
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 405
Title
Organo-Phosphate Chemicals induced inhibition of AChE leading to impaired cognitive function
Short name
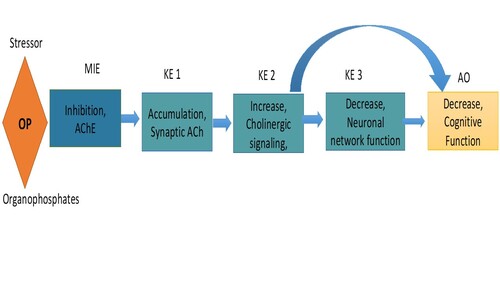
Graphical Representation
Point of Contact
Contributors
- SAROJ AMAR
- Kurt A. Gust
- Brendan Ferreri-Hanberry
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Acetylcholinesterase (AchE) Inhibition | April 29, 2020 17:21 |
| Acetylcholine accumulation in synapses | June 26, 2020 13:06 |
| Increased Cholinergic Signaling | December 20, 2019 17:32 |
| Decrease of neuronal network function | May 28, 2018 11:36 |
| Cognitive Function, Decreased | August 09, 2018 11:55 |
| AchE Inhibition leads to ACh Synaptic Accumulation | September 10, 2023 19:16 |
| Neuronal network function, Decreased leads to Cognitive Function, Decreased | August 27, 2021 12:24 |
| ACh Synaptic Accumulation leads to Increased Cholinergic Signaling | December 20, 2019 09:16 |
| Increased Cholinergic Signaling leads to Cognitive Function, Decreased | September 01, 2021 13:47 |
| Increased Cholinergic Signaling leads to Neuronal network function, Decreased | September 01, 2021 18:27 |
| Organophosphates | November 29, 2016 21:20 |
| Paraoxon | June 24, 2021 18:47 |
| Methyl parathion | June 24, 2021 18:49 |
| Ethyl Parathion | June 24, 2021 18:52 |
| N-methyl Carbamates | October 07, 2019 14:19 |
Abstract
Organophosphate compounds (OP) are extensively used as pesticides/insecticides including paraoxon, ethyl-parathion, methyl-parathion, but have also been developed as warfare nerve agents such as soman, sarin, tabun and others. OP-induced cognitive deficits were observe not only among farm worker but also among environmentally exposed individual (Corral SA et al., 2017). The present adverse outcome pathways (AOP) describes the risk associated with a molecular initiating event (MIE) characteristic of OP exposure in which inhibition of acetylcholinesterase (AChE) activity causes a series of key events (KEs) that ultimately manifest as the adverse outcome (AO) of cognitive defects. The MIE of inhibited AChE triggers the KEs: accumulation of synaptic AChE (KE 1), increase of cholinergic signaling (KE 2), decrease of neuronal network function (KE 3), and decrease of cognitive function as an adverse outcome. The content of this AOP draws upon content from other AOPs in the AOPwiki page and has expanded interpretation in the interconnected network of AOPs linking the MIE of AChE inhibition to other AOs, including acute mortality. The common threads between this and the other AOPs include common KEs of acetylcholine (ACh) accumulation at the synapses (KE 1), which results in (KE 2) excessive signaling from cholinergic neurons on a broad range of tissues throughout the body. The MIE is engaged when OP binds to AChE causing an irreversible phosphorylation status of the enzyme. AChE is an enzyme responsible for controlling the level of the excitatory neurotransmitter, ACh, at neural synapses and neuromuscular junctions. AChE negatively regulates ACh via hydrolysis to acetic acid and choline (Wilson 2010). Inhibition of AChE (MIE) prevents degradation of ACh, which leads to (KE1) ACh accumulation at neural synapses and neuromuscular junctions in the central and peripheral nervous systems. (https://aopwiki.org/aops/16, https://aopwiki.org/aops/312, Soreq and Seidman, 2001; Lushington 2006, Prado, 2017). ACh is generated in presynaptic neurons and released into the synaptic cleft where it can bind to both pre- and postsynaptic receptors. ACh availability is decreased when this neurotransmitter is degraded by AChE and by negative feedback loops controlled by muscarinic M2 receptors on the presynaptic neuron within the synapse (Soreq and Seidman, 2001). Affinity of ACh for metabotropic muscarinic receptors (mAChRs) and ionotropic nicotinic receptors (nAChRs), as well as rates of synaptic clearance (mediated through AChE activity) and local concentration of ACh in and outside the synapse, is critical for the control and specificity of cholinergic signaling (KE 2) (Sarter M et al., 2009, Picciotto MR, et al., 2012). Excessive accumulation of ACh at neural synapses (KE 1) and at neural-muscular junctions results in increased cholinergic signaling (KE 2) (AOP 16, https://aopwiki.org/aops/16). Endogenously released ACh regulates cognitive functions (AO), by acting as a neuromodulator and/or acting as a direct transmitter via nicotinic and muscarinic receptors in CNS by cholinergic signaling (KE 2) (Luchicchi A et al., 2014), which is evidence of direct relationship between KE2 and AO. The ability of a neuron to communicate is based on neural network formation (KE 3) that relies on functional synapse establishment by cholinergic neurons (KE 2) (Colón-Ramos, 2009) this is evidence that KE 2 leads to KE3. Muscarinic cholinergic activity influences sensory processing by facilitating or depressing neuronal responses to specific stimuli, and by modulating