
This AOP is open for adoption and licensed under the BY-SA license. The BY-SA license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 213
Title
Inhibition of fatty acid beta oxidation leading to nonalcoholic steatohepatitis (NASH)
Short name
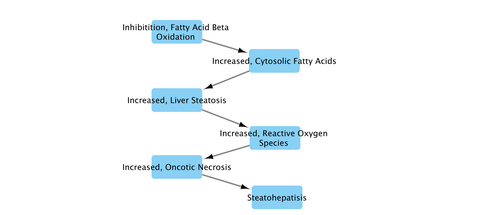
Graphical Representation
Point of Contact
Contributors
- Edward Perkins
- Lyle Burgoon
- Arthur Author
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increase, cytosolic fatty acid | November 27, 2017 12:20 |
| N/A, Steatohepatisis | November 27, 2017 13:41 |
| Inhibition, Fatty Acid Beta Oxidation | November 27, 2017 12:15 |
| Increased, Liver Steatosis | May 21, 2024 10:00 |
| Increased, Reactive oxygen species | January 29, 2025 12:41 |
| Increased, Oncotic Necrosis | November 27, 2017 13:31 |
| Inhibition of fatty acid beta oxidation leads to Increase, cytosolic fatty acid | November 13, 2017 12:51 |
| Increase, cytosolic fatty acid leads to Increased, Liver Steatosis | November 14, 2017 10:25 |
| Increased, Liver Steatosis leads to Increased, Reactive oxygen species | November 14, 2017 10:26 |
| Increased, Reactive oxygen species leads to increased, oncotic necrosis | November 14, 2017 10:27 |
| increased, oncotic necrosis leads to steatohepatitis | November 14, 2017 10:28 |
Abstract
Non-alcoholic steatohepatitis (NASH) is a significant disease of the liver. NASH presents as steatosis (fatty liver) and hepatitis (liver inflammation). NASH is on a spectrum of liver disease, starting at steatosis and ultimately leading to cirrhosis (liver fibrosis) if chemical exposure and injury continues. This AOP is focused on fatty acid beta oxidation and its contribution as a molecular initiating event in the formation of NASH. Steatosis is ultimately a net increase in fatty acids within hepatocytes. This can be from a net decrease in efflux (e.g., influx >> efflux), a net decrease in overall fatty acid oxidation/metabolism to glucose and intermediates, or a combination of these factors. In this AOP, our MIE is the inhibition of fatty acid beta oxidation (FABO). This leads to an overall increase in fatty acids. These fatty acids undergo lipid peroxidation resulting in fatty acid free radicals. When the reduction potential of the cell is overwhelmed, the free radicals lead to oncotic cell death, and the release of signals that stimulate inflammatory cell infiltration.
AOP Development Strategy
Context
Steatosis is the condition where an abnormal amount of fat is being stored within the liver. The liver is the site where sugars and fats are converted for the purposes of supplying energy to the rest of the body. The liver will convert glucose to fatty acids and package them as triglycerides for distribution throughout the body via the bloodstream and storage in adipose tissue. The liver also takes in fatty acids and triglycerides, and oxidizes them back to glucose for distribution throughout the body. When the influx/efflux and metabolism of fatty acids is altered, leading to a net increase in cellular fatty acids, the result is steatosis. As steatosis progresses, these fatty acids may lead to oxidative stress that ultimately leads to oncotic necrosis (cell death) and inflammatory cell infiltration (inflammation). This is termed steatohepatitis.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1490 | Inhibition, Fatty Acid Beta Oxidation | Inhibition of fatty acid beta oxidation |
| KE | 1305 | Increase, cytosolic fatty acid | Increase, cytosolic fatty acid |
| KE | 459 | Increased, Liver Steatosis | Increased, Liver Steatosis |
| KE | 1115 | Increased, Reactive oxygen species | Increased, Reactive oxygen species |
| KE | 1491 | Increased, Oncotic Necrosis | increased, oncotic necrosis |
| AO | 1489 | N/A, Steatohepatisis | steatohepatitis |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Inhibition of fatty acid beta oxidation leads to Increase, cytosolic fatty acid | adjacent | High | High |
| Increase, cytosolic fatty acid leads to Increased, Liver Steatosis | adjacent | High | High |
| Increased, Liver Steatosis leads to Increased, Reactive oxygen species | adjacent | High | Low |
| Increased, Reactive oxygen species leads to increased, oncotic necrosis | adjacent | High | Low |
| increased, oncotic necrosis leads to steatohepatitis | adjacent | High | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| Vertebrates | Vertebrates | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP
Overall, the confidence in this AOP is high. Most of the components of fatty acid metabolism are well known biochemical pathways that are well studied. Although components of this AOP occur within cells, the overall diagnosis of this condition is typically at the organ level. The adverse outcome is known to occur in vertebrates in the liver. Steatosis has been used as a regulatory endpoint or potential regulatory endpoint in many assessments from the US EPA.
