Bens, M., Chassin, C., and Werts, C. (2014). Cell Communication and Signaling 12: 8
- Weiwad, M., Edlich, F., Kilka ,S., Erdmann, F., Jarczowski, F., Dorn, M., Moutty, M.C. and Fischer, G. (2006). Biochemistry 45(51): 15776-84.
- Wicker, L.S., Boltz, R.C. Jr., Matt, V., Nichols. E.A., Peterson, L.B. and Sigal, N.H. (1990). European journal of immunology 20(10): 2277-83.
- Yoshida, T., Yamashita, K., Watanabe, M., Koshizuka, Y., Kuraya, D., Ogura, M., Asahi, Y., Ono, H., Emoto, S., Mizukami, T., Kobayashi, N., Shibasaki, S., Tomaru, U., Kamachi, H., Matsushita, M., Shiozawa, S., Hirono, S. and Todo, S. (2015). Am J Transplant. 15(10): 2565-75.
- Yoshimura, N., Matsui, S., Hamashima, T. and Oka, T. (1989). Transplantation 47(2): 356-9.
- Yoshino, T., Nakase, H., Honzawa, Y., Matsumura, K., Yamamoto, S., Takeda, Y., Ueno, S., Uza, N., Masuda, S., Inui, K. and Chiba, T. (2010). Inflammatory bowel disease. 16(12): 2022-33
- Zhang, B.W., Zimmer, G., Chen, J., Ladd, D., Li, E., Alt, F.W., Wiederrecht, G., Cryan, J., O'Neill, E.A., Seidman, C.E., Abbas, A.K. and Seidman, J.G.. (1996). Journal of experimental medicine 183(2): 413-20.
- Zhu, J. and McKeon, F. (1999). Nature. 398(6724): 256-60.
- de Paulis, A., Cirillo, R., Ciccarelli, A., de Crescenzo, G., Oriente, A. and Marone, G. (1991). Journal of immunology 147(12): 4278-85.
- de Paulis, A., Stellato, C., Cirillo, R., Ciccarelli, A., Oriente, A. and Marone, G. (1992). Journal of investigative dermatology 99(6): 723-8.
- van Dieren, J.M., Lambers, M.E.H., Kuipers, E.J., Samsom, J.N., van der Woude, C.J. and Nieuwenhuis, E.E.S. (2010). Digestive diseases and sciences 55(9): 2514-19.
- van Lierop, P.P., de Haar, C., Lindenbergh-Kortleve, D.J., Simons-Oosterhuis, Y., van Rijt, L.S., Lambrecht, B.N., Escher, J.C., Samsom, J.N. and Nieuwenhuis, E.E. (2010). Inflammatory bowel disease 16(3): 442-51.
- Maruho Co.,Ltd. (2014) Drug interview form Protopic ointment 0.1% Revised 16th edition.
- Astellas Pharma Inc. (2014) Drug interview form Prograf capsules 0.5mg, 1mg, 5mg, granules 0.2mg, 1mg. Revised 34th edition
- Astellas Pharma Inc. (2014) Drug interview form Prograf capsules 0.5 mg, 1 mg, 5 mg, granules 0.2 mg, 1 mg. Revised 34th edition
- Novartis Pharma K.K. (2016). Drug interview form Simulect i.v. injection 20 mg. 10th edition.
- Sanofi K.K. (2018) Drug interview form Dupixent subcutaneous injection 300 mg syringe. 2nd edition.
- Taiho Pharmaceutical Co.,Ltd. (2013) Drug interview form IPD capsule 50 and 100. Revised 5th edition.
- Fyjii Y., Gogi H., Takamura K., Sakuma A. and Goto T. Kisotorinsyo 31(8): 2693-2700 (in Japanese)
- Sengoku T., Morita K., Sato A., Sakuma S., Ogawa T., Hiroi J., Fujii T and Goto T. (1998) Folia Pharmacol. Jpn. (Nippon Yakurigaku Zasshi) 112, 221-232