
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 376
Title
Androgen receptor agonism leading to male-biased sex ratio
Short name
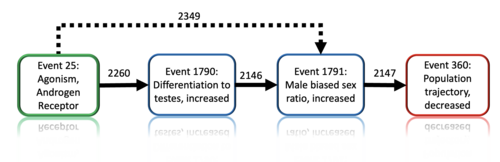
Graphical Representation
Point of Contact
Contributors
- Dan Villeneuve
- Kelvin Santana Rodriguez
- Gerald Ankley
- Evgeniia Kazymova
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.12 | WPHA/WNT Endorsed |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Agonism, Androgen receptor | March 20, 2017 17:44 |
| Increased, Differentiation to Testis | December 28, 2022 10:15 |
| Increased, Male Biased Sex Ratio | January 03, 2023 08:22 |
| Decrease, Population growth rate | January 03, 2023 09:09 |
| Agonism, Androgen receptor leads to Increased, Differentiation to Testis | January 03, 2023 08:30 |
| Agonism, Androgen receptor leads to Increased, Male Biased Sex Ratio | January 03, 2023 08:47 |
| Increased, Differentiation to Testis leads to Increased, Male Biased Sex Ratio | January 03, 2023 08:35 |
| Increased, Male Biased Sex Ratio leads to Decrease, Population growth rate | January 03, 2023 08:37 |
| 17beta-Trenbolone | November 29, 2016 18:42 |
| Chemical:33664 17-Methyltestosterone | March 23, 2021 13:34 |
| 5alpha-Dihydrotestosterone | March 14, 2017 12:44 |
| Methyldihydrotestosterone | May 07, 2021 15:36 |
| 11-Keto-testosterone | May 07, 2021 15:36 |
Abstract
This adverse outcome pathway links androgen receptor agonism in teleost fish during gonadogenesis to male-biased sexual differentiation and consequently, reduced population growth rate. Sex determination in teleost fishes is highly plastic; it can be genetically or environmentally influenced. Species with environmentally-based sex determination in particular can be very sensitive to exogenous chemicals during the period of differentiation. Exogenous hormones are of ecological concern because they have the potential to alter gonad development and sex differentiation. Activation of the androgen receptor (AR) by endogenous androgens plays a crucial role in normal sex differentiation, sexual maturation, and spermatogenesis in vertebrates and inappropriate signaling by exogenous AR agonists can disrupt theses processes. For example, studies have shown that during early development in some teleost species, exposure to androgenic steroids can induce complete gonadal sex inversion, resuting in male-biased sex ratios. This can lead to impacts on population growth rates due to the decreased number of reproductively viable females in the population.
AOP Development Strategy
Context
This AOP shares multiple KEs and KERs with AOP 346 which links aromatase inhibition to male-biased sex ratios in vertebrates with environmental sex determination.
Strategy
This AOP was initiated by Dr. Daniel Villeneuve, a leader in the field of AOP development. Dr. Gerald Ankley also contributed to AOP development, particularly at later stages of the effort. Dr. Ankley is experienced with AOP development, and is a widely-recognized international expert concerning the effects of EDCs on fish. The AOP is based on evaluation of published peer-reviewed literature derived from focused searches guided by the expertise of the authors. Mr. Kelvin Santana-Rodriguez contributed to literature searches for the AOP, and Ms. Kathleen Jensen, who also has worked for many years on EDCs/fish endocrinology, provided a secondary QA review of key papers supporting the AOP.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 25 | Agonism, Androgen receptor | Agonism, Androgen receptor |
| KE | 1790 | Increased, Differentiation to Testis | Increased, Differentiation to Testis |
| KE | 1791 | Increased, Male Biased Sex Ratio | Increased, Male Biased Sex Ratio |
| AO | 360 | Decrease, Population growth rate | Decrease, Population growth rate |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Development | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP
See details below.
Domain of Applicability
Life Stage
The life stage applicable to this AOP is developing embryos and juveniles prior to- or during the gonadal developmental stage. This AOP is not applicable to sexually differentiated adults.
Sex
The molecular initiating event for this AOP occurs prior to gonad differentiation. Therefore, this AOP is only applicable to sexually undifferentiated individuals.
