
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 315
Title
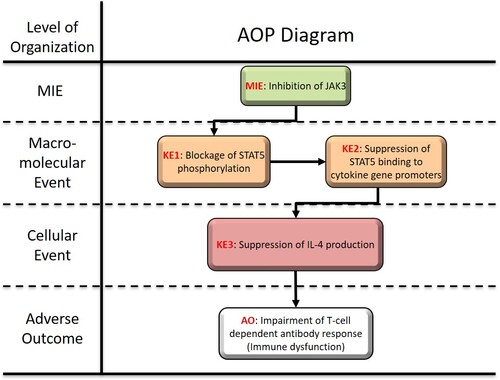
Inhibition of JAK3 leading to impairment of T-Cell Dependent Antibody Response
Short name
Graphical Representation
Point of Contact
Contributors
- Takumi Ohishi
- Yasuhiro Yoshida
- Brendan Ferreri-Hanberry
Coaches
- Shihori Tanabe
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.74 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition of JAK3 | February 15, 2021 06:25 |
| Blockade of STAT5 phosphorylation | February 15, 2021 06:27 |
| Suppression of STAT5 binding to cytokine gene promoters | February 24, 2021 06:52 |
| Suppression of IL-4 production | February 24, 2021 06:55 |
| Impairment of T-cell dependent antibody response | February 24, 2021 06:58 |
| Inhibition of JAK3 leads to STAT5 inhibition | November 03, 2021 12:44 |
| STAT5 inhibition leads to Suppression of STAT5 binding to cytokine gene promoters | November 03, 2021 12:53 |
| Suppression of STAT5 binding to cytokine gene promoters leads to Suppression of IL-4 production | March 30, 2021 11:07 |
| Suppression of IL-4 production leads to Impairment, TDAR | April 04, 2021 08:42 |
| PF-06651600 (CAS No:1792180-81-4), | September 15, 2020 05:38 |
| RB1 | September 15, 2020 05:38 |
Abstract
Signal transduction between immune-related cells depends in many cases on cytokines. The transduction involves cell surface cytokine receptors as well as direct cell-to-cell interaction. Cytokines influence the movement, proliferation, differentiation, and activation of lymphocytes and other leukocytes in a variety of ways. Some cytokine receptors require an activation step through a Janus-kinase (JAK)/signal transducer and activator of transcription (STAT) system. When cytokines bind to specific cytokine receptors, the receptors form dimers, which more closely resemble JAK molecules. JAK is activated and phosphorylates adjacent cytokine receptors. STATs bind to the phosphorylated receptor sites and are in turn phosphorylated by the activated JAK. The phosphorylated STAT is dimerized and translocated into the nucleus. There it binds to the promoter regions of cytokine genes, which initiates the transcription of these genes in the nucleus.
In mammals, four JAK families of enzymes (JAK1, JAK2, JAK3, and TYK2) and seven STATs (STAT1, STAT2, STAT3, STAT4, STAT5a, STAT5b, and STAT6) are utilized by more than 50 cytokines and growth factors to mediate intracellular signaling. In particular, pro-inflammatory cytokines such as interferon-γ (IFN-γ), interleukin-2 (IL-2), IL-4, IL-6, IL-13, IL-21, and IL-23 have been implicated in inflammatory diseases that utilize the JAK pathway. In addition, TH2 derived cytokines, including IL-31 and thymic stromal lymphopoietin (TSLP), are ligands for murine and human sensory nerves. These cytokines have critical roles in evoking itchiness. Because these cytokines also interact with JAK, several JAK inhibitors have received a lot of attention recently as therapeutic agents for major inflammatory diseases and pruritic diseases.
This proposed AOP consists of JAK3 inhibition as a MIE, blockade of STAT5 phosphorylation as the first key event (KE1), suppression of STAT5 binding to the promoter regions of cytokine genes as KE2, suppression of IL-4 production as KE3, and suppression of T cell dependent antibody response (TDAR) as an AO. This AOP especially focuses on the inhibition of JAK3, which is required for signal transduction by cytokines through the common γ chain of the receptors for IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21. In the proposed AOP, JAK3 selective inhibitors that include PF-06651600 (CAS No: 1792180-81-4) and the 4-aminopiperidine-based compound RB1 are stressors. STAT5 that is phosphorylated by JAK3 forms a homo-dimer that translocate to the nucleus and induces expressions of genes, such as IL-4. Therefore, JAK3 inhibition leads to the suppressed binding of STAT5 to the promoter regions of cytokine genes and the subsequent suppression of IL-4 production. Thus, JAK/STAT regulation plays an important role in the TDAR. TDAR is frequently affected by immunosuppressive conditions and is a major endpoint in many preclinical immunotoxicity studies.
