
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 282
Title
Adverse outcome pathway on photochemical toxicity initiated by light exposure
Short name
Graphical Representation
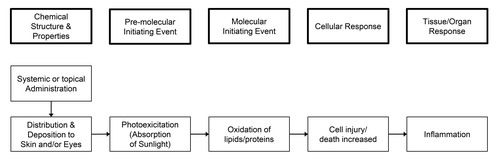
Point of Contact
Contributors
- Yoshiki Seto
- Satomi Onoue
- Evgeniia Kazymova
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.49 | Under Review |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| ROS generation from photoactivated chemicals | July 04, 2019 04:19 |
| Oxidation of membrane lipids | July 04, 2019 21:15 |
| Oxidation/denatuation of membrane proteins | July 04, 2019 21:19 |
| Inflamatory events in light-exposed tissues | July 04, 2019 21:25 |
| ROS generation leads to Oxidation of membrane lipids | July 09, 2019 05:54 |
| ROS generation leads to Oxidation/denatuation of membrane proteins | July 09, 2019 05:56 |
| Oxidation of membrane lipids leads to Inflammatory events | July 09, 2019 06:00 |
| Oxidation/denatuation of membrane proteins leads to Inflammatory events | July 09, 2019 06:03 |
| Light (290-700 nm) | February 18, 2019 20:30 |
| Photoreactive chemicals | March 25, 2019 22:35 |
| Reactive oxygen species | August 15, 2017 10:43 |
Abstract
Phototoxicity is an adverse reaction in the light-exposed tissues triggered by normally harmless doses of sunlight (Moore, 1998; Moore, 2002; Roberts, 2001). Recently, high-intensity UV rays from the sun have reached the Earth’s surface with the destruction of the ozone layer, and interest in phototoxic events has increased enormously. Notably, phototoxic reactions against exogenous agents are caused by the combined effects of environmental light and external agents, including drugs, cosmetics, and foods (Epstein, 1983; Stein and Scheinfeld, 2007).
In this AOP, the primary trigger for a compound to be considered with respect to potential to create photochemical and photobiological reactions is the absorption of photon energy from light ranging from 290 to 700 nm. The extent of absorption depends on the wavelength of light and the type of absorbing chromophores in the light-exposed tissues. A molecule is excited by absorption of photon energy, and the photoactivated molecule induces photochemical reactions via energy transfer (type I photochemical reaction) and free radical generation (type II photochemical reaction). These photochemical reactions result in generation of radicals and reactive oxygen species, and the reactive species react with biomolecules. Generated radicals of a target chemical bind to DNA and proteins, resulting in formation of these photo-adducts, and reactive oxygen species (ROS), including singlet oxygen and superoxide, induce oxidation of biomolecules. These key events bring inflammatory events in the light-exposed tissues (Brendler-Schwaab et al. , 2004; Epstein and Wintroub, 1985; Quintero and Miranda, 2000).
This AOP describes the pathway of photochemical toxicity between attack of ROS generated from photoactivated chemicals to membranes and inflammatory events in light-exposed tissues.
AOP Development Strategy
Context
The primary event in any photosensitization process can be the absorption of photons of the appropriate wavelength, which allows chromophore to reach an excited state. The excitation energy is often transferred to oxygen molecules, followed by generation of ROS. These appear to be the principal intermediate species in the phototoxic response. In cells, this cascade gives rise to local oxidative stress and damage to genomic DNA, proteins, and lipids within cell membranes. From the standpoint of risk assessment, ROS generation from photoirradiated chemicals has been considered to be one of key determinants in recognizing their phototoxic potential. The ROS assay has been designed to assess photoreactivity of pharmaceuticals, of which the principle is to monitor types I and II photochemical reactions of the test chemicals when exposed to simulated sunlight. This simple analytical test could be used to screen potential chemical scaffolds, leads, and candidate drugs to identify and/or select away from those having phototoxic potential. The validation study for the ROS assay has been carried out by the Japan Pharmaceutical Manufacturers Association (JPMA), supervised by the Japanese Center for the Validation of Alternative Methods (JaCVAM). The validation study indicates satisfactory outcomes in terms of transferability, intra- and inter-laboratory variability, and predictive capacity. Thus, a negative result in this ROS assay would indicate a very low probability of phototoxicity, whereas a positive result would be a flag for follow-up assessment. ROS assay was successfully adopted as ICH S10 guideline (2014) and OECD test guideline 495 (2019).
