
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 379
Title
Binding to ACE2 leading to thrombosis and disseminated intravascular coagulation
Short name
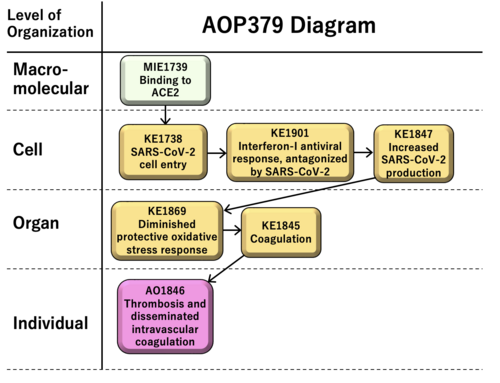
Graphical Representation
Point of Contact
Contributors
- Shihori Tanabe
- Arthur Author
Coaches
- Cinzia La Rocca
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.96 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Coagulation | August 30, 2023 20:39 |
| Thrombosis and Disseminated Intravascular Coagulation | November 25, 2022 01:38 |
| Increased SARS-CoV-2 production | June 14, 2022 08:49 |
| SARS-CoV-2 cell entry | April 04, 2023 07:39 |
| Diminished protective oxidative stress response | October 30, 2023 03:36 |
| Binding to ACE2 | August 30, 2023 20:36 |
| Interferon-I antiviral response, antagonized by SARS-CoV-2 | December 15, 2023 14:27 |
| Binding to ACE2 leads to SARS-CoV-2 cell entry | February 07, 2023 23:24 |
| SARS-CoV-2 cell entry leads to IFN-I response, antagonized | December 12, 2023 15:15 |
| IFN-I response, antagonized leads to SARS-CoV-2 production | December 14, 2023 15:23 |
| SARS-CoV-2 production leads to Diminished Protective Response to ROS | September 26, 2023 01:47 |
| Diminished Protective Response to ROS leads to Coagulation | January 26, 2024 00:52 |
| Coagulation leads to Diminished Protective Response to ROS | October 17, 2023 00:34 |
| Coagulation leads to Thrombosis and DIC | February 05, 2023 20:49 |
| Stressor:624 SARS-CoV-2 | April 20, 2021 03:40 |
Abstract
Coronavirus disease-19 (COVID-19) is circulating all over the world. To understand and find a way of the COVID-19 treatment, the signaling pathway and therapeutic mechanism of COVID-19 should be investigated. The pathogenesis of COVID-19 includes molecular networks such as the binding of the membrane proteins, signaling pathways, and RNA replication. The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which is a new type of coronavirus causing COVID-19, infects the cells via the binding of the membrane proteins of human cells and is internalized by the cells. The viral genome is replicated by RNA-dependent RNA polymerase (RdRp), followed by the packaging and releasing of the viral particles. These steps can be the main targets for the therapeutics of COVID-19. The AOP379 "Binding to ACE2 leading to thrombosis and disseminated intravascular coagulation" consists of the molecular initiating event (MIE) as “Binding to ACE2” (KE1739), key events (KEs) as “SARS-CoV-2 cell entry” (KE1738), “Interferon-I antiviral response, antagonized by SARS-CoV-2” (KE1901), "Increased SARS-CoV-2 production" (KE1847), “Diminished protective oxidative stress response" (KE1869) and "Coagulation" (KE1845), and adverse outcome (AO) as "Thrombosis and Disseminated Intravascular Coagulation" (KE1846).
AOP Development Strategy
Context
Strategy
This AOP379 has been developed in the collaboration of Modelling the Pathogenesis of COVID-19 using the AOP framework (CIAO) international consortium (Clerbaux et al., 2022). Extensive efforts in discussion and literature search of the group crystalized into many AOPs related to coronavirus pathogenesis and coronavirus infectious disease 2019 (COVID-19), where the AOP379 focuses on the thrombosis and disseminated intravascular coagulation as adverse outcome of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection through reactive oxygen species-induced oxidative stress response (Tanabe et al., 2022).