AOP Development Strategy
Context
Although many stressors inhibit JAK3 activity, this AOP is based on immunosuppression caused by the recently developed and highly selective JAK3 inhibitors PF-06651600 and RB1. A significant body of scientific literature has been published concerning these two inhibitors. We look forward to future amendments to this AOP with up-to-date information on other stressors, which will clarify the link between inhibition of JAK activity and impairment of TDAR.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1715 | Inhibition of JAK3 | Inhibition of JAK3 |
| KE | 1716 | Blockade of STAT5 phosphorylation | STAT5 inhibition |
| KE | 1717 | Suppression of STAT5 binding to cytokine gene promoters | Suppression of STAT5 binding to cytokine gene promoters |
| KE | 1718 | Suppression of IL-4 production | Suppression of IL-4 production |
| AO | 1719 | Impairment of T-cell dependent antibody response | Impairment, TDAR |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Inhibition of JAK3 leads to STAT5 inhibition | adjacent | High | High |
| STAT5 inhibition leads to Suppression of STAT5 binding to cytokine gene promoters | adjacent | High | High |
| Suppression of STAT5 binding to cytokine gene promoters leads to Suppression of IL-4 production | adjacent | High | High |
| Suppression of IL-4 production leads to Impairment, TDAR | adjacent | High | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP

JAKs are a family of nonreceptor tyrosine kinases and consist of four members: JAK1, JAK2, JAK3, and Tyk2 (Johnston, et al. 1994). All four mediate signals initiated by cytokines through interactions with receptors for IL-2, IL-5, IL-7, IL-9, and IL-15 via the common γ chain (Witthuhn, et al. 1994). Different studies have shown that JAK3 is widely expressed in different organs (Witthuhn, et al. 1994). Previous studies with IL-2Rγ-null mice showed that JAK3 is related to the development of spontaneous inflammatory bowel disease symptoms (Miyazaki, et al. 1994). Moreover, abnormal activation of JAK3 was associated with human hematology (Ihle, et al. 1997), indicating that a tight balance of its activity is essential for normal hematopoietic development.
Although JAK1, JAK2, and Tyk2 are widely expressed, JAK3 is predominantly expressed in hematopoietic cells and is associated only with the common γ chain of the IL-2, IL-4, IL-7, IL-9, and IL-15 receptors (Nosaka, et al. 1995). IL-4 is a very well-known cytokine that is crucial in the polarization of naïve T cells to type 2 helper T cells. IL-4 plays a major role in the growth and proliferation of many immune cells, such as natural killer (NK) cells and T cells (Dhupkar and Gordon 2017). Homozygous mutant mice harboring a disrupted JAK3 gene display profound reductions in thymocytes and severe B cell and T cell lymphopenia, similar to severe combined immunodeficiency disease (SCID), and functionally deficient residual T cells and B cells. Thus, JAK3 plays a critical role in γ chain signaling and lymphoid development.
Domain of Applicability
The proposed AOP involves inhibition of JAK activity, which leads to suppression of TDAR independent of life stage, sex, or age. Since JAK3 inhibitors (PF-06651600, RB1) are currently under phase 2 clinical evaluation for the treatment of rheumatoid arthritis, the AOP appears to be applicable to all life stages. JAK3 inhibitor-induced outcomes in humans are mimicked by similar responses in a variety of animal models, including non-human primates and rodents. Thus, immunosuppression induced by inhibition of JAK3 activity is considered to occur across a variety of mammalian species. For example, PF-06651600 was reported to reduce paw swelling with an unbound EC50 of 169 nM in rat adjuvant-induced arthritis. Similarly, PF-06651600 significantly reduced disease severity in an experimental autoimmune encephalomyelitis mouse model at 30 or 100 mg/kg or prophylactically at 20 and 60 mg/kg. PF-06651600 will be evaluated in clinical trials (Telliez, et al. 2016).
