
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 27
Title
Cholestatic Liver Injury induced by Inhibition of the Bile Salt Export Pump (ABCB11)
Short name
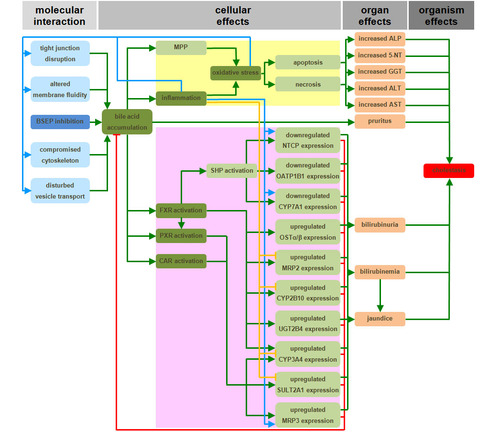
Graphical Representation
Point of Contact
Contributors
- Mathieu Vinken
- Arthur Author
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.19 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition, Bile Salt Export Pump (ABCB11) | September 16, 2017 10:14 |
| Activation of specific nuclear receptors, Transcriptional change | September 16, 2017 10:14 |
| Bile accumulation, Pathological condition | September 16, 2017 10:14 |
| Release, Cytokine | September 16, 2017 10:14 |
| Increase, Inflammation | February 28, 2024 06:33 |
| Production, Reactive oxygen species | December 03, 2016 16:37 |
| Peptide Oxidation | November 13, 2017 10:22 |
| Cholestasis, Pathology | December 03, 2016 16:33 |
| Bile accumulation, Pathological condition leads to Activation of specific nuclear receptors, Transcriptional change | December 03, 2016 16:37 |
| Activation of specific nuclear receptors, Transcriptional change leads to Cholestasis, Pathology | December 03, 2016 16:37 |
| Release, Cytokine leads to Increase, Inflammation | December 03, 2016 16:37 |
| Production, Reactive oxygen species leads to Peptide Oxidation | December 03, 2016 16:37 |
| Inhibition, Bile Salt Export Pump (ABCB11) leads to Bile accumulation, Pathological condition | December 03, 2016 16:38 |
| Bile accumulation, Pathological condition leads to Release, Cytokine | December 03, 2016 16:38 |
| Increase, Inflammation leads to Cholestasis, Pathology | December 03, 2016 16:38 |
| Bile accumulation, Pathological condition leads to Production, Reactive oxygen species | December 03, 2016 16:38 |
| Peptide Oxidation leads to Cholestasis, Pathology | December 03, 2016 16:38 |
Abstract
Adverse outcome pathways (AOPs) have been recently introduced in human risk assessment as pragmatic tools with multiple applications. As such, AOPs intend to provide a clear-cut mechanistic representation of pertinent toxicological effects. AOPs are typically composed of a molecular initiating event, a series of intermediate steps and key events, and an adverse outcome. In the current study, an AOP framework is proposed for cholestasis triggered by drug-mediated inhibition of the bile salt export pump transporter protein. For this purpose, an in-depth survey of relevant scientific literature was carried out in order to identify intermediate steps and key events. The latter include bile accumulation, the induction of oxidative stress and inflammation, and the activation of specific nuclear receptors. Collectively, these mechanisms drive both a deteriorative cellular response, which underlies directly caused cholestatic injury, and an adaptive cellular response, which is aimed at counteracting cholestatic insults. AOP development was performed according to OECD guidance, including critical consideration of the Bradford Hill criteria for weight of evidence assessment and the OECD key questions for evaluating AOP confidence. The postulated AOP is expected to serve as the basis for the development of new in vitro tests and the characterization of novel biomarkers of drug-induced cholestasis.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 41 | Inhibition, Bile Salt Export Pump (ABCB11) | Inhibition, Bile Salt Export Pump (ABCB11) |
| KE | 288 | Activation of specific nuclear receptors, Transcriptional change | Activation of specific nuclear receptors, Transcriptional change |
| KE | 214 | Bile accumulation, Pathological condition | Bile accumulation, Pathological condition |
| KE | 87 | Release, Cytokine | Release, Cytokine |
| KE | 149 | Increase, Inflammation | Increase, Inflammation |
| KE | 249 | Production, Reactive oxygen species | Production, Reactive oxygen species |
| KE | 209 | Peptide Oxidation | Peptide Oxidation |
| AO | 357 | Cholestasis, Pathology | Cholestasis, Pathology |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult, reproductively mature | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| humans | Homo sapiens | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Mixed | High |
Overall Assessment of the AOP
1. Concordance of dose-response relationships: Morgan and colleagues investigated the potential of more than 200 benchmark drugs to inhibit BSEP. As much as 16% of the tested drugs displayed high potency of BSEP inhibition (IC50 ≤ 25 µM), the majority of which are associated with liver liabilities in humans (Morgan et al., 2010). Likewise, 17 of 85 pharmaceuticals tested by Dawson and coworkers inhibited BSEP (IC50 ≤ 100 µM), all which are known to cause DILI (Dawson et al., 2011). Furthermore, several of the BSEP-inhibiting drugs cause cholestastic liver injury in a dose-dependent way, such as is the case for troglitazone and bosentan in rats (Funk et al., 2001) and humans (Fattinger et al., 2001), respectively. Thus, there is a clear relationship between the IC50 of BSEP inhibition and the occurrence of (cholestatic) DILI.
