
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 393
Title
AOP for thyroid disorder caused by triphenyl phosphate via TRβ activation
Short name
Graphical Representation
Point of Contact
Contributors
- Fei Li
- Brendan Ferreri-Hanberry
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increased, Hepatic thyroid hormone uptake/transport | September 16, 2017 10:17 |
| Cell cycle, disrupted | June 30, 2021 02:56 |
| systemic inflammatory response syndrome | March 19, 2021 09:50 |
| Increase cell proliferation | December 19, 2018 11:25 |
| Apoptosis | February 28, 2024 09:40 |
| Altered, Thyroid hormone-dependent gene expression | December 09, 2020 14:22 |
| Triphenyl phosphate | March 30, 2020 16:54 |
Abstract
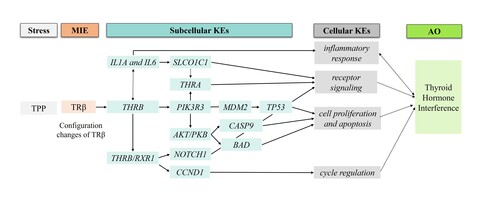
The critical molecular initiating event (MIE) was the configuration changes of C-terminal helix 12 (H12) of thyroid hormone receptor β (TRβ) induced by triphenyl phosphate (TPP) binding. The transcriptomic analysis extended the related key event (KE1) at the subcellular level, such as changes in gene expression levels for coding cycle regulation (CCND1), inflammatory response (IL1A and IL6), and cell proliferation and apoptosis (BAD, TP53 and CASP9). Then, the KE2 at cellular levels such as apoptosis, cell cycle control, and cell proliferation were influenced accordingly. As a result, these alterations led to thyroid disorder as adverse outcomes.
AOP Development Strategy
Context
A large number of studies reported that triphenyl phosphate (TPP) was detected from environmental and human samples, indicating potential environmental and health risks. Previous toxicology researches suggested that TPP was a potential endocrine disruptor, which was harmful to thyroid function . However, the potential thyroid hormone-disrupting effects of TPP were scarce and the mechanism remained unclear. Additional studies are required to further explore the toxicity pathway of thyroid dysfunction exposed by TPP.
Strategy
This study retrieved the data from comparative toxicogenomics database (CTD) and screened out the core gene. Molecular dynamics (MD) analysis was used to explore configuration changes and confirm molecular initiating event (MIE). The transcriptomic analysis was further utilized to supplement the relationships between MIEs and key events (KEs) of AOP.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1158 | Increased, Hepatic thyroid hormone uptake/transport | Increased, Hepatic thyroid hormone uptake/transport |
| KE | 1505 | Cell cycle, disrupted | Cell cycle, disrupted |
| KE | 1829 | Altered, Thyroid hormone-dependent gene expression | Altered, TH-dependent gene expression |
| KE | 1844 | systemic inflammatory response syndrome | SIRS |
| KE | 1555 | Increase cell proliferation | Increase cell proliferation |
| KE | 1262 | Apoptosis | Apoptosis |
Relationships Between Two Key Events (Including MIEs and AOs)
Network View
Prototypical Stressors
| Name |
|---|
| Triphenyl phosphate |
Life Stage Applicability
Taxonomic Applicability
Sex Applicability
Overall Assessment of the AOP
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|