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1592 | ROS generation from photoactivated chemicals | ROS generation |
| KE | 1594 | Oxidation of membrane lipids | Oxidation of membrane lipids |
| KE | 1595 | Oxidation/denatuation of membrane proteins | Oxidation/denatuation of membrane proteins |
| AO | 1599 | Inflamatory events in light-exposed tissues | Inflammatory events |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| ROS generation leads to Oxidation of membrane lipids | adjacent | High | Low |
| ROS generation leads to Oxidation/denatuation of membrane proteins | adjacent | High | Low |
| Oxidation of membrane lipids leads to Inflammatory events | adjacent | High | Low |
| Oxidation/denatuation of membrane proteins leads to Inflammatory events | adjacent | High | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| human | Homo sapiens | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Mixed | High |
Overall Assessment of the AOP
The focus of this AOP is on photochemical toxicity, especially photoactivation of target chemicals followed by generation of ROS. ROS generated from photoirradiated chemicals can react with molecules on the membranes, including lipids and proteins, and the reactions may lead to inflammatory events in the UV-exposed tissues.
Phototoxicity is an adverse reaction triggered by normally harmless doses of sunlight. There are two types of photosensitive disorders, endogenous and exogenous phototoxicity, and the observable changes to the sunlight-exposed tissues are essentially detrimental, and include the following appearance; (i) immediate faint erythema during exposure, (ii) delayed erythemal responses, (iii) abnormal keratinisation and vacuolated cells, (iv) formation of desquamating layer, and (v) desquamation (peeling) (Moore, 1998; Moore, 2002; Roberts, 2001). Recently, high-intensity UV rays from the sun have reached the Earth’s surface with the destruction of the ozone layer, and interest in phototoxic events has increased enormously. Notably, phototoxic reactions against exogenous agents are caused by the combined effects of UV irradiation and external agents, including drugs, cosmetics and foods (Stein and Scheinfeld, 2007). Phototoxic skin responses after administration of photosensitive drugs, so-called drug-induced phototoxicity, have been recognized as undesirable side effects, and several classes of drugs, even when not toxic by themselves, may become reactive under exposure to environmental light, inducing undesired phototoxic responses (Epstein, 1983).
The primary trigger for a compound to be considered with respect to potential to create photochemical and photobiological reaction is the absorption of UV and visible light ranging from 290 to 700 nm. The extent of absorption depends on the wavelength of light and the type of absorbing chromophores in the UV-exposed tissues. UV radiation is usually divided into several ranges based on its physiologic effects: (1) UVA (near UV): 320–400 nm (UVA I: 340–400 nm and UVA II: 320–340 nm), (2) UVB (middle UV): 290–320 nm, and (3) UVC (far UV): 180–290 nm (Svensson et al., 2001; Vassileva et al., 1998). The sun emits ultraviolet radiation in the UVA, UVB, and UVC bands, but because of absorption by the atmosphere's ozone layer, the main ultraviolet radiation that reaches the Earth's surface is UVA (Dubakiene and Kupriene, 2006). Absorption of light through the skin and eyes, primarily in the 290–700 nm range, varies with wavelength, such that light in the red region of the spectrum reaches well into the subcutis layer; whereas at 300 nm or shorter wavelength, only an estimated 10% passes through the epidermis (Epstein, 1989). Thus, penetration and absorption of light in the UV-exposed tissues is important factor in drug-induced phototoxicity as Grotthus-Draper law of photobiology states; only light that is absorbed can be active in photochemical and photobiological processes.
When a drug molecule absorbs a photon energy, electrons can be prompted from occupied orbitals (the ground state) to an unoccupied orbital (S1, S2) depending upon bond type and associated energy level. Furthermore, unpaired singlet state electrons (opposite spin) may be converted to triplet state (parallel spin) by inversion of the spin via intersystem crossing of the absorbed energy. To return to the ground state from S1, S2/T1, T2, energy must be dissipated by internal conversion, fluorescence (from singlet state), phosphorescence (from triplet state) or via chemical reaction, giving rise to photoproducts and/or potential external reactions with biomolecules.