2. Temporal concordance among the key events and adverse effect: Inhibition of BSEP activity and the resulting accumulation of bile acids primarily triggers a direct cellular response, which is associated with deteriorative processes, such as inflammation, oxidative stress and cell death. It also causes a secondary and rather indirect cellular response, which is adaptive in nature. Indeed, a well-orchestrated network of mechanisms is activated, all of which are targeted towards the elimination of bile from the liver (Boyer, 2009; Zollner and Trauner, 2006 and 2008; Wagner et al., 2009). The temporal concordance between these cellular responses is not clear. It is, however, conceivable to assume that the adaptive response becomes manifested at a somewhat later stage when compared to the primary events, especially since this secondary response highly depends on transcriptional regulation. Nonetheless, it is clear that the graphical linear representation of cholestatic DILI resulting from BSEP inhibition as a sequence of events is an oversimplification of a probably very complex network of entangled consecutive and parallel reactions.
3. Strength, consistency, and specificity of association of adverse effect and initiating event: BSEP is considered as the major apical transporter protein that pumps bile salts from hepatocytes into bile canaliculi. As a part of this pivotal task, BSEP has a very narrow substrate specificity with only a few known non-bile substrates (Dawson et al., 2011; Kis et al., 2012; Morgan et al., 2010). Defects in BSEP expression or function therefore can be anticipated to have drastic consequences with respect to bile homeostasis. Indeed, a plethora of studies has demonstrated that BSEP inhibition or impairment is causally linked to the induction of cholestasis in a dose-dependent way, both in experimental animals and in humans (Zollner and Trauner, 2006 and 2008; Wagner et al., 2009). Thus, there is a well-established, direct, specific and quantitative association between BSEP inhibition and the onset of cholestatic DILI.
4. Biological plausibility, coherence, and consistency of the experimental evidence: In essence, BSEP inhibition (i.e. the MIE) activates a number of mechanisms that drive a deteriorative cellular response, which underlies directly caused cholestatic injury, as well as an adaptive cellular response, which is aimed at counteracting cholestatic insults. Both these responses contribute to the clinical manifestation of cholestasis (i.e. the AO). Serum concentrations of ALT, AST, ALP, GGT and 5’-NT indeed increase because of bile acid-induced membrane damage of hepatocytes and cholangiocytes (Hofmann, 2009; Kuntz and Kuntz, 2008; Padda et al., 2011). At the same time, elevated concentrations of bilirubin in serum and urine are observed, reflecting the compensatory response of the organism to counteract bile acid accumulation. Hyperbilirubinemia causes jaundice, while the increased presence of bile acids in serum is thought to induce pruritus (Hofmann, 2009; Kuntz and Kuntz, 2008; Padda et al., 2011; Zollner and Trauner, 2008).
5. Alternative mechanisms that logically present themselves and the extent to which they may distract from the postulated AOP. It should be noted that alternative mechanisms of action, if supported, require a separate AOP: Although predominant, BSEP inhibition is not the sole MIE in cholestatic DILI, as depicted in the established AOP. In this regard, cyclosporine A not only inhibits BSEP (Dawnson et al., 2011; Kis et al., 2012; Morgan et al., 2010), but also induces cholestasis by inhibition of intrahepatic vesicle transport (Roman et al., 1990) and by affecting canalicular membrane fluidity (Yasumiba et al., 2001). Several of these events, in particular the cytoskeletal changes, might be considered as secondary and non-specific phenomena (Trauner et al., 1998b). Nevertheless, separate AOPs could be drafted for each of these alternative mechanisms in cholestatic DILI.
6. Uncertainties, inconsistencies and data gaps: Although a clear causal and dose-dependent relationship has been established between BSEP inhibition and the clinical onset of cholestasis (Zollner and Trauner, 2006 and 2008; Wagner et al., 2009), several mechanisms of the intermediate steps and key events as well as their linkage are not fully understood. A prominent discussion in this respect relates to the nature of the cell death mode, namely apoptosis or necrosis, associated with cholestasis (Woolbright and Jaeschke, 2012). High concentrations of hydrophobic bile acids induce apoptotic cell death in cultures of primary hepatocytes (Gores et al., 1998; Botla et al., 1995; Schoemaker et al., 2004), yet such concentrations are not achieved in vivo (Zhang et al., 2012). It has therefore been suggested that the main mechanism of cell death in cholestasis in vivo is necrosis (Woolbright and Jaeschke, 2012). In fact, this seems to be a general consideration of the AOP, as several other constituting data also have been derived from in vitro experimentation and need to be substantiated in vivo. On the other hand, a number of data are still lacking, including the full identification of FXR, PXR and CAR target genes, which may additionally contribute to the adaptive response to BSEP inhibition. Furthermore, ongoing research regarding the regulation of these nuclear receptors in cholestatic DILI might add complexity to the AOP. It is known that they act, at least in part, by recruiting co-activators and co-repressors (Gollamudi et al., 2008; Wagner et al., 2009). Moreover, compelling evidence suggests that nuclear receptors are regulated epigenetically, which might necessitate inclusion in the AOP (Elloranta and Kullak-Ublick, 2005; Wagner et al., 2009). Additional uncertainties, inconsistencies and data gaps associated with the established AOP relate to the temporal concordance of the intermediate steps and key events, the consistency of the available experimental data, alternative mechanisms involved, interspecies and intraspecies differences.
