
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 410
Title
GSK3beta inactivation leading to increased mortality via defects in developing inner ear
Short name
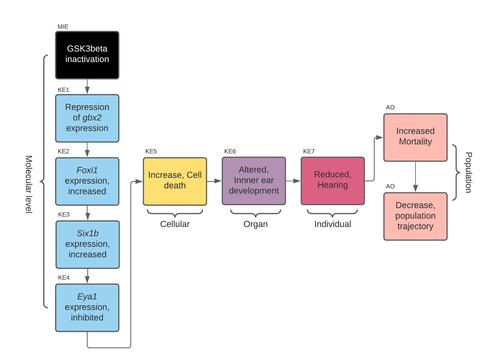
Graphical Representation
Point of Contact
Contributors
- Vid Modic
- Anze Zupanic
- Cataia Ives
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| foxi1 expression, increased | October 28, 2021 12:09 |
| six1b expression, increased | August 12, 2021 14:13 |
| eya1 expression, inhibited | October 07, 2021 12:59 |
| Increase, Cell death | March 22, 2023 11:07 |
| altered, inner ear development | August 24, 2021 07:50 |
| Reduced, Hearing | February 18, 2019 10:50 |
| GSK3beta inactivation | October 05, 2021 06:20 |
| Repression of Gbx2 expression | October 07, 2021 12:58 |
| Increased Mortality | July 08, 2022 07:32 |
| Decrease, Population growth rate | January 03, 2023 09:09 |
| GSK3beta inactivation leads to Repression of Gbx2 expression | December 12, 2021 12:23 |
| Repression of Gbx2 expression leads to foxi1 expression, increased | August 13, 2021 11:39 |
| foxi1 expression, increased leads to six1b expression, increased | October 28, 2021 12:58 |
| six1b expression, increased leads to eya1 expression, inhibited | August 13, 2021 14:44 |
| eya1 expression, inhibited leads to Increase, Cell death | August 22, 2021 15:17 |
| Increase, Cell death leads to Altered, inner ear development | August 29, 2021 09:05 |
| Altered, inner ear development leads to Reduced, Hearing | August 23, 2021 18:16 |
| Reduced, Hearing leads to Increased Mortality | December 08, 2020 03:26 |
| Increased Mortality leads to Decrease, Population growth rate | July 08, 2022 08:29 |
| BIO (6-bromoindirubin-3’-oxime) | May 29, 2019 21:19 |
Abstract
The focus of this AOP is on inactivation of Glycogen synthase kinase 3 beta (Gsk3b) by different chemicals which leads to defects in developing inner ear of zebrafish. Inactivation of Gsk3b leads to repressed expression of gbx2 (KE1) which consequently increases expression of two genes foxi1 (KE2) and six1b (KE3). Increase in six1b expression leads to inhibited expression of eya1 (KE4). Changes on molecular level (MIE-KE4) leads to changes at cellular level such as increased cell death in developing inner ear (KE5). Alterations in inner ear (KE6) translate to (AO) decrease in population trajectory through reduced hearing (KE7) and increased mortality (AO). An overall assessment of this AOP shows that there is low to moderate biological plausibility to suggest a qualitative link between the repression of Gsk3b expression to the KE4-cell death within developing inner ear and high evidence linking KE5 to increased mortality (AO). Currently there is not enough data for an appropriate assessment of essentiality of KEs and empirical support. KEs on molecular level have some uncertainties like foxi1 loss of function experiment resulting in no expression of six1b in otic placode and inconsistencies between zebrafish and mouse (six1b and eya1 role in otic placode development).
