
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 219
Title
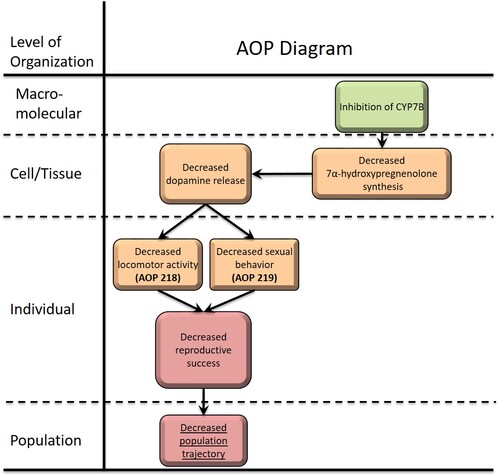
Inhibition of CYP7B activity leads to decreased reproductive success via decreased sexual behavior
Short name
Graphical Representation
Point of Contact
Contributors
- Florence Pagé-Larivière
- Arthur Author
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| CYP7B activity, inhibition | May 17, 2017 12:21 |
| 7α-hydroxypregnenolone synthesis in the brain, decreased | May 17, 2017 13:08 |
| Dopamine release in the brain, decreased | May 17, 2017 13:05 |
| Sexual behavior, decreased | May 17, 2017 21:17 |
| Decreased, Reproductive Success | December 03, 2016 16:37 |
| Decrease, Population growth rate | January 03, 2023 09:09 |
| CYP7B activity, inhibition leads to 7α-hydroxypregnenolone synthesis in the brain, decreased | May 17, 2017 21:09 |
| 7α-hydroxypregnenolone synthesis in the brain, decreased leads to Sexual behavior, decreased | May 17, 2017 22:00 |
| 7α-hydroxypregnenolone synthesis in the brain, decreased leads to Dopamine release in the brain, decreased | May 25, 2017 14:22 |
| Sexual behavior, decreased leads to Decreased, Reproductive Success | May 08, 2017 11:20 |
| Dopamine release in the brain, decreased leads to Sexual behavior, decreased | May 09, 2017 10:06 |
| Decreased, Reproductive Success leads to Decrease, Population growth rate | April 06, 2022 13:50 |
| Ketoconazole | May 02, 2017 11:08 |
Abstract
This AOP details the linkage between CYP7B inhibition and decreased sexual behavior that adversely impacts reproductive success. CYP7B is expressed in the brain and catalyzes the conversion of pregnenolone to 7α-hydroxypregnenolone, a neurosteroid that stimulates the release of dopamine in the telencephalon. When released through this pathway, dopamine binds D2 receptor which is involved in induction of sexual behaviors, among other effects. Ketoconazole and other azole fungicides are potent inhibitor of cytochrome P450s, including CYP7B. They bind to the heme site of the enzyme preventing its catalytic activity. When exposed to one of these molecules, 7α-hydroxypregnenolone synthesis decreases which, in turn, reduces dopamine release in the telencephalon and limits sexual behavior. Since sexual behaviors are closely associated to reproductive success, its inhibition negatively affects the fitness of animals.
7α-hydroxypregnenolone was recently discovered and its function and regulation remain unclear. The few studies that focused on this neurosteroid and that were used for this AOP are based on in vitro and in vivo experiments quail and newt. Since the function of this neurosteroid differs in mammals, this AOP is only applicable to non-mammalian vertebrates. It is also limited to male.
AOP Development Strategy
Context
This AOP shares most of its key events with AOP 218, with the exception of Locomotor activity, decreased (Event 1389). Due to this difference, the domain of applicability of the two AOPs differs and limits their compatibility. For that reason, two similar AOPs with different domain of applicability were created.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1386 | CYP7B activity, inhibition | CYP7B activity, inhibition |
| KE | 1387 | 7α-hydroxypregnenolone synthesis in the brain, decreased | 7α-hydroxypregnenolone synthesis in the brain, decreased |
| KE | 1388 | Dopamine release in the brain, decreased | Dopamine release in the brain, decreased |
| KE | 1390 | Sexual behavior, decreased | Sexual behavior, decreased |
| KE | 1141 | Decreased, Reproductive Success | Decreased, Reproductive Success |
| AO | 360 | Decrease, Population growth rate | Decrease, Population growth rate |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
| Name |
|---|
| Ketoconazole |
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult, reproductively mature |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male |
Overall Assessment of the AOP
Domain of Applicability
Taxons: This AOP is supported with evidence from studies conducted with newt and quail. Based on anticipated conservation of the biology associated with the KEs and KERs described, it is presumed to be applicable to all amphibian and bird.