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Allen, K., Jaeschke, H., Copple, B.L. (2011). Bile acids induce inflammatory genes in hepatocytes: a novel mechanism of inflammation during obstructive cholestasis. Am. J. Pathol. 178, 175-186.
Andersen, M.E., Clewell, R., Bhattacharya, S. (2012). Developing in vitro tools sufficient by themselves for 21st century risk assessment. In Towards the Replacement of In Vivo Repeated Dose Systemic Toxicity Testing Volume 2 (T. Gocht, M. Schwarz, Eds.), pp. 347-360. Imprimerie Mouzet, France.
Andersen, M.E., McMullen, P., Bhattacharya, S. (2013). Toxicogenomics for transcription factor-governed molecular pathways: moving on to roles beyond classification and prediction. Arch. Toxicol. 87, 7-11.
Ankley, G.T., Bennett, R.S., Erickson, R.J., Hoff, D.J., Hornung, M.W., Johnson, R.D., Mount, D.R., Nichols, J.W., Russom, C.L., Schmieder, P.K., Serrrano, J.A., Tietge, J.E., Villeneuve, D.L. (2010). Adverse outcome pathways: a conceptual framework to support ecotoxicology research and risk assessment. Environ. Toxicol. Chem. 29, 730-741.
Barbier, O., Torra, I.P., Sirvent, A., Claudel, T., Blanquart, C., Duran-Sandoval, D., Kuipers, F., Kosykh, V., Fruchart, J.C., Staels, B. (2003). FXR induces the UGT2B4 enzyme in hepatocytes: a potential mechanism of negative feedback control of FXR activity. Gastroenterology 124, 1926-1940.
Bernstein, H., Bernstein, C., Payne, C.M., Dvorakova, K., Garewal, H. (2005). Bile acids as carcinogens in human gastrointestinal cancers. Mutat. Res. 589, 47-65.
Bogdanffy, M.S., Daston, G., Faustman, E.M., Kimmel, C.A., Kimmel, G.L., Seed, J., Vu, V. (2001). Harmonization of cancer and noncancer risk assessment: proceedings of a consensus-building workshop. Toxicol. Sci. 61, 18-31.
Botla, R., Spivey, J.R., Aguilar, H., Bronk, S.F., Gores, G.J. (1995). Ursodeoxycholate (UDCA) inhibits the mitochondrial membrane permeability transition induced by glycochenodeoxycholate: a mechanism of UDCA cytoprotection. J. Pharmacol. Exp. Ther. 272, 930-938.
Boyer, J.L., Trauner, M., Mennone, A., Soroka, C.J., Cai, S.Y., Moustafa, T., Zollner, G., Lee, J.Y., Ballatori, N. (2006). Upregulation of a basolateral FXR-dependent bile acid efflux transporter OSTalpha-OSTbeta in cholestasis in humans and rodents. Am. J. Physiol. Gastrointest. Liver Physiol. 290, G1124-G1130.
Boyer, L.B. (2009). Adaptive regulation of hepatocyte transporters in cholestasis. In The Liver: Biology and Pathobiology 5th edition (I.M. Arias, H.J. Alter, J.L. Boyer, D.E. Cohen, N. Fausto, D.A. Shafritz, A.W. Wolkoff, Eds.), pp. 681-702. Wiley-Blackwell, Oxford.
Björnsson, E., Olsson, R. (2005). Outcome and prognostic markers in severe drug-induced liver disease. Hepatology 42, 481-489.
Blomme, E.A., Yang, Y., Waring, J.F. (2009). Use of toxicogenomics to understand mechanisms of drug-induced hepatotoxicity during drug discovery and development. Toxicol. Lett. 186, 22-31.
Chen, H.L., Chen, H.L., Liu, Y.J., Feng, C.H., Wu, C.Y., Shyu, M.K., Yuan, R.H., Chang, M.H. (2005). Developmental expression of canalicular transporter genes in human liver. J. Hepatol. 43, 472-477.
Cheng, X., Buckley, D., Klaassen, C.D. (2007). Regulation of hepatic bile acid transporters Ntcp and Bsep expression. Biochem. Pharmacol. 74, 1665-1676.
Cherrington, N.J., Slitt, A.L., Li, N., Klaassen, C.D. (2004). Lipopolysaccharide-mediated regulation of hepatic transporter mRNA levels in rats. Drug Metab. Dispos. 32, 734-741.
Dawson, S., Stahl, S., Paul, N., Barber, J., Kenna, J.G. (2011). In vitro inhibition of the bile salt export pump correlates with risk of cholestatic drug-induced liver injury in humans. Drug Metab. Dispos. 40, 130-138.