AOP Development Strategy
Context
The motivation behind building the AOP was methodological. Our team has recently developed molecular causal networks for developmental cardiotoxicity and neurotoxicity in zebrafish (doi.org/10.1021/acs.chemrestox.0c00095). These networks are highly curated, but rather large, going from adverse outcomes on the organ level upstream to wherever evidence takes us (many times finishing at what would be called MIEs). As there are many causal networks already present on the http://causalbionet.com/ (mostly for humans and for lung conditions), we were wondering how the rich knowledge available in causal pathways could be translated to AOPs. The AOP described in this document is one such example.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1647 | GSK3beta inactivation | GSK3beta inactivation |
| KE | 1902 | Repression of Gbx2 expression | Repression of Gbx2 expression |
| KE | 1903 | foxi1 expression, increased | foxi1 expression, increased |
| KE | 1904 | six1b expression, increased | six1b expression, increased |
| KE | 1905 | eya1 expression, inhibited | eya1 expression, inhibited |
| KE | 1825 | Increase, Cell death | Increase, Cell death |
| KE | 1930 | altered, inner ear development | Altered, inner ear development |
| KE | 1008 | Reduced, Hearing | Reduced, Hearing |
| AO | 351 | Increased Mortality | Increased Mortality |
| AO | 360 | Decrease, Population growth rate | Decrease, Population growth rate |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| GSK3beta inactivation leads to Repression of Gbx2 expression | adjacent | High | Low |
| Repression of Gbx2 expression leads to foxi1 expression, increased | adjacent | Moderate | Low |
| foxi1 expression, increased leads to six1b expression, increased | adjacent | Low | Low |
| six1b expression, increased leads to eya1 expression, inhibited | adjacent | Low | Low |
| eya1 expression, inhibited leads to Increase, Cell death | adjacent | High | Low |
| Increase, Cell death leads to Altered, inner ear development | adjacent | High | Low |
| Altered, inner ear development leads to Reduced, Hearing | adjacent | High | Low |
| Reduced, Hearing leads to Increased Mortality | adjacent | High | Low |
| Increased Mortality leads to Decrease, Population growth rate | adjacent | High | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| During brain development | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| zebrafish | Danio rerio | High | NCBI |
Sex Applicability
Overall Assessment of the AOP
An overall assessment of this AOP shows that there is low to moderate biological plausibility to suggest a qualitative link between the inactivation of Gsk3b to the KE4-cell death within developing inner ear and high evidence linking KE5 to increased mortality (AO). Biological plausibility is considered moderate because there is ample evidence from gain- and loss- of function experiments and knock out animal models that support the relationships between key events which are consistent with current biological knowledge, but there is mostly indirect evidence linking KEs on molecular level. KEs on molecular level have some uncertainties like foxi1 loss of function experiment resulting in no expression of six1b in otic placode (due to absence of otic placode) and inconsistencies across species (zebrafish, mouse). The evidence for essentiality of the KEs is mostly missing therefore the overall assessment of essentiality is low. The same goes for empirical support, currently there is no evidence for empirical support. Additional studies are needed to obtain data for empirical support, therefore, the empirical support of KERs is considered is low.
Domain of Applicability
Life stage: The current AOP is applicable from 2-8 cell stage (1,25 hpf; start of Gsk3b expression in zebrafish) (Valenti, 2015) up to 96 hpf wich is the expression limit of six1b in the developing inner ear (Webb & Shirey, 2003).
Taxonomic: This AOP is based on experimental evidence from studies on zebrafish, but is potentially also relevant to other vertebrates, because of conservation of all involved key events (Wnt signalling-Gsk3b, Gbx2, Eya1). But there are certain differences especially between zebrafish and mouse. Foxi1 gene is critical for zebrafish otic induction (Solomon et al., 2003), while it is not essential for this process in mice (Hulander et al., 2003). Interactions between Six1b and other members ofthe Pax–Six–Eya–Dach gene network, such as Eya1, also seem to differ between mouse and zebrafish (Li et al., 2003; Zheng et al., 2003).
Sex: Sex differences are typically not investigated in tests using early life stages of zebrafish and it is currently unclear whether sex-related differences are important in this AOP.
Essentiality of the Key Events
| Support for Essentiality of KEs |
Defining Question: Are downstream KEs and/or the AO prevented if an upstream KE is blocked?
|
| MIE: Gsk3b inactivation | Low: No experimental evidence of essentiality. |
| KE1: Repression of gbx2 expression | Low: No experimental evidence of essentiality. |
| KE2: Increased foxi1 expression | High: When foxi1 is knock down no expression of six1b is detected in otocyst (Bricaud and Collazo, 2006). |
| KE3: Increased six1b expression | Moderate: Six1b gain/loss-of-function experiment results indicate that in both cases normal development of inner ear is affected (KE5) (Bricaud and Collazo, 2006). |
| KE4: Inhibited eya1 expression | Low: No experimental evidence of essentiality. |
| KE5: Increased cell death | High: One of key players in normal development of sensory organs (KE6) (Whitfield et al., 2002; Kozlowski et al., 2005). |
| KE6: Altered inner ear development | Low: No experimental evidence of essentiality. |
| KE7: Reduced hearing | Moderate: One of the factors that are responsible for higher rate of mortality in fish (KE8) (Kasumyan, 2009). |
| AO: Increased mortality | High: Inability to perceive the environment leads to increase in mortality (Besson et al., 2020). |
| AO: Decrease of population trajectory | High: decrease in population trajectory is an imminent result of increased mortality (Rearick et al., 2018). |
Evidence Assessment
| Support for Biological Plausibility of KERs |
Defining Question: Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge?