Taxonomic
Most evidence for this AOP is derived from fish in the class Osteichthyes. Both phylogenetic analysis and evaluation of protein sequence conservation via SeqAPASS (https://seqapass.epa.gov/seqapass/) has shown that the structure of the AR is well conserved among most jawed vertebrates (Gnathostomata). This AOP is not expected to apply to mammals, birds, or other jawed vertebrates with genetic sex determination. However, it may be applicable to fishes, amphibians, and reptiles with environmentally-dependent sex determination, although outcomes may differ across physiologically different taxa. The present AOP is not considered relevant to Agnathans since the AR appears not to be present in jawless fishes (Thornton 2001; Hossain et al 2008).
Essentiality of the Key Events
Support for the essentiality of several of the key events in the AOP is provided by both in vivo and in vitro studies with chemicals acting as AR agonists and antagonists. Important in vivo studies typically have been conducted during the critical period of sexual differentiation.
Golan & Levavi-Sivian (2014) exposed genetic females of Nile tilapia (Oreochromis niloticus) to 17α-methyltestosterone (MT) and dihydrotestosterone (DHT), and showed that the two AR agonists induced a male biased sex ratio in a dose-dependent manner. However, in combined exposures with the AR antagonist flutamide (a pharmaceutical), the sex inversion potency of MT and DHT was reduced in a dose-dependent manner. This provides direct evidence that activation of the AR is required for the subsequent key events to occur.
Crowder et al. (2018) generated zebrafish (Danio rerio) with a mutation in the AR gene (aruab105/105). Most mutants developed ovaries and displayed female secondary sexual characteristics. The small percentage of mutants that developed as males displayed female secondary sexual characteristics with structurally disorganized testes, and were unable to produce normal levels of sperm. This demonstrates that the AR is required for proper testis development and fertility and supports the essentiality of AR activation in testis differentiation.
In a similar study with zebrafish, Yu et al. (2018) generated an AR gene mutant line using CRISPR/Cas9 technology. The number of female offspring was increased and the resulting AR-null males had female secondary sex characteristics and were infertile due to defective spermatogenesis. This study again supports the essentiality of AR agonism for the development of testis and how inappropriate (increased) signaling in the pathway could result in a male biased sex ratio.
|
Key Event |
Evidence |
Essentiality/Assessment |
|
|
Agonism, Androgen |
moderate |
There is good evidence from a sex inversion treatment via the direct blocking of AR using androgen antagonist that support the specificity of androgen receptor agonism for the subsequent key events to occur. |
|
|
Differentiation to Testis |
moderate |
Biological plausibility provides strong support for the essentiality of this event for the subsequent key events to occur. By definition, males have testis. |
|
|
Male Biased Sex Ratio |
moderate |
Breeding females (and both sexes) are necessary for population sustainability. A male biased sex population suggests a reduced offspring production, due to reduced numbers of females in the population, and consequentially reduced population growth rates. |
|
|
Population Sustainability |
weak |
|
Evidence Assessment
Biological Plausibility
The biological plausibility linking AR activation to increased differentiation to testis is very strong. Actions of androgens are mediated by the AR which is part of the nuclear receptor superfamily. ARs are ligand-dependent transcription factors (Hossain et al., 2008). Steroidal androgens act by entering the cell and forming a complex with the AR, resulting in conformational change (Bohen et al., 1995; Pratt and Toft, 1997). The ligand-AR complex is translocated to the nucleus where it binds to specific short DNA sequences (Androgen Responsive Elements), thereby activating transcripton of androgen regulated genes (Harbott et al., 2009). During sexual development, endogenous androgen can therefore induce the upregulation of many genes involved in the male developmental pathway.