Essentiality of the Key Events
MIE: Inhibition of JAK3
JAK3 was initially identified (Johnston, et al. 1994, Witthuhn, et al. 1994) in studies designed to identify the JAK family member involved in the signaling of a group of cytokines with shared utilization of the γ chain first identified in the IL-2 receptor complex. It was subsequently demonstrated that JAK3 physically associates with the γ chain and is activated in a receptor complex that also contains JAK1, which associates with the ligand-specific α or β chain of the receptors (Miyazaki, et al. 1994). JAK3 is somewhat unique within the JAK family in that it is predominantly expressed in hematopoietic cells and is only activated in response to cytokines that use the γ chain (Ihle, et al. 1997). The phenotype of the JAK3 deletion mice is striking, with a range of deficiencies that collectively constitute SCID (Nosaka, et al. 1995, Thomis, et al. 1995). At the same time, two groups identified individuals that lacked JAK3 and exhibited somatically acquired SCID (Macchi, et al. 1995, Russell, et al. 1995). One of the most striking components of the phenotype are the dramatic reductions in both the T and B-cell lineages. Comparable reductions are seen in mice that lack IL-7 (von Freeden-Jeffry, et al. 1995), the IL-7 receptor α chain (Peschon, et al. 1994), or the γ chain. Despite the reduced numbers, the cells that do develop are phenotypically normal. These results are consistent with the hypothesis that activation of JAK3 is critical in the expansion, but not differentiation, of early lymphoid lineage-committed cells. In addition to the reduced numbers, the differentiated lymphoid cells that are generated fail to respond to the spectrum of cytokines that utilize the γ chain and activate JAK3 normally. In addition, there are other examples of JAK3 mutant mice. Primary immunodeficiencies (PIDs) are inborn errors that cause developmental and/or functional defects in the immune system (Picard, et al. 2015). PIDs are usually rare and monogenic. They present clinically with a broad array of phenotypes, including increased susceptibility to infection. One of the most deadly categories of PID is SCID. SCID is invariably caused by severe developmental and/or functional defects of T lymphocytes. However, SCID may also present with variable defects of B and/or NK cells. The B6.Cg-Nr1d1tm1Ven/LazJ mouse line harbors a spontaneous mutation in JAK3, which generates the SCID phenotype (Robinette, et al. 2018). STAT5 plays a major role in regulating vital cellular functions, such as proliferation, differentiation, and apoptosis of hematopoietic and immune cells (Rani and Murphy 2016, Wittig and Groner 2005). The activation of STAT5 is transient and tightly regulated in normal cells (Quezada Urban, et al. 2018). The transcription factor STAT5 is expressed in all lymphocytes and plays a key role in multiple aspects of lymphocyte development and function (Owen and Farrar 2017). STAT5 was initially identified as a transcription factor activated by prolactin in mammary gland epithelial cells (Schmitt-Ney, et al. 1992, Wakao, et al. 1992). Subsequent studies identified STAT5 binding activity in T cells (Beadling, et al. 1994). Other authors described that the expression of STAT5 in multiple cell types and its’ activation by a number of cytokines, including the common γ-chain-dependent cytokines IL-2, IL-4, IL-7, IL-13, and IL-15 (Lin, et al. 1995).
|
KE |
description |
|
KE1: Blockade of STAT5 phosphorylation
|
STAT5 refer to two proteins that share 94% structural homology and are transcribed from separate genes, STAT5A and STAT5B. Binding of these extracellular ligands to their target receptors induces the activation of receptor-associated JAK kinases that phosphorylate key tyrosine residues within the receptor, providing docking sites for the SRC homology 2 (SH2) domains of the inactive cytoplasmic STAT5 monomers. STAT5 is then phosphorylated at specific tyrosine residues, either Y694 (STAT5A) or Y699 (STAT5B) of the C-terminus. Subsequently, STAT5 undergoes a conformational change and phosphorylated STAT5 monomers form either homo- or hetero- STAT5X-STATX dimers through reciprocal phosphotyrosine–SH2 interactions (Cumaraswamy, et al. 2014, Tothova, et al. 2021). This means that STAT5 will never be activated without this phosphorylation step. |
|
KE2: Suppression of STAT5 binding to cytokine gene promoters
|
Activated STAT5 dimers translocate to the nucleus, where they bind to STAT5 DNA response elements inducing transcription of genes involved in proliferation, cell differentiation, inflammation (cytokines) and cell survival. Since the STAT5 monomer does not bind directly to the DNA element, inhibiting the STAT5 phosphorylation step suppresses STAT5 activity. |
|
KE3: Suppression of IL-4 production
|
The observation that STAT5 is activated by multiple cytokines in T cells suggests that it might play a critical role in the development and/or function of these cells. Disruption of the Stat5a gene or Stat5b gene reportedly resulted in relatively modest phenotypes. For example, Stat5a-/- mice displayed defects in mammary gland development and lactation, while Stat5b-/- mice displayed defects in response to growth hormone in male mice and NK cell proliferation (Imada, et al. 1998, Liu, et al. 1997). To determine whether combined deletion of Stat5a and Stat5b might result in more profound immunodeficiencies, subsequent studies deleted the first coding exons of both Stat5a and Stat5b. This intervention resulted in the production of truncated forms of STAT5a and STAT5b, which acted as functional hypomorphs. These mice had surprisingly mild defects in lymphocyte development, although T cells were grossly dysfunctional as they could no longer proliferate in response to IL-2 (Moriggl, et al. 1999, Teglund, et al. 1998). Finally, complete deletion of Stat5a and Stat5b using Cre-LoxP approaches demonstrated that STAT5a and STAT5b are absolutely required for lymphocyte development, as Stat5a/b-/- mice had profound blocks in lymphocyte development, which mimicked that observed in Il7r-/- mice (Cui, et al. 2004, Yao, et al. 2006). These studies definitively demonstrated the retention of appreciable STAT5 function in STAT5 hypomorph mice. Thus, T cell damage due to STAT5 deficiency or inactivation leads to suppression of the production of cytokines such as IL-4. |
AOP: Impairment of T cell dependent antibody response (Immune dysfunction)
Evidence Assessment
T cell development is mainly regulated by the JAK-STAT system. JAK3 deficiency in T cells induces multiple types of immunosuppression, including TDAR.
JAK3-deficient mice reportedly displayed profound reductions in thymocytes and severe B cell and T cell lymphopenia, similar to SCID disease. The residual T cells and B cells were functionally deficient (Peschon, et al. 1994).