In addition, molecular oxygen, a triplet radical in its ground state, appears to be the predominant acceptor of excitation energy as its lowest excited level (singlet state) has a comparatively low value. An energy transfer from excited triplet photosensitizer to the oxygen (type II photochemical reaction) could produce excited singlet oxygen which might, in turn, participate in a lipid- and protein-membrane oxidation or induce DNA damage. An electron or hydrogen transfer could lead to the formation of free radical species (type I photochemical reaction), producing a direct attack on the biomolecules or in the presence of oxygen, to evolve towards secondary free radicals such as peroxyl radicals or the very reactive hydroxyl radical, a known intermediate in the oxidative damage of biomolecules. This toxic pathway corresponds to successive reactions which involve the appearance of superoxide anion radical, its dismutation to from hydrogen peroxide followed with the hydrogen peroxide reduction to form hydroxyl radical. Herein, excitation of the drug by light may give rise to ROS such as singlet oxygen and superoxide, which may be one of causative molecules for the drug-induced phototoxicity (Brendler-Schwaab et al., 2004; Epstein and Wintroub, 1985).
Domain of Applicability
Chemicals: This AOP applies to a wide range of chemicals. Phototoxic chemicals are recognized to have following characteristics: (i) absorption of light within the range of natural sunlight (290-700 nm); (ii) generation of a reactive species following absorption of UV-visible light; (iii) distribution to light-exposed tissues (e.g., skin and eye) (ICH S10).
Sex: This AOP applies to both males and females.
Life stages: The relevant life stages for this AOP are all life stages after born.
Taxonomic: This AOP mainly applies to human.
Essentiality of the Key Events
The essentiality of KEs for this AOP was rated high on the basis of experimental evidence in the investigations related to each of KEs and published guidelines. For details see the table on “Support for Essentiality of KEs”.
Evidence Assessment
Support for biological plausibility of KERs
|
MIE => KE 1 |
Generated ROS from photoactivated chemicals can react with membrane lipids, and oxidation of membrane lipids could be occurred. |
Biological Plausibility of the MIE => KE 1 is high. The relationship between MIE and KE 1 is consistent with chemical and biological knowledge (Girotti, 1990; Girotti, 2001; Onoue and Tsuda, 2006). |
|
MIE => KE 2 |
Generated ROS from photoactivated chemicals can react with membrane proteins, and oxidation/denaturation of membrane proteins could be occurred. |
Biological Plausibility of the MIE => KE 2 is high.
The relationship between MIE and KE 2 is consistent with biological knowledge (Dalle Carbonare and Pathak, 1992; Valenzeno, 1987). |
|
KE 1 => AO |
Oxidation of membrane lipids relates with damage produced in the cellular membrane, leading to inflammatory events. |
Biological Plausibility of the KE 1 => AO is high.
The relationship between KE 1 and AO is consistent with biological knowledge (Castell et al., 1994). |
|
KE 2 => AO |
Oxidation/denaturation of protein provides the necrosis of the living tissues as an inflammatory event. |
Biological Plausibility of the KE 2 => AO is high.
The relationship between KE 2 and AO is consistent with biological knowledge (Dalle Carbonare and Pathak, 1992; Opie, 1962). |
Support for Essentiality of KEs
|
MIE |
ROS generation from photoactivated chemicals |
High; well-accepted generation of reactive oxygen species from photo-activated chemicals associated with phototoxic reactions with 200 of chemicals evaluated in qualitative endpoints (Onoue et al., 2014; Onoue et al., 2013a; Onoue et al., 2008a; Onoue and Tsuda, 2006; Seto et al., 2013b). The event has described in ICH S10 guideline as a crucial factor of phototoxic reactions (ICH, 2014). |
|
KE 1 |
Oxidation of membrane lipids
|
High; Oxidative stress to lipids associated with the phototoxic reactions (Girotti, 1990; Girotti, 2001; Onoue and Tsuda, 2006). |
|
KE 2 |
Oxidation/denaturation of membrane proteins |
High; accepted oxidation/denaturation of proteins associated with the phototoxic reactions (Dalle Carbonare and Pathak, 1992; Valenzeno, 1987). |
|
Adverse outcome |
Inflammatory events in sunlight-exposed tissues |
Photoreactive agents indicated inflammatory events, including edema, dyskeratosis, and necrosis, in light-exposed tissues after sunlight exposure (Moore, 1998; Moore 2002; Roberts, 2001). |
Empirical Support for KERs
|