The direct link between increased differentiation to testis leading to a male biased sex ratio is highly plausible. If the conditions that favored a male producing phenotype (in this case, exposure to AR agonsts) overlap with the critical period of sex differentiation in a given population, it is reasonable that more phenotypic males will be produced (Orn et al., 2003; Seki et al., 2004; Bogers et al., 2006; Morthorst et al., 2010; Baumann et al., 2014; Golan & Levavi-Sivian 2014). Therefore, androgen exposure for repeated or prolonged periods of time conceptually will result in a male-biased population. Empirical evidence supporting the direct link between male-biased sex ratio and reduced population growth rate in fish species is lacking. However, altered sex ratios have the potential to affect fish populations (Marty et al. 2017). For example, a male-biased sex ratio suggests that the number of breeding females would be reduced. If the male-biased sex ratio persists and/or increases over time, the offspring produced per reproductive cycle would decrease, eventually leading to a diminished population size, assuming other demongraphic parameters remained largely constant (Brown et al. 2015; Grayson et al. 2014; Miller et al. 2022).
Concordance of Dose Response Relationships
The concentration-dependence of the key event responses with regard to the concentration of exogenous AR agonists has been established in vivo for some key events in the AOP. In general, effects on downstream key events occurred at concentrations equal to or greater than those at which upstream events occurred. However, binding to the androgen receptor (the MIE) was not directly measured in any of the in vivo studies. Nonetheless, AR binding of several of the agonists tested in vivo has been documented with in vitro studies with fish ARs (e.g., Wilson et al. 2004). In fish, phenotypic masculinization of female secondary sex characteristics has been used as an indirect measurement of in vivo AR agonism. However, in the of case this AOP specifically, AR agonism is occurring prior to sexual differentiation and the resultant “phenotypic measurement” for the in vivo study (gonad differentiation) is a discrete downstream key event. Consequently, in vitro evidence can reliably be used to identify specific chemicals as AR agonists (i.e., able to activate the MIE). That is, dependence of the severity of the downstream in vivo responses on concentration and potency of chemicals activating the AR in vitro can be used as indirect evidence of dose-response concordance between the MIE and downstream key events.
- Concentration dependent androgen receptor agonism (in vitro)
- COS Whole Cell Binding Assay with fathead minnow AR (fhAR) were used in competitive binding experiments testing several natural and synthetic steroids, some of which are environmental contaminants, such as R1881, 17beta-trenbolone , and 17alpha-trenbolone. All showed a concentration dependent displacement of [3H]R1881 binding proving to be high affinity ligands for the fmAR. (Wilson et al., 2004).
- The synthetic steroids, R1881 and methytestosterone, had the highest affinities of all the chemicals tested with IC50 values of about 1.6 nM, followed by the synthetic steroids 17α- and 17β-trenbolone with IC50 values of about 8 and 16 nM, respectively.
- Of the natural steroids, dihydrotestosterone was the strongest competitor with an IC50 of about 20 nM. The IC50 for the fish specific androgen, 11- ketotestosterone, was approximately 40 nM, followed by both testosterone and androstenedione at about 100 nM
- Important to note that all of the above steroids tested were used in the in vivo studies that were selected to support this AOP demonstrating that all bound to the fhAR with a higher affinity than 11- ketotestosterone.
- Concentration dependent increased differentiation to testes:
- Studies with zebrafish and Japanese medaka (Oryzias latipes) exposed to 17β-trenbolone during development resulted in masculinization in a concentration-dependent manner as evidenced from a significantly increased maturity of testes (Orn et al., 2006; Morthorst et al., 2010; Baumann et al., 2014) for some studies, this was determined ether by the abundance of spermatozoa and/or by the area of the testis.
- Concentration dependent increased male biased sex ratio:
- Exposure of developing zebrafish to different concentrations of 17β-trenbolone and dihydrotestosterone led to an increased number of males in a dose-dependent fashion (Orn et al., 2003; Holbech et al.,2006; Morthorst et al., 2010; Baumann et al., 2013; Baumann et al., 2014)
- Concentration dependent decline in population trajectory:
- Modeled population trajectories show a concentration-dependent reduction in projected population size (Brown et al 2015, Miller et al. 2022) based on the ratio of male to female. Population-level effects exposure of fish to AR agonists have not been measured directly.
Temporal concordance
Because this AOP involves actions during a specific development transition from an undifferentiated to differentiated gonad, the temporal concordance of the events is implicit. A male biased sex ratio cannot be observed until the population has undergone sexual differentiation. Likewise, reproduction and associated population growth rate cannot be assessed until the animals achieve sexual maturity.