Mice lacking JAK3 also showed a severe block in B cell development at the pre-B stage in the bone marrow. In contrast, although the thymuses of these mice were small, T cell maturation progressed relatively normally. In response to mitogenic signals, peripheral T cells in JAK3-deficient mice did not proliferate and secreted small amounts of IL-4. These data demonstrate that JAK3 is critical for the progression of B cell development in the bone marrow and for the functional competence of mature T cells (Nosaka, et al. 1995).
Furthermore, the abnormal architecture of lymphoid organs suggested the involvement of JAK3 in epithelial cells. T cells that developed in the mutant mice did not respond to IL-2, IL-4, or IL-7 (Ito, et al. 2017).
PF-06651600 and RB1 specifically inhibit JAK3 with over 100-fold preference over JAK2, JAK1, and TYK2 in kinase assays. Reduced inflammation and associated pathology have been described in collagen-induced arthritis mice. Importantly, the administration of PF-06651600 or RB1 results in decreased pro-inflammatory cytokines and JAK3 and STAT phosphorylation in mice. The findings suggest that the inhibition of JAK3/STAT signaling is closely correlated with the induction of multiple types of immunosuppression, including TDAR.
Known Modulating Factors
Quantitative Understanding
KER1 (MIE => KE1):
Treatment with the highly selective JAK3 inhibitor PF-06651600 or RB1 suppresses the complex formation of STAT5 in the nucleus. IL-2 stimulates STAT5 and induces tyrosine phosphorylation of STAT5 (Wakao, et al. 1995). RB1 inhibits the phosphorylation of STAT5 elicited by IL-2, as evidenced by an IC50 value of 31 nM in the peripheral blood mononuclear cells (PBMCs) of humans. PBMCs isolated from the buffy coats of healthy volunteers by density gradient centrifugation on Lymphoprep were cultured in complete RPMI 1640 medium (containing 10% fetal bovine serum, 100 μg/mL streptomycin and 100 U/mL penicillin) plus 10 μg/mL lectin phytohemagglutinin (PHA) for 3 days. The cells were then treated with recombinant human IL-6 (400 ng/mL), recombinant human IL-2 (100 ng/mL), or recombinant human granulocyte-macrophage colony-stimulating factor (GM-CSF; 50 ng/mL) at 37°C for 20 min. To terminate the stimulation, the cells were fixed with Lyse/Fix Buffer and then incubated with 100% methanol for 30 min. The cells were incubated with anti-pSTAT3 and anti-CD4 antibodies, or anti-pSTAT5 and anti-CD4 antibodies at 4°C overnight, washed twice with PBS, and analyzed with by flow cytometry (Ju, et al. 2011).
The fluorescence intensity of phospho-STAT5 in CD3-positive lymphocytes was observed to increase upon incubation of peripheral blood with IL-2. Peficitinib inhibited STAT5 phosphorylation in a concentration-dependent manner with a mean IC50 of 124 nM (101 and 147 nM for two rats). Additionally, the effect of peficitinib on IL-2 stimulated STAT5 phosphorylation in human peripheral T cells was evaluated. Parallel with the results in rats, the fluorescence intensity of phospho-STAT5 in CD3-positive lymphocytes increased in human peripheral blood after adding IL-2. Peficitinib inhibited STAT5 phosphorylation in a concentration-dependent manner with a mean IC50 of 127 nM in human lymphocytes (Ito, et al. 2017).
KER2 (KE1 => KE2):
STAT5 can be activated and phosphorylated by cytokines, such as IL-2 and IL-15. Tyrosine phosphorylation of STAT5 is important for the dimerization of STAT5 (Wakao, et al. 1995). The STAT5 dimer has an identical DNA binding specificity and immunoreactivity.
KER3 (KE2 => KE3):
STAT5 is phosphorylated by JAK kinases, allowing its dimerization and translocation into the nucleus where it can bind to its specific DNA binding sites. Electrophoretic mobility shift assay (EMSA) data revealed that IL-2 activation induced STAT5 dimerization and DNA binding to the gamma interferon activated site (GAS) motif in the IL-4 receptor alpha promoter region (John, et al. 1999). Other EMSA data showed that dexamethasone (10-6 M) inhibited STAT5 DNA binding in mononuclear cells in a dose-dependent fashion at dexamethasone concentrations of 10-8 to 10-7 M (Bianchi, et al. 2000). Dexamethasone could inhibit tyrosine phosphorylation, and nuclear translocation of STAT5 in primary T cells. The mechanism of inhibition involved suppression of IL-2 receptor and JAK3 expression.
KER4 (KE3 => AO):
Binding of IL-4 to the T cell receptor induces proliferation and differentiation into Th2 cells. Th2 cells assist B cells and promote class switching from IgM to IgG1 and IgE. Therefore, the suppression of IL-4 production leads to impairment of TDAR.
In co-cultured human T and B cells stimulated with anti-CD3 monoclonal antibody, the calcineurin inhibitors (CNIs) FK506 and cyclosporin A (CsA) lowered the levels of T cell cytokines, including IL-2 and IL-4, and inhibited IgM and IgG production in a dose-dependent manner (Heidt, et al. 2010).