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1739 | Binding to ACE2 | Binding to ACE2 |
| KE | 1738 | SARS-CoV-2 cell entry | SARS-CoV-2 cell entry |
| KE | 1901 | Interferon-I antiviral response, antagonized by SARS-CoV-2 | IFN-I response, antagonized |
| KE | 1847 | Increased SARS-CoV-2 production | SARS-CoV-2 production |
| KE | 1869 | Diminished protective oxidative stress response | Diminished Protective Response to ROS |
| KE | 1845 | Coagulation | Coagulation |
| AO | 1846 | Thrombosis and Disseminated Intravascular Coagulation | Thrombosis and DIC |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Binding to ACE2 leads to SARS-CoV-2 cell entry | adjacent | High | Moderate |
| SARS-CoV-2 cell entry leads to IFN-I response, antagonized | adjacent | Moderate | Moderate |
| IFN-I response, antagonized leads to SARS-CoV-2 production | adjacent | Moderate | Moderate |
| SARS-CoV-2 production leads to Diminished Protective Response to ROS | adjacent | Moderate | Not Specified |
| Diminished Protective Response to ROS leads to Coagulation | adjacent | Moderate | Not Specified |
| Coagulation leads to Diminished Protective Response to ROS | adjacent | Moderate | Not Specified |
| Coagulation leads to Thrombosis and DIC | adjacent | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | Moderate |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| Homo sapiens | Homo sapiens | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP
Domain of Applicability
This AOP is applicable to all sexes in Homo sapiens.
Essentiality of the Key Events
|
Essentiality of the KEs |
||||
|
Event |
ID and Title |
Direct Evidence |
Indirect Evidence |
No experimental evidence |
|
KE1 |
KE1738: SARS-CoV-2 cell entry |
** |
*** |
*** |
|
KE2 |
KE1901: Interferon-I antiviral response, antagonized by SARS-CoV-2 |
|
*** |
*** |
|
KE3 |
KE1847: Increased SARS-CoV-2 production |
** |
*** |
*** |
|
KE4 |
KE1869: Diminished protective oxidative stress response |
|
*** |
*** |
|
KE5 |
KE1845: Coagulation |
|
*** |
*** |
Evidence Assessment
|
[Evidence Assessment] |
|
|
1. Support for Biological Plausibility of KERs |
|
|
MIE => KE1: KER2056: Binding to ACE2 leads to SARS-CoV-2 cell entry |
Biological Plausibility of the MIE => KE1 is high. Rationale: Binding of SARS-CoV-2 to ACE2 cell-surface receptor initiates infection of SARS-CoV-2 where SARS-CoV-2 cell entry is essential (Zhou P et al., 2020, Benton DJ et al., 2020). |
|
KE1 => KE2: KER2496: SARS-CoV-2 cell entry leads to IFN-I response, antagonized |
Biological Plausibility of the KE1 => KE2 is moderate. Rationale: The expression of type I interferons (IFN-I) is antagonized by SARS-CoV-2 (Benton DJ et al., 2020). Individual viral proteins interact with and block host proteins in the IFN-I pathway or IFN-I stimulated genes. |
|
KE2 => KE3: KER2497: IFN-I response, antagonized leads to SARS-CoV-2 production |
Biological Plausibility of the KE2 => KE3 is moderate. Rationale: Inhibition of IFN-I response induces SARS-CoV-2 production. |
|
KE3 => KE4: KER2358: SARS-CoV-2 production leads to Diminished Protective Response to ROS |
Biological Plausibility of the KE3 => KE4 is moderate. Rationale: The fixation of SARS-CoV-2 in ACE2 receptor results in excessive production of pro-inflammatory and pro-oxidant agents (Ramdani and Bachari, 2020). |
|
KE4 => KE5: KER2359: Diminished Protective Response to ROS leads to Coagulation |
Biological Plausibility of the KE4 => KE5 is moderate. Rationale: The ROS results in lung injury and coagulopathy (Barrett CD et al., 2018). |
|
KE5 => KE4: KER2360: Coagulation leads to Diminished Protective Response to ROS |
Biological Plausibility of the KE5 => KE4 is moderate. Rationale: Coagulation imbalance induces oxidative stress (Robea MA et al., 2023). |
|
KE5 => AO: KER2290: Coagulation leads to Thrombosis and DIC |