Denson, L.A., Sturm, E., Echevarria, W., Zimmerman, T.L., Makishima, M., Mangelsdorf, D.J., Karpen, S.J. (2001). The orphan nuclear receptor, shp, mediates bile acid-induced inhibition of the rat bile acid transporter, ntcp. Gastroenterology 121, 140-147.
Echchgadda, I., Song, C.S., Oh, T., Ahmed, M., De La Cruz, I.J., Chatterjee, B. (2007). The xenobiotic-sensing nuclear receptors pregnane X receptor, constitutive androstane receptor, and orphan nuclear receptor hepatocyte nuclear factor 4alpha in the regulation of human steroid-/bile acid-sulfotransferase. Mol. Endocrinol. 21, 2099-2111.
Eloranta, J.J., Kullak-Ublick, G.A. (2005). Coordinate transcriptional regulation of bile acid homeostasis and drug metabolism. Arch. Biochem. Biophys. 433, 397-412.
EU. (2003). Directive 2003/15/EC of the European parliament, of the council of 27 February amending council directive 76/768/EEC on the approximation of the laws of the member states relating to cosmetic products. OV. J. L. 066, 26-35.
Fattinger, K., Funk, C., Pantze, M., Weber, C., Reichen, J., Stieger, B., Meier, P.J. (2001). The endothelin antagonist bosentan inhibits the canalicular bile salt export pump: a potential mechanism for hepatic adverse reactions. Clin. Pharmacol. Ther. 69, 223-231.
Faucette, S.R., Sueyoshi, T., Smith, C.M., Negishi, M., Lecluyse, E.L., Wang, H. (2006). Differential regulation of hepatic CYP2B6 and CYP3A4 genes by constitutive androstane receptor but not pregnane X receptor. J. Pharmacol. Exp. Ther. 317, 1200-1209.
Feingold, K.R., Spady, D.K., Pollock, A.S., Moser, A.H., Grunfeld, C. (1996). Endotoxin, TNF, and IL-1 decrease cholesterol 7 alpha-hydroxylase mRNA levels and activity. J. Lipid Res. 37, 223-228.
Funk, C., Pantze, M., Jehle, L., Ponelle, C., Scheuermann, G., Lazendic, M., Gasser, R. (2001). Troglitazone-induced intrahepatic cholestasis by an interference with the hepatobiliary export of bile acids in male and female rats: correlation with the gender difference in troglitazone sulfate formation and the inhibition of the canalicular bile salt export pump (Bsep) by troglitazone and troglitazone sulfate. Toxicology 167, 83-98.
Gnerre, C., Blättler, S., Kaufmann, M.R., Looser, R., Meyer, U.A. (2004). Regulation of CYP3A4 by the bile acid receptor FXR: evidence for functional binding sites in the CYP3A4 gene. Pharmacogenetics 14, 635-645.
Gollamudi, R., Gupta, D., Goel, S., Mani, S. (2008). Novel orphan nuclear receptors-coregulator interactions controlling anti-cancer drug metabolism. Curr. Drug Metab. 9, 611-613.
Goodwin, B., Jones, S.A., Price, R.R., Watson, M.A., McKee, D.D., Moore, L.B., Galardi, C., Wilson, J.G., Lewis, M.C., Roth, M.E., Maloney, P.R., Willson, T.M., Kliewer, S.A. (2000). A regulatory cascade of the nuclear receptors FXR, SHP-1, and LRH-1 represses bile acid biosynthesis. Mol. Cell 6, 517-526.
Gores, G.J., Miyoshi, H., Botla, R., Aguilar, H.I., Bronk, S.F. (1998). Induction of the mitochondrial permeability transition as a mechanism of liver injury during cholestasis: a potential role for mitochondrial proteases. Biochim. Biophys. Acta 1366, 167-175.
Green, R.M., Hoda, F., Ward, K.L. (2000). Molecular cloning and characterization of the murine bile salt export pump. Gene 241, 117-123.
Gu, X., Ke S., Liu, D., Sheng, T., Thomas, P.E., Rabson, A.B., Gallo, M.A., Xie ,W., Tian, Y. (2006). Role of NF-kappaB in regulation of PXR-mediated gene expression: a mechanism for the suppression of cytochrome P-450 3A4 by proinflammatory agents. J. Biol. Chem. 281, 17882-17889.
Gujral, J.S., Farhood, A., Bajt, M.L., Jaeschke, H. (2003). Neutrophils aggravate acute liver injury during obstructive cholestasis in bile duct-ligated mice. Hepatology 38, 355-363.
Gujral, J.S., Liu, J., Farhood, A., Hinson, J.A., Jaeschke, H. (2004). Functional importance of ICAM-1 in the mechanism of neutrophil-induced liver injury in bile duct-ligated mice. Am. J. Physiol. Gastrointest. Liver Physiol. 286, G499-G507.
Hill, A.B. (1965). The environment and disease: association or causation? Proc. R. Soc. Med. 58, 295-300.
Hofmann, A.F. (2009). Bile acids and the enterohepatic circulation. In The Liver: Biology and Pathobiology (I.M.; Arias, H.J., Alter, J.L., Boyer, D.E., Cohen, N., Fausto, D.A., Shafritz, A.W., Wolkoff Eds.), pp. 289-304. Wiley-Blackwell, Oxford.
http://caat.jhsph.edu/ (consulted June 2013).
http://www.seurat-1.eu/ (consulted June 2013).