The collective results demonstrate the quantitative relationships between the inhibition of IL-4 by specific antibodies or CNI and suppression of antibody production.
|
1. Support for Biological Plausibility of KER |
|
|
MIE => KE1: Inhibition of JAK3 to blockade of STAT5 phosphorylation |
Biological Plausibility of the MIE => KE1 is Strong. Rationale: Administration of PF-06651600 or RB1 results in decreased pro-inflammatory cytokines and JAK3 and STAT phosphorylation in mice. |
|
KE1 => KE2: Blockade of STAT5 phosphorylation to suppression of STAT5 binding to cytokine gene promoters |
Biological Plausibility of the KE1 => KE2 is Strong. Rationale: STAT5 plays a major role in regulating vital cellular functions, such as proliferation, differentiation, and apoptosis of hematopoietic and immune cells. STAT5 is activated by phosphorylation of a single constituent tyrosine residue (Y694) and is negatively regulated by dephosphorylation. A wide variety of growth factors and cytokines can activate STAT5 through the JAK-STAT pathway. The activation of STAT5 is transient and tightly regulated in normal cells. |
|
KE2 => KE3: Suppression of STAT5 binding to cytokine gene promoters to impairment of T cell dependent antibody response (Immune dysfunction) |
Biological Plausibility of the KE2 => KE3 is strong. Rationale: In response to mitogenic signals, peripheral T cells in JAK3-deficient mice did not proliferate and secreted small amounts of IL-4. These data demonstrate that JAK3 is critical for the progression of B cell development in the bone marrow and for the functional competence of mature T cells. |
|
KE3 => AO: Suppression, IL-4 production to impairment, T cell dependent Antibody response |
Biological Plausibility of the KE3 => KE4 is strong. Rationale: In T cells, binding of IL-4 to its receptor induces proliferation and differentiation into Th2 cells. Th2 cells provide help for B cells and promote class switching from IgM to IgG1 and IgE. Therefore, the suppression of IL-4 production leads to the impairment of TDAR. |
|
2. Support for Essentiality of AOP |
Rationale for Essentiality of KEs in the AOP is strong: |
|
3. Empirical Support for KERs |
|
|
MIE => KE1: Inhibition of JAK3 to blockade of STAT5 phosphorylation |
Empirical Support of the MIE => KE1 is strong. Rationale: Treatment with the highly selective JAK3 inhibitor PF-06651600 or RB1 suppresses the complex formation of STAT5 in the nucleus. IL-2 stimulates STAT5 and induces tyrosine phosphorylation of STAT5. RB1 inhibits the phosphorylation of STAT5 elicited by IL-2, as evidenced by an IC50 value of 31 nM in the peripheral blood mononuclear cells (PBMCs) of humans. PBMCs isolated from the buffy coats of healthy volunteers by density gradient centrifugation on Lymphoprep were cultured in complete RPMI 1640 medium (containing 10% fetal bovine serum, 100 μg/mL streptomycin and 100 U/mL penicillin) plus 10 μg/mL lectin phytohemagglutinin (PHA) for 3 days. The cells were then treated with recombinant human IL-6 (400 ng/mL), recombinant human IL-2 (100 ng/mL), or recombinant human granulocyte-macrophage colony-stimulating factor (GM-CSF; 50 ng/mL) at 37°C for 20 min. To terminate the stimulation, the cells were fixed with Lyse/Fix Buffer and then incubated with 100% methanol for 30 min. The cells were incubated with anti-pSTAT3 and anti-CD4 antibodies, or anti-pSTAT5 and anti-CD4 antibodies at 4°C overnight, washed twice with PBS, and analyzed with by flow cytometry. The fluorescence intensity of phospho-STAT5 in CD3-positive lymphocytes was observed to increase upon incubation of peripheral blood with IL-2. Peficitinib inhibited STAT5 phosphorylation in a concentration-dependent manner with a mean IC50 of 124 nM (101 and 147 nM for two rats). Additionally, the effect of peficitinib on IL-2 stimulated STAT5 phosphorylation in human peripheral T cells was evaluated. Parallel with the results in rats, the fluorescence intensity of phospho-STAT5 in CD3-positive lymphocytes increased in human peripheral blood after adding IL-2. Peficitinib inhibited STAT5 phosphorylation in a concentration-dependent manner with a mean IC50 of 127 nM in human lymphocytes. |
|
KE1 => KE2: Blockade of STAT5 phosphorylation to suppression of STAT5 binding to cytokine gene promoters |
Empirical Support of the KE1 => KE2 is strong. Rationale: STAT5 can be activated and phosphorylated by cytokines, such as IL-2 and IL-15. Tyrosine phosphorylation of STAT5 is important for the dimerization of STAT5. The STAT5 dimer has an identical DNA binding specificity and immunoreactivity. |