connection strength and neural synchronization: this results in the fine-tuning of cellular and network properties of neurons during developmental processes, the execution of attention tasks and perceptual learning (Colangelo C et al., 2019, Groleau et al., 2015). Damage or destruction of neurons during development when they are in the process of synapse formation, integration, and formation of neural networks, disrupts the organization and function of these networks (KE3), thereby setting the stage for subsequent impairment of learning and memory as sign of cognitive defects (AO), thus evident that KE 3 leads to AO. (AOP 13, https://aopwiki.org/aops/13). Therefore, if exposure to OP occurs during neuronal differentiation and synaptogenesis processes, there is potential to initiate KE3, functional neuronal network damage, leading to the cognitive defects AO. Thus, this AOP provides a needed link to between chronic AChE inhibition and detrimental long-term impacts on cognitive function, which is relevant for understanding the impacts of long-term environmental and occupational OP pesticide exposures.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 12 | Acetylcholinesterase (AchE) Inhibition | AchE Inhibition |
| KE | 10 | Acetylcholine accumulation in synapses | ACh Synaptic Accumulation |
| KE | 39 | Increased Cholinergic Signaling | Increased Cholinergic Signaling |
| KE | 386 | Decrease of neuronal network function | Neuronal network function, Decreased |
| AO | 402 | Cognitive Function, Decreased | Cognitive Function, Decreased |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| AchE Inhibition leads to ACh Synaptic Accumulation | adjacent | High | Moderate |
| ACh Synaptic Accumulation leads to Increased Cholinergic Signaling | adjacent | High | Low |
| Increased Cholinergic Signaling leads to Neuronal network function, Decreased | adjacent | Moderate | Moderate |
| Neuronal network function, Decreased leads to Cognitive Function, Decreased | non-adjacent | Moderate | Moderate |
| Increased Cholinergic Signaling leads to Cognitive Function, Decreased | non-adjacent | Moderate | Moderate |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Nursing Child | High |
| Adults | Moderate |
| All life stages | Moderate |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | Moderate |
| Male | High |
Overall Assessment of the AOP
Acute and chronic exposure to organophosphate, act by inhibiting cholinesterases which is widely associated with cognitive and motor impairments that can be observed even several months post-intoxication (Roldán-Tapia et al., 2006). Previous studies reported that the cognitive deficits were frequently observed not only in agricultural workers that directly manipulate pesticides but also individuals indirectly exposed during environmental applications (Corral SA et al., 2017). The multifactorial nature of pesticide-related cognitive impairment is consistent with experimental results showing distinct roles for pesticide exposure, duration of exposure and age of exposed individuals. The extent to which each of the aforementioned factors contributes to cognitive deficits remains under-explored (Aloizou AM et al., 2020). AChE inhibition is initiated by electrostatic interaction at the anionic site of the enzyme and binding with the serine hydroxyl radicals at the esteratic site of AChE (Wilson 2010; Fukuto 1990). This irreversible binding between AChE and OP pesticides is due to phosphorylation of enzyme. ACh is crucial for a number of important task including normal function of CNS, learning and memory, cognitive function as well as emotional and behavioral functions (Kilgard and Merzenich, 1998), reward (Leslie et al., 2013) and attention (Klinkenberg et al., 2011; Picciotto et al., 2012). This AChE inhibition (MIE) leads directly to the KE1 where ACh has unmitigated accumulation in neuronal synapses. This KER is directly supported the given observations demonstrating that AChE catalyzes degradation of ACh into choline and acetate (Wilson 2010). For goal-directed behavior, an appropriate levels of ACh are require to stimulate relevant sensory information (Sarter et al., 2009). An increase in cholinergic tone (KE 2) appears to be sufficient to induce depression-like symptoms in humans (Piccioto et al., 2012) and increasing ACh levels (KE 1) increases symptoms of depression (Overstreet, 1993). ACh can induce heterogeneous effects in different brain areas that appear to have opposite behavioral consequences depending on the specific anatomical location (Piccioto et al., 2012). Regardless, the predominant response of excessive accumulation of ACh at neural synapses (KE 1) and at neural-muscular junctions results in increased cholinergic signaling (KE 2) (AOP 16, https://aopwiki.org/aops/16). The ability of Ach (KE1) to induce synaptic plasticity through actions on pre- and post-synaptic nAChRs and mAChRs is likely to modulate learning and memory – symptoms of cognitive defects (AO), including memory of stressful events (Piccioto et al., 2012, Nijholt et al., 2004), and a role for ACh in regulation of hippocampal excitability (responsible for cognitive task) through presynaptic release of glutamate and GABA has also been well-characterized (Alkondon et al., 1997). It is clear that ACh (KE1), released from the cholinergic inputs (KE 2) of the basal forebrain, striatal, and the pontomesencephalic (PM) areas, plays an important role in supporting neurocognitive (AO) and motivational functions of the prefrontal cortical, hippocampal, and ventral tegmental projections to the striatum (Berman JA et al., 2007, Cragg, 2006; Sarter et al., 2005; Wonnacott et al., 2005). The integrated-information processing and communication role of neurons is dependent to neural network formation (KE 3) that count on functional synapse formation by cholinergic neurons (KE 2) (Colón-Ramos, 2009) which is influential in KE 2 leading to KE3. Still, the most difficult remaining gap for neuroscientific investigation of OP effects center on connecting the impacts on neuronal network function (KE3) to (AO) cognition, including learning and memory. It is unknown which alterations in neuronal circuits are essential to change motor behavior as per learning and memory test record (Mayford et al., 2012), meaning that there is no clear understanding about how this KE and AO are connected (AOP 13, https://aopwiki.org/aops/13). It’s difficult to establish the relationship between alteration of neural network function and cognitive deficits due to complexity of synaptic interactions in even the simplest brain circuit. Linking of neurophysiological assessments to learning and memory processes have been made across simple monosynaptic connections and largely focused on the hippocampus (AOP 13, https://aopwiki.org/aops/13). There is very limited information on the degree of quantitative change in neural network function (KE 3) required to alter cognitive behaviors (AO). This is a result of the diversity of methods for measuring both neuronal network function and learning and memory deficits, which hamper cross-study analysis (AOP 13, https://aopwiki.org/aops/13). In humans, the hippocampus is involved in recollection of an event’s rich spatial-temporal contexts and distinguished from simple semantic memory, which is memory of a list of facts (https://aopwiki.org/events/402, Burgess et al., 2002).
Domain of Applicability
Life Stage Applicability
The key molecular target is the AChE enzyme, which appears to be available in all life stages of different species (https://aopwiki.org/aops/16).
|
Life Stage |
Evidence |
|
Child |
High |
|
Adult |
Moderate |
Taxonomic Applicability
Though AChE enzyme can be traced in all vertebrate and invertebrate but the activity among taxa differs (https://aopwiki.org/aops/16)
Sex Applicability
Study with 44 children for scanning the OP pesticide exposure risk witnessed differences for sex of the child, with male levels higher than female levels (Loewenherz C et al., 1997).
Essentiality of the Key Events
- MIE: Inhibition, AChE: AChE is a serine hydrolase that terminates the action of the neurotransmitter ACh by hydrolyzing it into acetic acid and choline. (McHardy SF et al., 2017) The AChE inhibitors (pesticides) bind to the enzyme and interfere with the breakdown of ACh, leading to the deposition of ACh (KE1) in the nerve synapses and causing disrupted neurotransmission (Thapa S et al.,2017, https://aopwiki.org/aops/16, https://aopwiki.org/aops/312). Previous studies with vertebrate and invertebrate validate the dependence of AChE activity to the dose of OP and increasing inhibition of AChE in dose dependent manner with OP as reported in fish, birds, nematodes, rodents and mollusk (https://aopwiki.org/events/12).
- Key Event 1: Accumulation, synaptic ACh: ACh is stored in nerve endings at cholinergic synapses in the central and peripheral nervous systems (Soreq and Seidman, 2001; Lushington 2006). OP anticholinesterases potentially have a mechanism of toxicity in common, that is, phosphorylation of AChE causing accumulation of ACh (KE 1), overstimulation of cholinergic receptors, and consequent clinical signs of cholinergic toxicity. However, some OP pesticides appear capable of altering noncholinergic neurochemical processes. These additional actions may contribute to qualitative and quantitative differences in toxicity sometimes noted in the presence of similar levels of AChE inhibition induced by different OP pesticides (Pope CN., 1999). Epidemiological studies have reported statistically significant correlations between prenatal subacute exposures to OP insecticides and neurological deficits that range from cognitive impairments to tremors in childhood (Burke RD et al., 2017). Excessive accumulation of ACh at neural synapses (KE 1) and at neural-muscular junctions results in increased cholinergic signaling (KE 2) (AOP 16, https://aopwiki.org/aops/16,AOP 312 https://aopwiki.org/aops/312).