Jaeschke, H., Hasegawa, T. (2006). Role of neutrophils in acute inflammatory liver injury. Liver Int. 26, 912-919.
Jemnitz, K., Veres, Z., Vereczkey, L. (2010). Contribution of high basolateral bile salt efflux to the lack of hepatotoxicity in rat in response to drugs inducing cholestasis in human. Toxicol. Sci. 115, 80-88.
Julien, E., Boobis, A.R., Olin, S.S. (2009). The key events dose-response framework: a cross-disciplinary mode-of-action based approach to examining dose-response and thresholds. Crit. Rev. Food Sci. Nutr. 49, 682-689.
Jung, D., Mangelsdorf, D.J., Meyer, U.A. (2006). Pregnane X receptor is a target of farnesoid X receptor. J. Biol. Chem. 281, 19081-19091.
Jung, D., Elferink, M.G., Stellaard, F., Groothuis, G.M. (2007). Analysis of bile acid-induced regulation of FXR target genes in human liver slices. Liver Int. 27, 137-144.
Kast, H.R., Goodwin, B., Tarr, P.T., Jones, S.A., Anisfeld, A.M., Stoltz, C.M., Tontonoz, P., Kliewer, S., Willson, T.M., Edwards, P.A. (2002). Regulation of multidrug resistance-associated protein 2 (ABCC2) by the nuclear receptors pregnane X receptor, farnesoid X-activated receptor, and constitutive androstane receptor. J. Biol. Chem. 277, 2908-2915.
Kim, M.S., Shigenaga, J., Moser, A., Grunfeld, C., Feingold, K.R. (2004). Suppression of DHEA sulfotransferase (Sult2A1) during the acute-phase response. Am. J. Physiol. Endocrinol. Metab. 287, E731-E738.
Kis, E., Ioja, E., Rajnai, Z., Jani, M., Méhn, D., Herédi-Szabó, K., Krajcsi, P. (2012). BSEP inhibition: in vitro screens to assess cholestatic potential of drugs. Toxicol. In Vitro 26, 1294-1299.
Knisely, A.S., Strautnieks, S.S., Meier, Y., Stieger, B., Byrne, J.A., Portmann, B.C., Bull, L.N., Pawlikowska, L., Bilezikçi, B., Ozçay, F., László, A., Tiszlavicz, L., Moore, L., Raftos, J., Arnell, H., Fischler, B., Németh, A., Papadogiannakis, N., Cielecka-Kuszyk, J., Jankowska, I. (2006). Hepatocellular carcinoma in ten children under five years of age with bile salt export pump deficiency. Hepatology 44, 478-486.
Kocarek, T.A., Shenoy, S.D., Mercer-Haines, N.A., Runge-Morris, M. (2002). Use of dominant negative nuclear receptors to study xenobiotic-inducible gene expression in primary cultured hepatocytes. J. Pharmacol. Toxicol. Methods 47, 177-187.
Kostrubsky, V.E., Vore, M., Kindt, E., Burliegh, J., Rogers, K., Peter, G., Altrogge, D., Sinz, M.W. (2001). The effect of troglitazone biliary excretion on metabolite distribution and cholestasis in transporter-deficient rats. Drug Metab. Dispos. 29, 1561-1566.
Krasowski, M.D., Yasuda, K., Hagey, L.R., Schuetz, E.G. (2005). Evolution of the pregnane X receptor: adaptation to cross-species differences in biliary bile salts. Mol. Endocrinol. 19, 1720-1739.
Kuntz, E., Kuntz, H.D. (2008). Cholestasis. In Hepatology: Textbook and Atlas : History, Morphology, Biochemistry, Diagnostics, Clinic, Therapy 3rd edition (E. Kuntz E., H.-D. Kuntz Eds.), pp. 235-250. Springer, Heidelberg.
Landesmann, B., Goumenou, M., Munn, S., Whelan, M. (2012). Description of prototype modes-of-action related to repeated dose toxicity. JRC Scientific and Policy Report 75689.
Lang, C., Meier, Y., Stieger, B., Beuers, U., Lang, T., Kerb, R., Kullak-Ublick, G.A., Meier, P.J., Pauli-Magnus, C. (2007). Mutations and polymorphisms in the bile salt export pump and the multidrug resistance protein 3 associated with drug-induced liver injury. Pharmacogenet. Genomics 17, 47-60.
Lang, T., Haberl, M., Jung, D., Drescher, A., Schlagenhaufer, R., Keil, A., Mornhinweg, E., Stieger, B., Kullak-Ublick, G.A., Kerb, R. (2006). Genetic variability, haplotype structures, and ethnic diversity of hepatic transporters MDR3 (ABCB4) and bile salt export pump (ABCB11). Drug Metab. Dispos. 34, 1582-1599.
Lecureur, V., Sun, D., Hargrove, P., Schuetz, E.G., Kim, R.B., Lan, L.B., Schuetz, J.D. (2000). Cloning and expression of murine sister of P-glycoprotein reveals a more discriminating transporter than MDR1/P-glycoprotein. Mol. Pharmacol. 57, 24-35.