|
KE2 => KE3: Suppression of STAT5 binding to cytokine gene promoters to impairment of T cell dependent antibody response (Immune dysfunction) |
Empirical Support of the KE2 => KE3 is strong. Rationale: STAT5 is phosphorylated by JAK kinases, allowing its dimerization and translocation into the nucleus where it can bind to its specific DNA binding sites. Electrophoretic mobility shift assay (EMSA) data revealed that IL-2 activation induced STAT5 dimerization and DNA binding to the gamma interferon activated site (GAS) motif in the IL-4 receptor alpha promoter region. Other EMSA data showed that dexamethasone (10-6 M) inhibited STAT5 DNA binding in mononuclear cells in a dose-dependent fashion at dexamethasone concentrations of 10-8 to 10-7 M. Dexamethasone could inhibit tyrosine phosphorylation, and nuclear translocation of STAT5 in primary T cells. The mechanism of inhibition involved suppression of IL-2 receptor and JAK3 expression. |
|
KE3 => AO: Suppression, IL-4 production to impairment, T cell dependent Antibody response |
Empirical Support of the KE3 => KE4 is strong. Rationale: Binding of IL-4 to the T cell receptor induces proliferation and differentiation into Th2 cells. Th2 cells assist B cells and promote class switching from IgM to IgG1 and IgE. Therefore, the suppression of IL-4 production leads to impairment of TDAR. In co-cultured human T and B cells stimulated with anti-CD3 monoclonal antibody, the calcineurin inhibitors (CNIs) FK506 and cyclosporin A (CsA) lowered the levels of T cell cytokines, including IL-2 and IL-4, and inhibited IgM and IgG production in a dose-dependent manner. The collective results demonstrate the quantitative relationships between the inhibition of IL-4 by specific antibodies or CNI and suppression of antibody production. |
Considerations for Potential Applications of the AOP (optional)
References
Beadling C, Guschin D, Witthuhn BA, Ziemiecki A, Ihle JN, Kerr IM, Cantrell DA. 1994. Activation of JAK kinases and STAT proteins by interleukin-2 and interferon alpha, but not the T cell antigen receptor, in human T lymphocytes. EMBO J 13:5605-5615.
Bianchi M, Meng C, Ivashkiv LB. 2000. Inhibition of IL-2-induced Jak-STAT signaling by glucocorticoids. Proc Natl Acad Sci U S A 97:9573-9578. DOI: 10.1073/pnas.160099797.
Cui Y, Riedlinger G, Miyoshi K, Tang W, Li C, Deng CX, Robinson GW, Hennighausen L. 2004. Inactivation of Stat5 in mouse mammary epithelium during pregnancy reveals distinct functions in cell proliferation, survival, and differentiation. Mol Cell Biol 24:8037-8047. DOI: 10.1128/MCB.24.18.8037-8047.2004.
Cumaraswamy AA, Lewis AM, Geletu M, Todic A, Diaz DB, Cheng XR, Brown CE, Laister RC, Muench D, Kerman K, Grimes HL, Minden MD, Gunning PT. 2014. Nanomolar-Potency Small Molecule Inhibitor of STAT5 Protein. ACS Med Chem Lett 5:1202-1206. DOI: 10.1021/ml500165r.
Dhupkar P, Gordon N. 2017. Interleukin-2: Old and New Approaches to Enhance Immune-Therapeutic Efficacy. Adv Exp Med Biol 995:33-51. DOI: 10.1007/978-3-319-53156-4_2.
Heidt S, Roelen DL, Eijsink C, Eikmans M, van Kooten C, Claas FH, Mulder A. 2010. Calcineurin inhibitors affect B cell antibody responses indirectly by interfering with T cell help. Clin Exp Immunol 159:199-207. DOI: 10.1111/j.1365-2249.2009.04051.x.
Ihle JN, Nosaka T, Thierfelder W, Quelle FW, Shimoda K. 1997. Jaks and Stats in cytokine signaling. Stem Cells 15 Suppl 1:105-111; discussion 112. DOI: 10.1002/stem.5530150814.
Imada K, Bloom ET, Nakajima H, Horvath-Arcidiacono JA, Udy GB, Davey HW, Leonard WJ. 1998. Stat5b is essential for natural killer cell-mediated proliferation and cytolytic activity. J Exp Med 188:2067-2074.
Ito M, Yamazaki S, Yamagami K, Kuno M, Morita Y, Okuma K, Nakamura K, Chida N, Inami M, Inoue T, Shirakami S, Higashi Y. 2017. A novel JAK inhibitor, peficitinib, demonstrates potent efficacy in a rat adjuvant-induced arthritis model. J Pharmacol Sci 133:25-33. DOI: 10.1016/j.jphs.2016.12.001.
John S, Vinkemeier U, Soldaini E, Darnell JE, Jr., Leonard WJ. 1999. The significance of tetramerization in promoter recruitment by Stat5. Mol Cell Biol 19:1910-1918.
Johnston JA, Bacon CM, Finbloom DS, Rees RC, Kaplan D, Shibuya K, Ortaldo JR, Gupta S, Chen YQ, Giri JD, et al. 1995. Tyrosine phosphorylation and activation of STAT5, STAT3, and Janus kinases by interleukins 2 and 15. Proc Natl Acad Sci U S A 92:8705-8709.
