
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 307
Title
Decreased testosterone synthesis leading to short anogenital distance (AGD) in male (mammalian) offspring
Short name
Graphical Representation
Point of Contact
Contributors
- Terje Svingen
- Cataia Ives
Coaches
- Judy Choi
- Shihori Tanabe
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.90 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Reduction, Testosterone synthesis in Leydig cells | September 16, 2017 10:14 |
| Decrease, testosterone levels | May 24, 2024 12:27 |
| Decrease, dihydrotestosterone (DHT) level | April 05, 2024 08:10 |
| Decrease, androgen receptor activation | April 05, 2024 08:19 |
| Altered, Transcription of genes by the androgen receptor | April 05, 2024 09:28 |
| anogenital distance (AGD), decreased | December 22, 2022 05:18 |
| Reduction, Testosterone synthesis in Leydig cells leads to Decrease, testosterone levels | May 01, 2024 16:10 |
| Decrease, testosterone levels leads to Decrease, AR activation | April 05, 2024 09:18 |
| Decrease, testosterone levels leads to Decrease, DHT level | October 19, 2023 08:31 |
| Decrease, DHT level leads to Decrease, AR activation | April 05, 2024 08:48 |
| Decrease, AR activation leads to Altered, Transcription of genes by the AR | April 05, 2024 08:50 |
| Altered, Transcription of genes by the AR leads to AGD, decreased | May 11, 2020 07:04 |
| Decrease, AR activation leads to AGD, decreased | December 22, 2022 05:20 |
| Dibutyl phthalate | November 29, 2016 18:42 |
| Bis(2-ethylhexyl) phthalate | November 29, 2016 18:42 |
Abstract
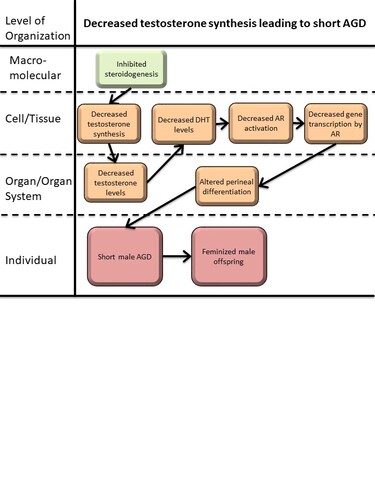
This AOP links decreased testosterone synthesis by fetal Leydig cells with short anogenital distance (AGD) in male offspring. A short AGD around birth is a marker for feminization of male fetuses and is associated with male reproductive disorders, including reduced fertility in adulthood. Although a short AGD is not necessarily ‘adverse’ from a human health perspective, it is considered an ‘adverse outcome’ in OECD test guidelines; AGD measurements are mandatory in specific tests for developmental and reproductive toxicity in chemical risk assessment (TG 443, TG 421/422, TG 414).
Testosterone is primarily synthesized by fetal Leydig cells of the fetal testes by the process of steroidogenesis. The precursor molecule cholesterol is converted to testosterone via several enzymatic steps and includes for instance key CYP enzymes, CYP11 and CYP17. Following synthesis, testosterone is released into the circulation and transported to target tissues and organs where it initiates masculinization processes. Under normal physiological conditions, testosterone produced by the testicles, is converted in peripheral tissues by 5α-reductase into DHT, which in turn binds AR and activates downstream target genes. AR signaling is necessary for masculinization of the developing fetus, including differentiation of the levator ani/bulbocavernosus (LABC) muscle complex in males. The LABC complex does not develop in the absence, or low levels of, androgen signaling, as in female fetuses.
The key events in this pathway is inhibition of testosterone synthesis in the fetal Leydig cells. In turn, this results in reduced circulating testosterone levels and less DHT (converted by 5α-reductase). Low DHT fails to properly activate AR in target tissues, including the developing perineal region, which leads to failure to properly masculinize the perineum/LABC complex and ultimately a short AGD.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| KE | 413 | Reduction, Testosterone synthesis in Leydig cells | Reduction, Testosterone synthesis in Leydig cells |
| KE | 1690 | Decrease, testosterone levels | Decrease, testosterone levels |
| KE | 1613 | Decrease, dihydrotestosterone (DHT) level | Decrease, DHT level |
| KE | 1614 | Decrease, androgen receptor activation | Decrease, AR activation |
| KE | 286 | Altered, Transcription of genes by the androgen receptor | Altered, Transcription of genes by the AR |
| AO | 1688 | anogenital distance (AGD), decreased | AGD, decreased |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Reduction, Testosterone synthesis in Leydig cells leads to Decrease, testosterone levels | adjacent | High | Moderate |
| Decrease, testosterone levels leads to Decrease, DHT level | adjacent | Moderate | Low |
| Decrease, DHT level leads to Decrease, AR activation | adjacent | High | Moderate |
| Decrease, AR activation leads to Altered, Transcription of genes by the AR | adjacent | High | Moderate |
| Altered, Transcription of genes by the AR leads to AGD, decreased | adjacent | Moderate | Moderate |
| Decrease, testosterone levels leads to Decrease, AR activation | non-adjacent | Moderate | Moderate |
| Decrease, AR activation leads to AGD, decreased | non-adjacent |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Foetal | High |
| Pregnancy | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male | High |
Overall Assessment of the AOP
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
1. Schwartz CL, Christiansen S, Vinggaard AM, Axelstad M, Hass U and Svingen T (2019), Anogenital distance as a toxicological or clinical marker for fetal androgen action and risk for reproductive disorders. Arch Toxicol 93: 253-272.