|
| KER1: Gsk3b inactivation leads to repression of gbx2 expression | High: There is extensive evidence linking inhibition of Gsk3b to activation of canonical Wnt pathway for which Gbx2 is representative marker. |
| KER2: Repression of gbx2 expression leads to increased foxi1 expression | Moderate: Extensive evidence that Gbx2 represses many developmental regulatory genes such as foxi1, but multifunctional nature of Gbx2 is still unknown. |
| KER3: Increased foxi1 expression leads to increased six1b expression | Low: Relationship was confirmed with loss-of-function experiment, but the connection could be secondary to the overall absence of otic placode. |
| KER4: Increased six1b expression leads to inhibited eya1 expression | Low: Mutual regulation and interactions of both entities have not yet been well researched and described. Inconsistencies in zebrafish and mouse models. |
| KER5: Inhibited eya1 expression leads to increased cell death | High: Extensive evidence of relationship in vertebrate models. |
| KER6: Increased cell death leads to altered inner ear development | High: Extensive understanding that inner ear development depends on correct regulation of cell death in precursor cells and tissues. |
| KER7: Altered inner ear development leads to reduced hearing | High: Extensive understanding of defects in the development of inner ear and outcomes suggestive of deafness. |
| KER8: Reduced hearing leads to increased mortality | High: Extensive understanding that defective hearing decreases survival in natural setting. |
| KER9: Increased mortality leads to decrease of population trajectory | High: Extensive understanding that increased mortality on individual level decreases population trajectory. |
Empirical support: Currently there is no sufficient evidence to estimate the weight of the evidence of empirical support for KERs in this AOP. Further more specific research on the relationships between the entities involved in the AOP is needed.
Known Modulating Factors
Quantitative Understanding
Data to support the quantitative understanding of this AOP is currently lacking.
Considerations for Potential Applications of the AOP (optional)
References
Besson, M. et. al. 2020. „Anthropogenic stressors impact fish sensory development and survival via thyroid disruption“. Nature Communications 2020 11:1 11(1): 1–10.
Bricaud, O., Leslie, A. C., & Gonda, S. (2006). Development/Plasticity/Repair The Transcription Factor six1 Inhibits Neuronal and Promotes Hair Cell Fate in the Developing Zebrafish (Danio rerio) Inner Ear. Journal of Neuroscience, 26(41), 10438–10451. https://doi.org/10.1523/JNEUROSCI.1025-06.2006
Hulander, M., Kiernan, A., Blomqvist, S., Carlsson, P., Samuelsson, E., Johansson, B., Steel, K., & Enerbäck, S. (2003). Lack of pendrin expression leads to deafness and expansion of the endolymphatic compartment in inner ears of Foxi1null mutant mice 2013. Development, 130, 2013–2025. https://doi.org/10.1242/dev.00376
Kasumyan, A. O. 2009. „Acoustic signaling in fish“. Journal of Ichthyology 2009 49:11 49(11): 963–1020.
Kozlowski, D. J., Whitfield, T. T., Hukriede, N. A., Lam, W. K., & Weinberg, E. S. (2005). The zebrafish dog-eared mutation disrupts eya1, a gene required for cell survival and differentiation in the inner ear and lateral line. Developmental Biology, 277(1), 27–41. https://doi.org/10.1016/j.ydbio.2004.08.033
Li, X., Oghi, K. A., Zhang, J., Krones, A., Bush, K. T., Glass, C. K., Nigam, S. K., Aggarwal, A. K., Maas, R., Rose, D. W., & Rosenfeld, M. G. (2003). Eya protein phosphatase activity regulates Six1-Dach-Eya transcriptional effects in mammalian organogenesis. Nature, 426(6964), 247–254. https://doi.org/10.1038/nature02083
Rearick, Daniel C., Jessica Ward, Paul Venturelli and Heiko Schoenfuss. 2018. „Environmental oestrogens cause predation-induced population decline in a freshwater fish“. Royal Society Open Science 5(10).
Sklirou, A. D. et al. (2017) ‘6-bromo-indirubin-3′-oxime (6BIO), a Glycogen synthase kinase-3β inhibitor, activates cytoprotective cellular modules and suppresses cellular senescence-mediated biomolecular damage in human fibroblasts’, Sci Rep, 7, p. 11713. doi: 10.1038/s41598-017-11662-7.
Solomon, K. S., Kudoh, T., Dawid, I. B., & Fritz, A. (2003). Zebrafish foxi1 mediates otic placode formation and jaw development. Development, 130(5), 929–940. https://doi.org/10.1242/dev.00308
Valenti, Fabio et al. 2015. „The Increase in Maternal Expression of axin1 and axin2 Contribute to the Zebrafish Mutant Ichabod Ventralized Phenotype“. Journal of Cellular Biochemistry 116(3): 418–30.
Wang, Z. et al. (2018) ‘The role of gastrulation brain homeobox 2 (gbx2) in the development of the ventral telencephalon in zebrafish embryos’, Differentiation, 99(September 2017), pp. 28–40. doi: 10.1016/j.diff.2017.12.005.
Webb, J. F., & Shirey, J. E. (2003). Postembryonic Development of the Cranial Lateral Line Canals and Neuromasts in Zebrafish. Developmental Dynamics, 228(3), 370–385. https://doi.org/10.1002/dvdy.10385
Whitfield, T. T., Riley, B. B., Chiang, M. Y., & Phillips, B. (2002). Development of the zebrafish inner ear. Developmental Dynamics, 223(4), 427–458. https://doi.org/10.1002/dvdy.10073
Zheng, W., Huang, L., Wei, Z.-B., Silvius, D., Tang, B., & Pin-Xian, X. (2003). The role of Six1 in mammalian auditory system development. Development, 130, 3989–4000. https://doi.org/10.1242/dev.00628