Johnston JA, Kawamura M, Kirken RA, Chen YQ, Blake TB, Shibuya K, Ortaldo JR, McVicar DW, O'Shea JJ. 1994. Phosphorylation and activation of the Jak-3 Janus kinase in response to interleukin-2. Nature 370:151-153. DOI: 10.1038/370151a0.
Ju W, Zhang M, Jiang JK, Thomas CJ, Oh U, Bryant BR, Chen J, Sato N, Tagaya Y, Morris JC, Janik JE, Jacobson S, Waldmann TA. 2011. CP-690,550, a therapeutic agent, inhibits cytokine-mediated Jak3 activation and proliferation of T cells from patients with ATL and HAM/TSP. Blood 117:1938-1946. DOI: 10.1182/blood-2010-09-305425.
Liao W, Schones DE, Oh J, Cui Y, Cui K, Roh TY, Zhao K, Leonard WJ. 2008. Priming for T helper type 2 differentiation by interleukin 2-mediated induction of interleukin 4 receptor alpha-chain expression. Nat Immunol 9:1288-1296. DOI: 10.1038/ni.1656.
Lin JX, Migone TS, Tsang M, Friedmann M, Weatherbee JA, Zhou L, Yamauchi A, Bloom ET, Mietz J, John S, et al. 1995. The role of shared receptor motifs and common Stat proteins in the generation of cytokine pleiotropy and redundancy by IL-2, IL-4, IL-7, IL-13, and IL-15. Immunity 2:331-339.
Liu X, Robinson GW, Wagner KU, Garrett L, Wynshaw-Boris A, Hennighausen L. 1997. Stat5a is mandatory for adult mammary gland development and lactogenesis. Genes Dev 11:179-186.
Macchi P, Villa A, Giliani S, Sacco MG, Frattini A, Porta F, Ugazio AG, Johnston JA, Candotti F, O'Shea JJ, et al. 1995. Mutations of Jak-3 gene in patients with autosomal severe combined immune deficiency (SCID). Nature 377:65-68. DOI: 10.1038/377065a0.
Miyazaki T, Kawahara A, Fujii H, Nakagawa Y, Minami Y, Liu ZJ, Oishi I, Silvennoinen O, Witthuhn BA, Ihle JN, et al. 1994. Functional activation of Jak1 and Jak3 by selective association with IL-2 receptor subunits. Science 266:1045-1047.
Moriggl R, Topham DJ, Teglund S, Sexl V, McKay C, Wang D, Hoffmeyer A, van Deursen J, Sangster MY, Bunting KD, Grosveld GC, Ihle JN. 1999. Stat5 is required for IL-2-induced cell cycle progression of peripheral T cells. Immunity 10:249-259.
Nosaka T, van Deursen JM, Tripp RA, Thierfelder WE, Witthuhn BA, McMickle AP, Doherty PC, Grosveld GC, Ihle JN. 1995. Defective lymphoid development in mice lacking Jak3. Science 270:800-802.
Owen DL, Farrar MA. 2017. STAT5 and CD4 (+) T Cell Immunity. F1000Res 6:32. DOI: 10.12688/f1000research.9838.1.
Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC, Park LS, Ziegler SF, Williams DE, Ware CB, Meyer JD, Davison BL. 1994. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med 180:1955-1960.
Picard C, Al-Herz W, Bousfiha A, Casanova JL, Chatila T, Conley ME, Cunningham-Rundles C, Etzioni A, Holland SM, Klein C, Nonoyama S, Ochs HD, Oksenhendler E, Puck JM, Sullivan KE, Tang ML, Franco JL, Gaspar HB. 2015. Primary Immunodeficiency Diseases: an Update on the Classification from the International Union of Immunological Societies Expert Committee for Primary Immunodeficiency 2015. J Clin Immunol 35:696-726. DOI: 10.1007/s10875-015-0201-1.
Quezada Urban R, Diaz Velasquez CE, Gitler R, Rojo Castillo MP, Sirota Toporek M, Figueroa Morales A, Moreno Garcia O, Garcia Esquivel L, Torres Mejia G, Dean M, Delgado Enciso I, Ochoa Diaz Lopez H, Rodriguez Leon F, Jan V, Garzon Barrientos VH, Ruiz Flores P, Espino Silva PK, Haro Santa Cruz J, Martinez Gregorio H, Rojas Jimenez EA, Romero Cruz LE, Mendez Catala CF, Alvarez Gomez RM, Fragoso Ontiveros V, Herrera LA, Romieu I, Terrazas LI, Chirino YI, Frecha C, Oliver J, Perdomo S, Vaca Paniagua F. 2018. Comprehensive Analysis of Germline Variants in Mexican Patients with Hereditary Breast and Ovarian Cancer Susceptibility. Cancers (Basel) 10. DOI: 10.3390/cancers10100361.