MIE => KE 1: ROS generation leads to Oxidation of membrane lipids |
Empirical support of the MIE => KE 1 is strong. Rationale: Lipid peroxidation was occurred by ROS-generating chemicals under exposure to simulated sunlight (Onoue et al., 2011, Onoue and Tsuda, 2006). A photoreactive chemical indicated dose-dependent increases in ROS generation and lipid peroxidation after exposure to a fixed dose of simulated sunlight (Seto et al., 2013a). |
|
MIE => KE 2: ROS generation leads to Oxidation/denaturation of membrane proteins |
Empirical support of the MIE => KE 2 is moderate. Rationale: ROS generated from photosensitizing agents led to oxidation and denaturation of proteins (Dalle Carbonare and Pathak, 1992). |
|
KE 1 => AO: Oxidation of membrane lipids leads to Inflammatory events |
Empirical support of the KE 1=> AO is strong. Rationale: Increases in lipid peroxidation and inflammatory-related cytokines were observed in the murine skin, and naringenin, an anti-oxidant, attenuated these increases in a dose-dependent manner (Martinez et al., 2015). Benzoyl peroxide, a ROS generator, led to lipid peroxidation and GSH depletion, and the changes caused the gene expression of pro-inflammatory cytokines (Valacchi et al., 2001). |
|
KE 2 => AO: Oxidation/denaturation of membrane proteins leads to Inflammatory events |
Empirical support of the KE 2=> AO is moderate. Rationale: Denaturation of proteins induced necrosis and inflammatory in the skin (Opie, 1962). |
Known Modulating Factors
Quantitative Understanding
Although there is empirical information on KERs as described above sections, the overall quantitative understanding of the AOP is insufficient to directly link a measure of KEs to a quantitative prediction of KERs.
As a pre-MIE, light absorption of chemicals is an important event for phototoxic reactions induced by photoreactive chemicals. Quantitative endpoint on absorption of light (290–700 nm) was recognized in the previous report (Henry et al., 2009), and, for photoreactive chemicals, the criterion on molar extinction coefficient (MEC) was determined to be 1,000 M-1·cm-1. Most of chemicals with MEC values of over 1,000 M-1·cm-1 generated significant ROS, including singlet oxygen and/or superoxide (Onoue et al., 2013b; Onoue and Tsuda, 2006), and the qualitative criteria on ROS generation was determined to evaluate chemical phototoxicity (Onoue et al., 2014; Onoue et al., 2013a; Onoue et al., 2008b).
Considerations for Potential Applications of the AOP (optional)
The MIE and KEs in this AOP could contribute to assays development for photosafety evaluation and an AOP-based IATA construction. AOP-based IATA can be applied for various aims including screening of chemicals, prioritization of chemicals for further testing, and risk assessment.
The regulatory applicability of the AOP would be to use experimental results from assays based on MIE and KEs as indictors for the risk of phototoxic reactions.
Combined use of photobiochemical properties and tissue exposure data would be of help for photosafety evaluation of chemicals. Risk assessment would be possible when exposure data in light-exposed tissues combined with assay data based on AOP.
References
Brendler-Schwaab S, Czich A, Epe B, Gocke E, Kaina B, Muller L, et al. Photochemical genotoxicity: principles and test methods. Report of a GUM task force. Mutation research. 2004;566:65-91.
Castell JV, Gomez-Lechon MJ, Grassa C, Martinez LA, Miranda MA, Tarrega P. Photodynamic lipid peroxidation by the photosensitizing nonsteroidal antiinflammatory drugs suprofen and tiaprofenic acid. Photochem Photobiol. 1994;59:35-9.
Dalle Carbonare M, Pathak MA. Skin photosensitizing agents and the role of reactive oxygen species in photoaging. J Photochem Photobiol B. 1992;14:105-24.
Dubakiene R, Kupriene M. Scientific problems of photosensitivity. Medicina (Kaunas). 2006;42:619-24.
Epstein JH. Phototoxicity and photoallergy in man. J Am Acad Dermatol. 1983;8:141-7.
Epstein JH. Photomedicine. New York: Plenum press; 1989.
Epstein JH, Wintroub BU. Photosensitivity due to drugs. Drugs. 1985;30:42-57.
Girotti AW. Photodynamic lipid peroxidation in biological systems. Photochem Photobiol. 1990;51:497-509.
Girotti AW. Photosensitized oxidation of membrane lipids: reaction pathways, cytotoxic effects, and cytoprotective mechanisms. J Photochem Photobiol B. 2001;63:103-13.
Henry B, Foti C, Alsante K. Can light absorption and photostability data be used to assess the photosafety risks in patients for a new drug molecule? J Photochem Photobiol B. 2009;96:57-62.