Biological Plausibility of the KE5 => AO is high. Rationale: Extreme aggravation of blood coagulation induces multiple thrombi in the microvasculature, which leads to consumption coagulopathy followed by disseminated intravascular disease. |
|
2. Support for Essentiality of KEs |
|
|
AOP379 |
Rationale for Essentiality of KEs in the AOP is Moderate. |
|
3. Empirical Support for KERs |
|
|
MIE => KE1: KER2056: Binding to ACE2 leads to SARS-CoV-2 cell entry |
Empirical Support of the MIE => KE1 is moderate. Rationale: The SARS-CoV-2 binding to ACE2 induce fusion of the virus and cell membranes to release the virus genome into the cell. (Zhou P et al., 2020, Benton DJ et al., 2020). |
|
KE1 => KE2: KER2496: SARS-CoV-2 cell entry leads to IFN-I response, antagonized |
Empirical Support of the KE1 => KE2 is moderate. Rationale: SARS-CoV-2-infection induces IFN-I pathway reduction (Sui C et al., 2022, Blanco-Melo, 2020). |
|
KE2 => KE3: KER2497: IFN-I response, antagonized leads to SARS-CoV-2 production |
Empirical Support of the KE2 => KE3 is moderate. Rationale: Reduced IFN-I response induces SARS-CoV-2 production. |
|
KE3 => KE4: KER2358: SARS-CoV-2 production leads to Diminished Protective Response to ROS |
Empirical Support of the KE3 => KE4 is moderate. Rationale: As SARS-CoV-2 is attached to ACE2, ACE2 is not available within the renin-angiotensin system to convert Ang II to angiotensin-(1,7) resulting in Ang II to accumulate. Ang II stimulates membrane-bound NADPH oxidase, which in turn generate ROS and oxidative stress (Janardhan et al., 2020). |
|
KE4 => KE5: KER2359: Diminished Protective Response to ROS leads to Coagulation |
Empirical Support of the KE4 => KE5 is moderate. Rationale: The presence of ROS assists in the transformation of a circulating, non-oxidized, circular-shaped beta2-glycoprotein 1 into an oxidized J-shape, which binds to antiphospholipid antibodies such as anticardiolipin, lupus anticoagulant, and anti-beta2-GP1 antibodies (Janardhan et al., 2020). Domain V of beta2gP1 binds with the phospholipid layer of platelets or endothelial cells via Annexin (Janardhan et al., 2020). |
|
KE5 => KE4: KER2360: Coagulation leads to Diminished Protective Response to ROS |
Empirical Support of the KE5 => KE4 is moderate. Rationale: Excessive coagulation induces oxidative stress. |
|
KE5 => AO: KER2290: Coagulation leads to Thrombosis and DIC |
Empirical Support of the KE5 => AO is high. Rationale: Extreme aggravation of blood coagulation induces multiple thrombi in the microvasculature, which leads to consumption coagulopathy followed by disseminated intravascular disease. |
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
The AOP379 focuses on the coronavirus-induced thrombosis and disseminated intravascular coagulation, which may contribute to the development of therapeutics of COVID-19 and long COVID syndrome. The understanding of the mechanism of the coronavirus-induced vascular adverse outcome may predict adverse responses of COVID-19 vaccines.
References
Banerjee AK, Blanco MR, Bruce EA, Honson DD, Chen LM, Chow A, et al. SARS-CoV-2 Disrupts Splicing, Translation, and Protein Trafficking to Suppress Host Defenses. Cell. 2020;183(5):1325-39.e21.
Barrett CD, Hsu AT, Ellson CD, B YM, Kong YW, Greenwood JD, et al. Blood clotting and traumatic injury with shock mediates complement-dependent neutrophil priming for extracellular ROS, ROS-dependent organ injury and coagulopathy. Clin Exp Immunol. 2018;194(1):103-17.
Benton DJ, Wrobel AG, Xu P, Roustan C, Martin SR, Rosenthal PB, et al. Receptor binding and priming of the spike protein of SARS-CoV-2 for membrane fusion. Nature. 2020;588(7837):327-30.
Blanco Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Møller R, et al. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell. 181;(5):1036-1045.
Chen B, Tian EK, He B, Tian L, Han R, Wang S, et al. Overview of lethal human Coronaviruses. Signal Transduction and Targeted Therapy, 2020;5(1):89.