Lee, J., Azzaroli, F., Wang, L., Soroka, C.J., Gigliozzi, A., Setchell, K.D., Kramer, W., Boyer, J.L. (2001). Adaptive regulation of bile salt transporters in kidney and liver in obstructive cholestasis in the rat. Gastroenterology 121, 1473-1484.
Li-Masters, T., Morgan, E.T. (2001). Effects of bacterial lipopolysaccharide on phenobarbital-induced CYP2B expression in mice. Drug Metab. Dispos. 29, 252-257.
Lucena, M.I., Andrade, R.J., Kaplowitz, N., García-Cortes, M., Fernández, M.C., Romero-Gomez, M., Bruguera, M., Hallal, H,. Robles-Diaz, M., Rodriguez-González, J.F., Navarro, J.M., Salmeron, J., Martinez-Odriozola, P., Pérez-Alvarez, R., Borraz, Y., Hidalgo, R. (2009). Phenotypic characterization of idiosyncratic drug-induced liver injury: the influence of age and sex. Hepatology 49, 2001-2009.
Maher, J.M., Cheng, X., Slitt, A.L., Dieter, M.Z., Klaassen, C.D. (2005). Induction of the multidrug resistance-associated protein family of transporters by chemical activators of receptor-mediated pathways in mouse liver. Drug Metab. Dispos. 33, 956-962.
Mazzon, E., Cuzzocrea, S. (2003). Role of iNOS in hepatocyte tight junction alteration in mouse model of experimental colitis. Cell. Mol. Biol. 49, 45-57.
Meek, M.E., Bucher, J.R., Cohen, S.M., Dellarco, V., Hill, R.N., Lehman-McKeeman, L.D., Longfellow, D.G., Pastoor, T., Seed, J., Patton, D.E. (2003). A framework for human relevance analysis of information on carcinogenic modes of action. Crit. Rev. Toxicol. 33, 591-653.
Meier, Y., Pauli-Magnus, C., Zanger, U.M., Klein, K., Schaeffeler, E., Nussler, A.K., Nussler, N., Eichelbaum, M., Meier, P.J., Stieger, B. (2006). Interindividual variability of canalicular ATP-binding-cassette (ABC)-transporter expression in human liver. Hepatology 44, 62-74.
Mennone, A., Soroka, C.J., Harry, K.M., Boyer, J.L. (2010). Role of breast cancer resistance protein in the adaptive response to cholestasis. Drug Metab. Dispos. 38, 1673-1678.
Morgan, R.E., Trauner, M., van Staden, C.J., Lee, P.H., Ramachandran, B., Eschenberg, M., Afshari, C.A., Qualls, C.W. Jr., Lightfoot-Dunn, R., Hamadeh, H.K. (2010). Interference with bile salt export pump function is a susceptibility factor for human liver injury in drug development. Toxicol. Sci. 118, 485-500.
NRC. (2007). Toxicity testing in the 21st century: a vision and a strategy. The National Academies Press, Washington DC.
OECD. (2011). Report of the workshop on using mechanistic information on forming chemical categories. Series on testing and assessment No. 138.
OECD. (2012a). Proposal for a template and guidance on developing and assessing the completeness of adverse outcome pathways.
OECD. (2012b). The adverse outcome pathway for skin sensitization initiated by covalent binding to proteins part I: scientific evidence. Series on testing and assessment No. 168.
Oude Elferink, R.P., Paulusma, C.C., Groen, A.K. (2006). Hepatocanalicular transport defects: pathophysiologic mechanisms of rare diseases. Gastroenterology 13, 908-925.
Ozer, J., Ratner, M., Shaw, M., Bailey, W., Schomaker, S. (2008). The current state of serum biomarkers of hepatotoxicity. Toxicology 245, 194-205.
Padda, M.S., Sanchez, M., Akhtar, A.J., Boyer, J.L. (2011). Drug-induced cholestasis. Hepatology 53, 1377-1387.
Pagani, R., Portolés, M.T., De La Viña, S., Melzner, I., Vergani, G. (2003). Alterations induced on cytoskeleton by Escherichia coli endotoxin in different types of rat liver cell cultures. Histol. Histopathol. 1, 837-848.
Pauli-Magnus, C., Meier, P.J. (2006). Hepatobiliary transporters and drug-induced cholestasis. Hepatology 44, 778-787.
Pauwels, M., Rogiers, V. (2010). Human health safety evaluation of cosmetics in the EU: a legally imposed challenge to science. Toxicol. Appl. Pharmacol. 243, 260-274.
Roman, I.D., Monte, M.J., Gonzalez-Buitrago, J.M., Esteller, A., Jimenez, R. (1990). Inhibition of hepatocytary vesicular transport by cyclosporin A in the rat: relationship with cholestasis and hyperbilirubinemia. Hepatology 12, 83-91.
Salgia, R., Becker, J.H., Sayeed, M.M. (1993). Altered membrane fluidity in rat hepatocytes during endotoxic shock. Mol. Cell. Biochem. 121, 143-148.
Schoemaker, M.H., Conde de la Rosa, L., Buist-Homan, M., Vrenken, T.E., Havinga, R., Poelstra, K., Haisma, H.J., Jansen, P.L., Moshage, H. (2004). Tauroursodeoxycholic acid protects rat hepatocytes from bile acid-induced apoptosis via activation of survival pathways. Hepatology 39, 1563-1573.