Consistency
We are aware of no cases where substantial exposure of susceptible teleost fish species during sexual differentiation to known AR agonists did not impact male sex ratios (for reviews see Pandian and Sheela 1995 and Leet et al. 2011). There are cases however, in which exposure to aromatizable androgens such as methyltestosterone can lead both to masculinization and feminization of fish (e.g., Piferrer et al. 1993; Orn et al., 2003); this most likely is due to conversion of the androgen to its corresponding estrogen analogue (i.e., methylestradiol; e.g., Hornung et al. 2004 ). In other instances, non-aromatizable androgens (e.g., dihydrotestosterone) have been reported to feminize fish exposed during early development (e.g., Davis et al. 1992; Bogers et al. 2006). The mechanism underlying this is uncertain, but plausibly could involve binding to the estrogen receptor which is known to interact with a variety of steroids, including androgens at comparatively test concentrations.
The adverse outcome is not wholly specific to this AOP. Key events included overlap with AOPs linking other MIEs (e.g., aromatase inhibition, AOP 346) that can lead to male biased sex ratios.
Uncertainties, inconsistencies, and data gaps
Overall, there is strong empirical support for the proposed AOP. We did not find notable inconsistencies in the literature reviewed as part of this AOP development. However, there were several notable data gaps which could potentially be addressed through further research:
(1) The detailed gene expression and signaling mechanisms via with AR activation induces differentiation to testes is not well understood or defined. If key steps in this process could be defined, one or more additional key events could potentially be inserted between Event 25 (agonism, androgen receptor) and Event 1790 (differentiation to testes, increased).
(2) Population-level consequences of a male biased sex ratio are based on modeling. At present, we found no empirical studies that establish the effect of a male-biased sex ratio on population growth rates. Current models assume that other demographic variables such as predation rates, prey availability, habitat availability, etc. are unaffected by sex ratio. This may or may not be the case in real-world populations.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
At this time available data are insufficient to develop a quantitative AOP linking AR activation to male biased fish populations.
Considerations for Potential Applications of the AOP (optional)
Sex ratios can be a useful endpoint in risk and hazard assessment of chemicals. In July 2011, the Fish Sexual Development Test (FSDT) has officially been adopted as OECD test guideline no. 234 for the detection of EDCs within the OECD conceptual framework at level 4 (OECD, 2011b). The Fish Sexual Development Test (FSDT; OECD TG 234, OECD, 2011) covers endocrine disruption during the developmental period of sexual differentiation of particularly zebrafish and uses gonadal differentiation and sex ratio as endocrine disruption-associated endpoints for indicating EAS (estrogen, androgen and steroidogenesis) activity (Dang & Kienzler 2019). Therefore, this AOP can provide additional support to the use of alternative measurements in this type of tests by screening for androgen receptor agonists.
References
Baumann, L., Holbech, H., Keiter, S., Kinnberg, K. L., Knörr, S., Nagel, T., & Braunbeck, T. (2013). The maturity index as a tool to facilitate the interpretation of changes in vitellogenin production and sex ratio in the Fish Sexual Development Test. Aquatic toxicology (Amsterdam, Netherlands), 128-129, 34–42.