Clerbaux, L.-A., Amigó, N., Amorim, M. J., Bal-Price, A., Batista Leite, S., Beronius, A., Bezemer, G. F. G., Bostroem, A.-C., Carusi, A., Coecke, S., Concha, R., Daskalopoulos, E. P., De Bernardi, F., Edrosa, E., Edwards, S. W., Filipovska, J., Garcia-Reyero, N., Gavins, F. N. E., Halappanavar, S., Hargreaves, A. J., Hogberg, H. T., Huynh, M. T., Jacobson, D., Josephs-Spaulding, J., Kim, Y. J., Kong, H. J., Krebs, C. E., Lam, A., Landesmann, B., Layton, A., Lee, Y. O., Macmillan, D. S., Mantovani, A., Margiotta-Casaluci, L., Martens, M., Masereeuw, R., Mayasich, S. A., Mei, L. M., Mortensen, H., Munoz Pineiro, A., Nymark, P., Ohayon, E., Ojasi, J., Paini, A., Parissis, N., Parvatam, S., Pistollato, F., Sachana, M., Sørli, J. B., Sullivan, K. M., Sund, J., Tanabe, S., Tsaioun, K., Vinken, M., Viviani, L., Waspe, J., Willett, C. and Wittwehr, C. (2022) “COVID-19 through adverse outcome pathways: Building networks to better understand the disease – 3rd CIAO AOP Design Workshop”, ALTEX - Alternatives to animal experimentation, 39(2), pp. 322–335. doi: 10.14573/altex.2112161.
Cui J, Li F, Shi ZL. Origin and evolution of pathogenic Coronaviruses. Nature Reviews Microbiology. 2019;17(3):181-192.
Florindo HF, Kleiner R, Vaskovich-Koubi D, Acúrcio RC, Carreira B, Yeini,E, et al. Immune-mediated approaches against COVID-19. Nature Nanotechnology. 2020:15(8):630-45.
Janardhan V, Janardhan V, Kalousek V. COVID-19 as a Blood Clotting Disorder Masquerading as a Respiratory Illness: A Cerebrovascular Perspective and Therapeutic Implications for Stroke Thrombectomy. Journal of Neuroimaging. 2020;30(5):555-61.
Kowalewski J, Ray A. Predicting novel drugs for SARS-CoV-2 using machine learning from a & g 10 million chemical space. Heliyon. 2020;6(8).
Pizzorno A, Padey B, Julien T, Trouillet-Assant S, Traversier A, Errazuriz-Cerda E, et al. Characterization and Treatment of SARS-CoV-2 in Nasal and Bronchial Human Airway Epithelia. Cell Reports Medicine. 2020:1(4).
Ramdani LH and Bachari K. Potential therapeutic effects of resveratrol against SARS-CoV-2. Acta virologica 2020;64, 276-280
Riva L, Yuan S, Yin X, Martin-Sancho L, Matsunaga N, Pache L, et al. Discovery of SARS-CoV-2 antiviral drugs through large-scale compound repurposing. Nature. 2020.
Robea MA, Balmus I-M, Girleanu I, Huiban L, Muzica C, Ciobica A, et al. Coagulation Dysfunctions in Non-Alcoholic Fatty Liver Disease—Oxidative Stress and Inflammation Relevance. Medicina. 2023;59(9):1614.
Sui C, Xiao T, Zhang S, Zeng H, Zheng Y, Liu B, et al. SARS-CoV-2 NSP13 Inhibits Type I IFN Production by Degradation of TBK1 via p62-Dependent Selective Autophagy. The Journal of Immunology. 2022;208(3):753-61.
Tanabe S (2020a). Cellular Internalization and RNA Regulation of RNA virus. Adv Clin Med Res. 2020;1(1):1-3. https://www.genesispub.org/cellular-internalization-and-rna-regulation-of-rna-virus
Tanabe S (2020b). The Therapeutic Mechanism of COVID-19. J Clin Med Res. 2020;2(5):1-3. DOI: https://doi.org/10.37191/Mapsci-2582-4333-2(5)-048
Tanabe, S., Beaton, D., Chauhan, V., Choi, I., Danielsen, P. H., Delrue, N., Esterhuizen, M., Filipovska, J., FitzGerald, R., Fritsche, E., Gant, T., Garcia-Reyero, N., Helm, J., Huliganga, E., Jacobsen, N., Kay, J. E., Kim, Y.-J., Klose, J., La Rocca, C., Luettich, K., Mally, A., O’Brien, J., Poulsen, S. S., Rudel, R. A., Sovadinova, I., Tollefsen, K. E., Vogel, U., Yepiskoposyan, H. and Yauk, C. (2022) “Report of the 1st and 2nd Mystery of Reactive Oxygen Species Conferences”, ALTEX - Alternatives to animal experimentation, 39(2), pp. 336–338. doi: 10.14573/altex.2203011.
Zhou P, Yang X-L, Wang X-G, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270-3.