
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 285
Title
Inhibition of N-linked glycosylation leads to liver injury
Short name
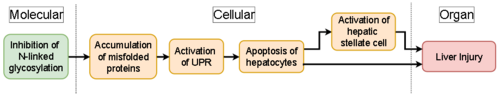
Graphical Representation
Point of Contact
Contributors
- Marvin Martens
- Kirsten Snijders
- Arthur Author
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition of N-linked glycosylation | March 06, 2019 07:44 |
| Accumulation of misfolded proteins | March 06, 2019 07:49 |
| Unfolded Protein Response | September 14, 2023 08:51 |
| Apoptosis | February 28, 2024 09:40 |
| Activation of hepatic stellate cells | March 06, 2019 07:50 |
| Increased, Liver Steatosis | May 21, 2024 10:00 |
| Liver Injury | March 07, 2019 04:23 |
| Inhibition of N-linked glycosylation leads to Accumulation, misfolded proteins | March 06, 2019 08:07 |
| Accumulation, misfolded proteins leads to Unfolded Protein Response | March 06, 2019 08:06 |
| Unfolded Protein Response leads to Apoptosis | March 06, 2019 08:09 |
| Apoptosis leads to Activation, hepatic stellate cells | March 06, 2019 08:10 |
| Activation, hepatic stellate cells leads to Liver Injury | March 06, 2019 08:10 |
| Apoptosis leads to Liver Injury | March 06, 2019 08:11 |
Abstract
The Endoplasmic Reticulum (ER) is responsible for protein synthesis and folding making it the main target for the unfolded protein response (UPR). Cell stress can induce an increase in misfolded proteins that lead to the activation of the UPR through upregulation of sensors, transcription factors and downstream targets to recover homeostasis and control the levels of unfolded proteins in the ER. This adverse outcome pathway (AOP) outlines the way in which inhibition of N-linked glycosylation activates and disrupts the UPR leading to livery injury. It will provide more in depth knowledge on the thresholds involved during the UPR for the maintenance of homeostasis and the induction of the adverse outcome on the target organ of the Liver.
All newly synthesized proteins undergo glycosylation before they are folded in the ER. Any misfolding of proteins is resolved by the ER- associated degradation (ERAD) that recognizes and clears misfolded proteins from the ER. This quality control of protein folding is glycosylation directed. Misfolded proteins that are not N-linked glycosylated fail to be recognized by the ERAD.
The molecular initiating event for this AOP is the inhibition of N-linked glycosylation. This can be achieved through directly inhibiting either the biosynthesis or the processing of N-linked oligosaccharide chains. Enzymes that synthesize N-linked oligosaccharide chain are often targets for inhibition of glycosylation. Unglycosylated misfolded proteins are unable to be recognized or cleared by the ERAD thus leading to key event 1, a buildup of misfolded proteins. This accumulation activates sensors and triggers key event 2: the (UPR).
Whilst the UPR is in place to maintain homeostasis and resolve the buildup of misfolded proteins in the ER. The activation of the UPR coupled with an inability to resolve the buildup of misfolded protein, through compromised ERAD clearance, leads to key event 3: apoptosis of hepatocytes, triggered by UPR downstream target CHOP. This in turn will lead to the adverse outcome: Liver Injury.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1604 | Inhibition of N-linked glycosylation | Inhibition of N-linked glycosylation |
| KE | 1605 | Accumulation of misfolded proteins | Accumulation, misfolded proteins |
| KE | 1512 | Unfolded Protein Response | Unfolded Protein Response |
| KE | 1262 | Apoptosis | Apoptosis |
| KE | 1606 | Activation of hepatic stellate cells | Activation, hepatic stellate cells |
| KE | 459 | Increased, Liver Steatosis | Increased, Liver Steatosis |
| AO | 1549 | Liver Injury | Liver Injury |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Inhibition of N-linked glycosylation leads to Accumulation, misfolded proteins | adjacent | Not Specified | Not Specified |
| Accumulation, misfolded proteins leads to Unfolded Protein Response | adjacent | Not Specified | Not Specified |
| Unfolded Protein Response leads to Apoptosis | adjacent | Not Specified | Not Specified |
| Apoptosis leads to Activation, hepatic stellate cells | adjacent | Not Specified | Not Specified |
| Activation, hepatic stellate cells leads to Liver Injury | adjacent | Not Specified | Not Specified |
| Apoptosis leads to Liver Injury | adjacent | Not Specified | Not Specified |