Baumann, L., Knörr, S., Keiter, S., Nagel, T., Rehberger, K., Volz, S., Oberrauch, S., Schiller, V., Fenske, M., Holbech, H., Segner, H., & Braunbeck, T. (2014). Persistence of endocrine disruption in zebrafish (Danio rerio) after discontinued exposure to the androgen 17β-trenbolone. Environmental toxicology and chemistry, 33(11), 2488–2496. https://doi.org/10.1002/etc.2698
Bohen, S. P., Kralli, A., & Yamamoto, K. R. (1995). Hold 'em and fold 'em: chaperones and signal transduction. Science (New York, N.Y.), 268(5215), 1303–1304. https://doi.org/10.1126/science.7761850
Bogers, R., De Vries-Buitenweg, S., Van Gils, M., Baltussen, E., Hargreaves, A., van de Waart, B., De Roode, D., Legler, J., & Murk, A. (2006). Development of chronic tests for endocrine active chemicals. Part 2: an extended fish early-life stage test with an androgenic chemical in the fathead minnow (Pimephales promelas). Aquatic toxicology (Amsterdam, Netherlands), 80(2), 119–130. https://doi.org/10.1016/j.aquatox.2006.07.020
Brown, A. R., Owen, S. F., Peters, J., Zhang, Y., Soffker, M., Paull, G. C., Hosken, D. J., Wahab, M. A., & Tyler, C. R. (2015). Climate change and pollution speed declines in zebrafish populations. Proceedings of the National Academy of Sciences of the United States of America, 112(11), E1237–E1246. https://doi.org/10.1073/pnas.1416269112
Crowder, C. M., Lassiter, C. S., & Gorelick, D. A. (2018). Nuclear Androgen Receptor Regulates Testes Organization and Oocyte Maturation in Zebrafish. Endocrinology, 159(2), 980–993. https://doi.org/10.1210/en.2017-00617
Davis, K. B., Goudie, C. A., Simco, B. A., Tiersch, T. R., & Carmichael, G. J. (1992). Influence of dihydrotestosterone on sex determination in channel catfish and blue catfish: period of developmental sensitivity. General and comparative endocrinology, 86(1), 147–151. https://doi.org/10.1016/0016-6480(92)90136-8
Galvez, J., Mazik, P., Phelps, R., Mulvaney, D. (1995) Masculinization of Channel Catfish Ictalurus punctatus by Oral Administration of Trenbolone Acetate. World Aquaculture Society, 26(4), 378-383. https://doi.org/10.1111/j.1749-7345.1995.tb00832.x
Golan, M., & Levavi-Sivan, B. (2014). Artificial masculinization in tilapia involves androgen receptor activation. General and comparative endocrinology, 207, 50–55. https://doi.org/10.1016/j.ygcen.2014.04.026
Grayson, K. L., Mitchell, N. J., Monks, J. M., Keall, S. N., Wilson, J. N., & Nelson, N. J. (2014). Sex ratio bias and extinction risk in an isolated population of Tuatara (Sphenodon punctatus). PloS one, 9(4), e94214. https://doi.org/10.1371/journal.pone.0094214
Harbott, L. K., Burmeister, S. S., White, R. B., Vagell, M., & Fernald, R. D. (2007). Androgen receptors in a cichlid fish, Astatotilapia burtoni: structure, localization, and expression levels. The Journal of comparative neurology, 504(1), 57–73. https://doi.org/10.1002/cne.21435
Hornung, M. W., Jensen, K. M., Korte, J. J., Kahl, M. D., Durhan, E. J., Denny, J. S., Henry, T. R., & Ankley, G. T. (2004). Mechanistic basis for estrogenic effects in fathead minnow (Pimephales promelas) following exposure to the androgen 17alpha-methyltestosterone: conversion of 17alpha-methyltestosterone to 17alpha-methylestradiol. Aquatic toxicology (Amsterdam, Netherlands), 66(1), 15–23. https://doi.org/10.1016/j.aquatox.2003.06.004
Hossain, M. S., Larsson, A., Scherbak, N., Olsson, P. E., & Orban, L. (2008). Zebrafish androgen receptor: isolation, molecular, and biochemical characterization. Biology of reproduction, 78(2), 361–369. https://doi.org/10.1095/biolreprod.107.062018
Larsen, M. G., & Baatrup, E. (2010). Functional behavior and reproduction in androgenic sex reversed zebrafish (Danio rerio). Environmental toxicology and chemistry, 29(8), 1828–1833. https://doi.org/10.1002/etc.214
Leet, J. K., Gall, H. E., & Sepúlveda, M. S. (2011). A review of studies on androgen and estrogen exposure in fish early life stages: effects on gene and hormonal control of sexual differentiation. Journal of applied toxicology : JAT, 31(5), 379–398. https://doi.org/10.1002/jat.1682
Marty, M. S., Blankinship, A., Chambers, J., Constantine, L., Kloas, W., Kumar, A., Lagadic, L., Meador, J., Pickford, D., Schwarz, T., & Verslycke, T. (2017). Population-relevant endpoints in the evaluation of endocrine-active substances (EAS) for ecotoxicological hazard and risk assessment. Integrated environmental assessment and management, 13(2), 317–330. https://doi.org/10.1002/ieam.1887
Miller, D.H., D.L. Villeneuve, K.J. Santana-Rodriguez and G.T. Ankley. 2022. A multi-dimensional matrix model for predicting the effects of male-biased sex ratios on fish populations. Environmental Toxicology and Chemistry. 41, 1066-1077.