Seed, J., Carney, E.W., Corley, R.A., Crofton, K.M., DeSesso, J.M., Foster, P.M., Kavlock, R., Kimmel, G., Klaunig, J., Meek, M.E., Preston, R.J., Slikker, W. Jr., Tabacova, S., Williams, G.M., Wiltse, J., Zoeller, R.T., Fenner-Crisp, P., Patton, D.E. (2005). Overview: using mode of action and life stage information to evaluate the human relevance of animal toxicity data. Crit. Rev. Toxicol. 35, 664-672.
Sekine, S., Yano K.,, Saeki, J., Hashimoto, N., Fuwa, T., Horie, T. (2010). Oxidative stress is a triggering factor for LPS-induced Mrp2 internalization in the cryopreserved rat and human liver slices. Biochem. Biophys. Res. Commun. 399, 279-285.
Seok, J., Warren, H.S., Cuenca, A.G., Mindrinos, M.N., Baker, H.V., Xu, W., Richards, D.R., McDonald-Smith, G.P., Gao, H., Hennessy, L., Finnerty, C.C., López, C.M., Honari, S., Moore, E.E., Minei, J.P., Cuschieri, J., Bankey, P.E., Johnson, J.L., Sperry, J., Nathens, A.B., Billiar, T.R., West, M.A., Jeschke, M.G., Klein, M.B., Gamelli, R.L., Gibran, N.S., Brownstein, B.H., Miller-Graziano, C., Calvano, S.E., Mason, P.H., Cobb, J.P., Rahme, L.G., Lowry, S.F., Maier, R.V., Moldawer, L.L., Herndon, D.N., Davis, R.W., Xiao, W., Tompkin, R.G. (2013). Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl. Acad. Sci. USA 110, 3507-3512.
Sokol, R.J., Dahl, R., Devereaux, M.W., Yerushalmi, B., Kobak, G.E., Gumpricht, E. (2005). Human hepatic mitochondria generate reactive oxygen species and undergo the permeability transition in response to hydrophobic bile acids. J. Pediatr. Gastroenterol. Nutr. 41, 235-243.
Trauner, M., Arrese, M., Lee, H., Boyer, J.L., Karpen, S.J. (1998a). Endotoxin downregulates rat hepatic ntcp gene expression via decreased activity of critical transcription factors. J. Clin. Invest. 101, 2092-2100.
Trauner, M., Meier, P.J., Boyer, J.L. (1998b). Molecular pathogenesis of cholestasis. N. Engl. J. Med. 339, 1217-1227.
US EPA. (2005). Guidelines for carcinogen risk assessment. Washington DC.
Vinken, M., Pauwels, M., Ates, G., Vivier, M., Vanhaecke, T., Rogiers, V. (2012). Screening of repeated dose toxicity data present in SCC(NF)P/SCCS safety evaluations of cosmetic ingredients. Arch. Toxicol. 86, 405-412.
Wagner, M., Zollner, G., Trauner, M. (2009). New molecular insights into the mechanisms of cholestasis. J. Hepatol. 51, 565-580.
Woolbright, B.L., Jaeschke, H. (2012). Novel insight into mechanisms of cholestatic liver injury. World. J. Gastroenterol. 18, 4985-4993.
Yasumiba, S., Tazuma, S., Ochi, H., Chayama, K., Kajiyama, G. (2001). Cyclosporin A reduces canalicular membrane fluidity and regulates transporter function in rats. Biochem. J. 354, 591-596.
Zhang, Y., Hong, J.Y., Rockwell, C.E., Copple, B.L., Jaeschke, H., Klaassen, C.D. (2012). Effect of bile duct ligation on bile acid composition in mouse serum and liver. Liver Int. 32, 58-69.
Zollner, G., Wagner, M., Moustafa, T., Fickert, P., Silbert, D., Gumhold, J., Fuchsbichler, A., Halilbasic, E., Denk, H., Marschall, H.U., Trauner, M. (2006). Coordinated induction of bile acid detoxification and alternative elimination in mice: role of FXR-regulated organic solute transporter-alpha/beta in the adaptive response to bile acids. J. Physiol. Gastrointest. Liver Physiol. 290, G923-G932.
Zollner, G., Trauner, M. (2006). Molecular mechanisms of cholestasis. Wien. Med. Wochenschr. 156, 380-385.
Zollner, G., Trauner, M. (2008). Mechanisms of cholestasis. Clin. Liver Dis. 12, 1-26.
Confidence in the AOP
Information from this section should be moved to the Key Event Relationship pages! 1. How well characterized is the AOP? Drug-induced cholestasis is a well understood AO that is causally and dose-dependently linked to BSEP inhibition, being the predominant MIE (Dawson et al., 2011; Kis et al., 2012; Morgan et al., 2010). Furthermore, the critical role of the key events, namely the accumulation of bile, the induction of inflammation and oxidative stress and the activation of specific nuclear receptors, as well as the different intermediate steps in the AOP, is supported by a wealth of experimental data. Thus, despite a number of limitations in scientific evidence, as will be discussed further, the established general structure and components of the AOP can be considered as being well-characterized.