This AOP begins at inhibition of fatty acid beta-oxidation. This results in an increase in the overall amount of cytosolic fatty acids in the cytoplasm (under certain assumptions mentioned below). To lead to toxicity, the cytoplasm must become a net sink of fatty acids, with the rate of fatty acid deposition being greater than the rate of fatty acid efflux. This increase in cytosolic fatty acids increases the likelihood that reactive oxygen species will have lipids to undergo peroxidation, leading to a cascade of protein and membrane damage once the cellular reduction system is depleted. This damage leads to oncotic necrosis which results in the spillage of cytoplasmic contents, which triggers an inflammatory response. The result is steatohepatitis.
Domain of Applicability
The domain of applicability for this AOP is all sexes in vertebrates at all life stages with a functional liver.
Essentiality of the Key Events
It has been demonstrated that pharmacological inhibition of fatty acid beta-oxidation is sufficient to cause steatosis (1-4).
Steatosis leading to inflammation (termed non-alcoholic steatohepatitis; NASH) is well known. Some hypotheses of how this may occur are discussed in (5). As steatosis is necessary for a diagnosis of NASH, it is essential by definition.
Evidence Assessment
NASH is a well-known adverse event that has been well-studied in the toxicology literature, especially in support of pharmaceutical development. The key events involved in NASH are relatively well-studied. There are many forking paths that lead to NASH, with inhibition of fatty acid beta-oxidation being just one. The progression from pre-steatosis to steatohepatitis is reviewed in (6). The key events are generally well-known; however, what is less well-known are the exact molecular key event components that lead from steatosis to activation of the hepatic Kuppfer cells (liver macrophages) which activate hepatitis. However, for the purposes of the AOP, this level of granular knowledge is not required.
Known Modulating Factors
Quantitative Understanding
Based on information available in (2-3) it is possible to estimate the amount of fatty acid beta-oxidation inhibition that is required to lead to steatosis. The quantitative evidence for the steps leading from steatosis to hepatitis are relatively weak. This process is not entirely well-known at the granular, molecular level, and there is a derth of quantitative studies at this time.
Considerations for Potential Applications of the AOP (optional)
This AOP was developed to support the development of probabilistic AOPs, specifically the AOP Bayesian Network (AOPBN) for Steatosis featured in the BISCT tool (BISCT: Bayesian Inference for Substance and Chemical Toxicity ). The Steatosis AOPBN is intended for use in:
- Chemical Screening, when coupled with HTS assays,
- Hazard Identification, for risk screening and risk assessment
The AOPBN in BISCT is compatible with data from HTS assays, high content assays (e.g., toxicogenomics), molecular assays, and more traditional assays.
BISCT is open source software, available at GitHub
References
- Begriche K, Igoudjil A, Pessayre D, Fromenty B. Mitochondrial dysfunction in NASH: causes, consequences and possible means to prevent it. Mitochondrion. 2006 Feb;6(1):1-28. Epub 2006 Jan 5.
- E Freneaux, B Fromenty, A Berson, G Labbe, C Degott, P Letteron, D Larrey and D Pessayre. Stereoselective and nonstereoselective effects of ibuprofen enantiomers on mitochondrial beta-oxidation of fatty acids. Journal of Pharmacology and Experimental Therapeutics November 1990, 255 (2) 529-535.
- J Geneve, B Hayat-Bonan, G Labbe, C Degott, P Letteron, E Freneaux, T L Dinh, D Larrey and D Pessayre. Inhibition of mitochondrial beta-oxidation of fatty acids by pirprofen. Role in microvesicular steatosis due to this nonsteroidal anti-inflammatory drug. Journal of Pharmacology and Experimental Therapeutics September 1987, 242 (3) 1133-1137.
- Seung-Hoi Koo. Nonalcoholic fatty liver disease: molecular mechanisms for the hepatic steatosis. Clin Mol Hepatol. 2013 Sep; 19(3): 210–215.
- Herbert Tilg, Alexander R. Moschen. Evolution of inflammation in nonalcoholic fatty liver disease: The multiple parallel hits hypothesis. Hepatology 2010;52:1836-1846.
- Satapathy SK, Kuwajima V, Nadelson J, Atiq O, Sanyal AJ. Drug-induced fatty liver disease: An overview of pathogenesis and management. Ann Hepatol. 2015 Nov-Dec;14(6):789-806. doi: 10.5604/16652681.1171749.