Morthorst, J. E., Holbech, H., & Bjerregaard, P. (2010). Trenbolone causes irreversible masculinization of zebrafish at environmentally relevant concentrations. Aquatic toxicology (Amsterdam, Netherlands), 98(4), 336–343. https://doi.org/10.1016/j.aquatox.2010.03.008
Örn, S., Holbech, H., & Norrgren, L. (2016). Sexual disruption in zebrafish (Danio rerio) exposed to mixtures of 17α-ethinylestradiol and 17β-trenbolone. Environmental toxicology and pharmacology, 41, 225–231. https://doi.org/10.1016/j.etap.2015.12.010
Orn, S., Holbech, H., Madsen, T. H., Norrgren, L., & Petersen, G. I. (2003). Gonad development and vitellogenin production in zebrafish (Danio rerio) exposed to ethinylestradiol and methyltestosterone. Aquatic toxicology (Amsterdam, Netherlands), 65(4), 397–411. https://doi.org/10.1016/s0166-445x(03)00177-2
Orn, S., Yamani, S., & Norrgren, L. (2006). Comparison of vitellogenin induction, sex ratio, and gonad morphology between zebrafish and Japanese medaka after exposure to 17alpha-ethinylestradiol and 17beta-trenbolone. Archives of environmental contamination and toxicology, 51(2), 237–243. https://doi.org/10.1007/s00244-005-0103-y
Pandian, T. & Sheela, s. (1995) Hormonal induction of sex reversal in fish. Aquaculture, 138 (1–4), 1-22. https://doi.org/10.1016/0044-8486(95)01075-0.
Piferrer, F., Baker, I. J., & Donaldson, E. M. (1993). Effects of natural, synthetic, aromatizable, and nonaromatizable androgens in inducing male sex differentiation in genotypic female chinook salmon (Oncorhynchus tshawytscha). General and comparative endocrinology, 91(1), 59–65. https://doi.org/10.1006/gcen.1993.1104
Pratt, W. B., & Toft, D. O. (1997). Steroid receptor interactions with heat shock protein and immunophilin chaperones. Endocrine reviews, 18(3), 306–360. https://doi.org/10.1210/edrv.18.3.0303
Seki, M., Yokota, H., Matsubara, H., Maeda, M., Tadokoro, H., & Kobayashi, K. (2004). Fish full life-cycle testing for androgen methyltestosterone on medaka (Oryzias latipes). Environmental toxicology and chemistry, 23(3), 774–781. https://doi.org/10.1897/03-26
Shi, W. J., Jiang, Y. X., Huang, G. Y., Zhao, J. L., Zhang, J. N., Liu, Y. S., Xie, L. T., & Ying, G. G. (2018). Dydrogesterone Causes Male Bias and Accelerates Sperm Maturation in Zebrafish ( Danio rerio). Environmental science & technology, 52(15), 8903–8911. https://doi.org/10.1021/acs.est.8b02556
Thornton J. W. (2001). Evolution of vertebrate steroid receptors from an ancestral estrogen receptor by ligand exploitation and serial genome expansions. Proceedings of the National Academy of Sciences of the United States of America, 98(10), 5671–5676. https://doi.org/10.1073/pnas.091553298
Wilson, V. S., Cardon, M. C., Thornton, J., Korte, J. J., Ankley, G. T., Welch, J., Gray, L. E., Jr, & Hartig, P. C. (2004). Cloning and in vitro expression and characterization of the androgen receptor and isolation of estrogen receptor alpha from the fathead Minnow (Pimephales promelas). Environmental science & technology, 38(23), 6314–6321. https://doi.org/10.1021/es049771j
Yu, G., Zhang, D., Liu, W., Wang, J., Liu, X., Zhou, C., Gui, J., & Xiao, W. (2018). Zebrafish androgen receptor is required for spermatogenesis and maintenance of ovarian function. Oncotarget, 9(36), 24320–24334. https://doi.org/10.18632/oncotarget.24407