2. How well are the initiating and other key events causally linked to the outcome? It has been demonstrated on numerous occasions that BSEP inhibition is causally linked to the induction of cholestasis in a dose-dependent way, both in experimental animals and in humans (Zollner and Trauner, 2006 and 2008; Wagner et al., 2009). BSEP inhibition directly leads to bile accumulation, which subsequently activates an inflammatory reaction as well as the occurrence of oxidative stress (Gujral et al., 2003 and 2004; Jaeschke and Hasegawa, 2006; Woolbright and Jaeschke, 2012). The resulting cell death and associated bile acid-induced membrane damage of hepatocytes and cholangiocytes underlie the increased serum concentrations of ALT, AST, ALP, GGT and 5’-NT, being a prominent clinical hallmark of cholestasis (Hofmann, 2009; Kuntz and Kuntz, 2008; Padda et al., 2011). To compensate for the cholestatic insults, an adaptive response is induced, which is initiated by nuclear receptor activation and that is targeted towards the elimination of bile from the organism (Boyer, 2009; Zollner and Trauner, 2006 and 2008; Wagner et al., 2009). Consequently, elevated concentrations of bilirubin in serum and urine are observed. The former causes jaundice, while the increased presence of bile acids in serum is thought to induce pruritus (Hofmann, 2009; Kuntz and Kuntz, 2008; Padda et al., 2011; Zollner and Trauner, 2008). Thus, there is a direct, causal and, at least in some cases, quantitative association between the MIE, the key events and the AO in the established AOP.
3. What are the limitations in the evidence in support of the AOP? There are a number of limitations in the scientific evidence in support of the AOP in relation to temporal concordance of the intermediate steps and key events, the consistency of the available experimental data, alternative mechanisms involved, interspecies and intraspecies differences, uncertainties, inconsistencies and data gaps. These shortcomings are addressed in previous sections.
4. Is the AOP specific to certain tissues, life stages or age classes? Although the entire process of drug-induced cholestasis mainly takes place in the liver, more specifically in hepatocytes, the adaptive changes to this insult also occur in other tissues, including the kidney and the intestine. In this context, expression of MRP2 is induced in renal tubular cells in experimental models of cholestasis (Lee et al., 2001). Alterations in transporter protein expression during cholestasis also occur in other tissues, such as the ileum (Mennone et al., 2010). Like in the liver, the overall goal of these alterations is to increase elimination of bile salts via the urine and feces (Zollner and Trauner, 2006 and 2008; Wagner et al., 2009). Regarding age-specificity, no significant quantitative differences in BSEP expression between fetal and human liver have been detected (Chen et al., 2005). Hepatocellular accumulation of bile acids causes giant cell hepatitis and progressive liver damage in children (Oude Elferink et al., 2006), which may burgeon into hepatocellular carcinoma (Bernstein et al., 2005; Knisely et al., 2006). On the other hand, it is well established that age over 50 years poses an increased risk to develop drug-induced hepatic damage (Pauli-Magnus and Meier, 2006) and that DILI in elder people is of cholestatic rather than of hepatocelluar nature (Lucena et al., 2009). In general, women are more susceptible for developing DILI than men (Pauli-Magnus and Meier, 2006), yet no gender differences exist in liver BSEP expression in humans (Cheng et al., 2007). In contrast, male rats are more prone to troglitazone-induced cholestasis than female rats because of higher rates of troglitazone sulphate formation (Funk et al., 2001; Kostrubsky et al., 2001). At the population level, genetic variability in the BSEP gene, leading to its decreased expression, may predispose different ethnic populations to drug-induced cholestasis (Lang et al., 2006 and 2007; Meier et al., 2006).
5. Are the initiating and key events expected to be conserved across taxa? Standard animal studies conducted during drug development, using mainly rodents, usually pick up about half of all human hepatotoxic compounds because of interspecies differences (Blomme et al., 2009; Ozer et al., 2008). In the case of BSEP inhibition, however, interspecies differences are mostly of quantitative nature. This could be due to the fact that there is a high amino acid similarity between human BSEP and its rodent counterparts, namely 80% in mouse and 82% in rat (Green et al., 2000; Lecureur et al., 2000). Accordingly, while IC50 values for BSEP inhibition differ only minimally between human and mouse for troglitazone, they differ by almost an order of magnitude for glibenclamide (Kis et al., 2012). Other reasons for the absence of hepatotoxicity induced by human-relevant cholestatic drugs in rats include higher rates of basolateral bile salt efflux, which could represent an additional protective mechanism against cholestasis (Jemnitz et al., 2010). In addition to BSEP inhibition, the key events of the proposed AOP are expected to be generally well conserved among taxa. Nevertheless, a recent report showed that considerable differences exist in inflammatory responses between human and mouse (Seok et al., 2013). Despite the occurrence of interspecies differences in their expression or ligand binding, such as shown for PXR (Krasowski et al., 2005), activation of nuclear receptors is a critical event in different animal models of cholestasis (Wagner et al., 2009; Zollner and Trauner, 2006 and 2008). It remains to be established whether data included in the AOP can be extrapolated from animals to humans and vice versa.