ICH. ICH Guideline S10 Guidance on Photosafety Evaluation of Pharmaceuticals.: International Council on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use; 2014.
Martinez RM, Pinho-Ribeiro FA, Steffen VS, Caviglione CV, Vignoli JA, Barbosa DS, et al. Naringenin Inhibits UVB Irradiation-Induced Inflammation and Oxidative Stress in the Skin of Hairless Mice. Journal of natural products. 2015;78:1647-55.
Moore DE. Mechanisms of photosensitization by phototoxic drugs. Mutation research. 1998;422:165-73.
Moore DE. Drug-induced cutaneous photosensitivity: incidence, mechanism, prevention and management. Drug safety. 2002;25:345-72.
Onoue S, Hosoi K, Toda T, Takagi H, Osaki N, Matsumoto Y, et al. Intra-/inter-laboratory validation study on reactive oxygen species assay for chemical photosafety evaluation using two different solar simulators. Toxicology in vitro : an international journal published in association with BIBRA. 2014;28:515-23.
Onoue S, Hosoi K, Wakuri S, Iwase Y, Yamamoto T, Matsuoka N, et al. Establishment and intra-/inter-laboratory validation of a standard protocol of reactive oxygen species assay for chemical photosafety evaluation. Journal of applied toxicology : JAT. 2013a;33:1241-50.
Onoue S, Igarashi N, Yamada S, Tsuda Y. High-throughput reactive oxygen species (ROS) assay: an enabling technology for screening the phototoxic potential of pharmaceutical substances. J Pharm Biomed Anal. 2008a;46:187-93.
Onoue S, Kawamura K, Igarashi N, Zhou Y, Fujikawa M, Yamada H, et al. Reactive oxygen species assay-based risk assessment of drug-induced phototoxicity: classification criteria and application to drug candidates. J Pharm Biomed Anal. 2008b;47:967-72.
Onoue S, Seto Y, Ochi M, Inoue R, Ito H, Hatano T, et al. In vitro photochemical and phototoxicological characterization of major constituents in St. John's Wort (Hypericum perforatum) extracts. Phytochemistry. 2011;72:1814-20.
Onoue S, Suzuki G, Kato M, Hirota M, Nishida H, Kitagaki M, et al. Non-animal photosafety assessment approaches for cosmetics based on the photochemical and photobiochemical properties. Toxicology in vitro : an international journal published in association with BIBRA. 2013b;27:2316-24.
Onoue S, Tsuda Y. Analytical studies on the prediction of photosensitive/phototoxic potential of pharmaceutical substances. Pharmaceutical research. 2006;23:156-64.
Opie EL. On the relation of necrosis and inflammation to denaturation of proteins. The Journal of experimental medicine. 1962;115:597-608.
Quintero B, Miranda MA. Mechanism of photosensitization induced by drugs: A general survey. Ars Pharmaceutica. 2000;41:27-46.
Roberts JE. Ocular phototoxicity. J Photochem Photobiol B. 2001;64:136-43.
Seto Y, Inoue R, Kato M, Yamada S, Onoue S. Photosafety assessments on pirfenidone: photochemical, photobiological, and pharmacokinetic characterization. J Photochem Photobiol B. 2013a;120:44-51.
Seto Y, Kato M, Yamada S, Onoue S. Development of micellar reactive oxygen species assay for photosafety evaluation of poorly water-soluble chemicals. Toxicology in vitro : an international journal published in association with BIBRA. 2013b;27:1838-46.
Stein KR, Scheinfeld NS. Drug-induced photoallergic and phototoxic reactions. Expert Opin Drug Saf. 2007;6:431-43.
Svensson CK, Cowen EW, Gaspari AA. Cutaneous drug reactions. Pharmacological reviews. 2001;53:357-79.
Valacchi G, Rimbach G, Saliou C, Weber SU, Packer L. Effect of benzoyl peroxide on antioxidant status, NF-kappaB activity and interleukin-1alpha gene expression in human keratinocytes. Toxicology. 2001;165:225-34.
Valenzeno DP. Photomodification of biological membranes with emphasis on singlet oxygen mechanisms. Photochem Photobiol. 1987;46:147-60.
Vassileva SG, Mateev G, Parish LC. Antimicrobial photosensitive reactions. Archives of internal medicine. 1998;158:1993-2000.