Previous evidence suggest that this AOP is not applicable to mammal. All the key events of this AOP are described or are biologically plausible in mammal, but the relationship between them might differ, as suggested by Yau et al. (2006).
Sex: This AOP is applicable to male only.
Life Stage: This AOP applies to sexually mature animals since the endpoint is related to reproduction.
Essentiality of the Key Events
Few studies measured multiple key events of this AOP. For this reason, the evidence for essentiality of the key events is mainly indirect and provided by a series of antagonist/exogenous supplementation experiments. The animal models used for these investigations were newt and quail.
|
Key event |
Essentiality |
Rational |
|
MIE Inhibition of CYP7B |
Moderate |
At present, no CYP7B knock-out experiments were conducted in species of interest. However, several indirect evidences linking CYP7B inhibition to a decreased locomotor activity suggest an important correlation between the two events.
|
|
KE1 7α-hydroxypregnenolone, decreased |
Strong |
Direct evidences connecting this neurosteroid to sexual behavior were described.
|
|
KE2 Dopamine release, decreased |
Moderate |
There is strong evidence demonstrating the involvement of dopamine in sexual behavior among all vertebrates. However, only indirect evidence relates CYP7B inhibition to a decreased dopamine release. The rational is stronger for 7α-hydroxypregnenolone in relation to dopamine release, although this neurosteroid receptor remains to be identified.
|
|
KE3 Locomotor activity, decreased |
Strong |
All the previous key events can decrease sexual behavior in male quail and newt. |
Evidence Assessment
Biological plausibility
This AOP connects the CYP7B catalyzed synthesis on an important neurosteroid to a well characterized sequence of events. For instance, the involvement of dopamine in sexual behavior that in turn impacts on reproductive success is well described and undisputed (Melis et al., 1995; Hull et al., 2004). What is less characterized is the relation between 7α-hydroxypregnenolone and dopamine release. Since the neurosteroid receptor has yet to be identified, no direct interaction between 7α-hydroxypregnenolone and dopaminergic neuron has been demonstrated. It is thus possible that an intermediate event takes place in between to indirectly connect the neurosteroid to dopamine release.
In terms of structural plausibility, the brain expresses the steroidogenic enzymes required for pregnenolone synthesis, the main substrate of CYP7B. It also expresses CYP7B which synthesizes high concentration of 7α-hydroxypregnenolone in the diencephalon. This region of the brain is populated by neurons projecting into the striatum which is known to express a high quantity of D1- and D2-like dopamine receptor and control motor activity (Orgen S. et al., 1986; Mezey S. et al., 2002; Callier S. et al., 2003).
Uncertainties or inconsistencie
At present, there are no inconsistencies reported in the literature, but some gaps remain to be filled.
The most important ones are 7α-hydroxypregnenolone receptor localization and the connection between 7α-hydroxypregnenolone and dopamine release discussed in the previous section.
In addition, mammalian CYP7B not only catalyzes the 7α-hydroxylation of pregnenolone but also that of dehydroepiandrosterone (DHEA). Although no clear information reported this enzymatic reaction in the bird, it is plausible that CYP7B catalyzes the hydroxylation of DHEA. Thus, the phenotypic effect of CYP7B inhibition in the brain cannot be uniquely attributed to a depletion in 7α-hydroxypregnenolone. Additionally, ketoconazole is known to inhibit a variety of CYPs, which suggest that animal exposed to it are likely to have several other enzymes inhibited. It is plausible that the impacts of ketoconazole are the result of multiple CYPs inhibition that all converge towards the same phenotype. These off target effects greatly limit the investigations on 7α-hydroxypregnenolone since its concentration cannot be specifically decreased.
If a CYP7B knock-out in the brain was to be performed in an animal species, 7α-hydroxyDHEA supplementation would be required to properly study 7α-hydroxypregnenolone function.
Known Modulating Factors
Quantitative Understanding
This information is not available for the moment.