Rani A, Murphy JJ. 2016. STAT5 in Cancer and Immunity. J Interferon Cytokine Res 36:226-237. DOI: 10.1089/jir.2015.0054.
Robinette ML, Cella M, Telliez JB, Ulland TK, Barrow AD, Capuder K, Gilfillan S, Lin LL, Notarangelo LD, Colonna M. 2018. Jak3 deficiency blocks innate lymphoid cell development. Mucosal Immunol 11:50-60. DOI: 10.1038/mi.2017.38.
Russell SM, Tayebi N, Nakajima H, Riedy MC, Roberts JL, Aman MJ, Migone TS, Noguchi M, Markert ML, Buckley RH, O'Shea JJ, Leonard WJ. 1995. Mutation of Jak3 in a patient with SCID: essential role of Jak3 in lymphoid development. Science 270:797-800.
Schmitt-Ney M, Happ B, Hofer P, Hynes NE, Groner B. 1992. Mammary gland-specific nuclear factor activity is positively regulated by lactogenic hormones and negatively by milk stasis. Mol Endocrinol 6:1988-1997. DOI: 10.1210/mend.6.12.1491685.
Teglund S, McKay C, Schuetz E, van Deursen JM, Stravopodis D, Wang D, Brown M, Bodner S, Grosveld G, Ihle JN. 1998. Stat5a and Stat5b proteins have essential and nonessential, or redundant, roles in cytokine responses. Cell 93:841-850.
Telliez JB, Dowty ME, Wang L, Jussif J, Lin T, Li L, Moy E, Balbo P, Li W, Zhao Y, Crouse K, Dickinson C, Symanowicz P, Hegen M, Banker ME, Vincent F, Unwalla R, Liang S, Gilbert AM, Brown MF, Hayward M, Montgomery J, Yang X, Bauman J, Trujillo JI, Casimiro-Garcia A, Vajdos FF, Leung L, Geoghegan KF, Quazi A, Xuan D, Jones L, Hett E, Wright K, Clark JD, Thorarensen A. 2016. Discovery of a JAK3-Selective Inhibitor: Functional Differentiation of JAK3-Selective Inhibition over pan-JAK or JAK1-Selective Inhibition. ACS Chem Biol 11:3442-3451. DOI: 10.1021/acschembio.6b00677.
Thomis DC, Gurniak CB, Tivol E, Sharpe AH, Berg LJ. 1995. Defects in B lymphocyte maturation and T lymphocyte activation in mice lacking Jak3. Science 270:794-797.
Tothova Z, Tomc J, Debeljak N, Solar P. 2021. STAT5 as a Key Protein of Erythropoietin Signalization. Int J Mol Sci 22. DOI: 7109 [pii]10.3390/ijms22137109ijms22137109 [pii].
von Freeden-Jeffry U, Vieira P, Lucian LA, McNeil T, Burdach SE, Murray R. 1995. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J Exp Med 181:1519-1526.
Wakao H, Harada N, Kitamura T, Mui AL, Miyajima A. 1995. Interleukin 2 and erythropoietin activate STAT5/MGF via distinct pathways. EMBO J 14:2527-2535.
Wakao H, Schmitt-Ney M, Groner B. 1992. Mammary gland-specific nuclear factor is present in lactating rodent and bovine mammary tissue and composed of a single polypeptide of 89 kDa. J Biol Chem 267:16365-16370.
Willerford DM, Chen J, Ferry JA, Davidson L, Ma A, Alt FW. 1995. Interleukin-2 receptor alpha chain regulates the size and content of the peripheral lymphoid compartment. Immunity 3:521-530.
Witthuhn BA, Silvennoinen O, Miura O, Lai KS, Cwik C, Liu ET, Ihle JN. 1994. Involvement of the Jak-3 Janus kinase in signalling by interleukins 2 and 4 in lymphoid and myeloid cells. Nature 370:153-157. DOI: 10.1038/370153a0.
Wittig I, Groner B. 2005. Signal transducer and activator of transcription 5 (STAT5), a crucial regulator of immune and cancer cells. Curr Drug Targets Immune Endocr Metabol Disord 5:449-463.
Yao Z, Cui Y, Watford WT, Bream JH, Yamaoka K, Hissong BD, Li D, Durum SK, Jiang Q, Bhandoola A, Hennighausen L, O'Shea JJ. 2006. Stat5a/b are essential for normal lymphoid development and differentiation. Proc Natl Acad Sci U S A 103:1000-1005. DOI: 10.1073/pnas.0507350103.
Zhu J, Cote-Sierra J, Guo L, Paul WE. 2003. Stat5 activation plays a critical role in Th2 differentiation. Immunity 19:739-748. DOI: 10.1016/s1074-7613(03)00292-9.
Zhu J, Min B, Hu-Li J, Watson CJ, Grinberg A, Wang Q, Killeen N, Urban JF, Jr., Guo L, Paul WE. 2004. Conditional deletion of Gata3 shows its essential function in T(H)1-T(H)2 responses. Nat Immunol 5:1157-1165. DOI: 10.1038/ni1128.