- Key Event 2: Increase, Cholinergic signaling: Acetylcholine is a neurotransmitter and neuromodulator that can exert either excitatory or inhibitory effects, depending on the receptor it binds to. ACh facilitates central and peripheral functions as well as somatic and autonomic functions. Excessive accumulation of acetylcholine at neural synapses and at neural-muscular junctions results in increased cholinergic signaling (https://aopwiki.org/relationships/456). The complexity of CNS cholinergic circuits and signaling mechanisms produces a system in which origins and end results may be easier to conclude than intervening intermediate steps. It is well reported that ACh, releases from the cholinergic inputs of the basal forebrain and striatal and from pontomesencephalic (PM) areas is supporting neurocognitive and motivational functions (Cragg, 2006; Sarter et al., 2005). Endogenously released ACh regulates cognitive functions (AO), by acting as a neuromodulator or acting as a direct transmitter via nicotinic and muscarinic receptors in CNS by cholinergic signaling (KE 2) (Luchicchi A et al., 2014), which is evidence of direct relationship between KE2 and AO. The capability of a neuron to communicate is centered on neural network formation (KE 3) that depend on functional synapse formation through cholinergic neurons (KE 2) (Colón-Ramos, 2009) this is evidence that KE 2 leads to KE3. The capacity of a neuron to communicate is dependent to neural network formation (KE 3) that depend on functional synapse formation by cholinergic neurons (KE 2) (Colón-Ramos, 2009) this is evidence that KE 2 leads to KE3.
- Key Event 3 Decrease, neuronal network function: Exposure to the potential developmental toxicants and OP during neuronal differentiation and synaptogenesis will increase the risk of functional neuronal network damage (KE3) leading to cognitive defects (AO), (https://aopwiki.org/aops/13). Moreover, it is well accepted that alterations in synaptic transmission and plasticity contribute to (AO) deficits in cognitive function (AOP 13, https://aopwiki.org/aops/13). Damage or destruction of neurons during development when they are in the process of synapse formation, integration, and formation of neural networks, disrupts the organization and function of these networks (KE3), thereby setting the stage for subsequent impairment of learning and memory as sign of cognitive defects (AO), thus evident that KE 3 leads to AO. (AOP 13, https://aopwiki.org/aops/13) Neuronal network formation and function are established via the process of synaptogenesis. The initial period of synaptogenesis is important for the formation of the basic circuitry of the nervous system, though neurons can form new synapses throughout life (Rodier, 1995). Proper neuronal communication is dependent to brain electrical activity and synapse formation. The main roles of synapses are responsible for the regulation of intercellular communication in nervous system as well as the information flow among neural networks. The connectivity and functionality of neural networks depends on where and when synapses are formed (Colón-Ramos, 2009). So, the decreased synapse formation during the process of synaptogenesis is vital and resulting to the decrease of neural network formation.
Evidence Assessment
- The overall weight of evidence supporting the indirect relationship between AChE inhibition and cognitive defects is very strong and there are many physiological activities associated with ACh neurotransmission that are plausibly linked with organism survival.
- Based on the current information assembled for this AOP, the essentiality of the key events downstream of ACh accumulation is less clear. While there are several key events that correspond with well-known symptoms of AChE inhibition, it is presently unclear which of these the major driver of cognitive defects are across different species. Given the abundance of literature on ACh signaling and adverse effects associated with AChE inhibition, this is an area of the AOP that warrants further development.
- Biological Plausibility
- ACh is a critical neurotransmitter localized to neuronal synapses. Biological plausibility to support the relationship between AChE inhibition and accumulation of ACh is rooted in evidence demonstrating that AChE catalyzes degradation of ACh into choline and acetate. Therefore, inhibition of the AChE leads to ACh accumulation.
- Biological plausibility for ACh accumulation at the synapse leading to nervous system dysfunction is rooted in the well-established understanding of ACh’s function as a neurotransmitter and neuromodulator. By acting upstream of a range of cellular and physiological functions, it is biologically plausible that accumulation of ACh at neurological synapses will lead to systemic dysfunctions, which are often freely evident and assessable in clinical and research settings.
- Neuronal network formation and function are established via the process of synaptogenesis. The initial period of synaptogenesis is vital for the formation of the elementary circuitry of the nervous system, while neurons can form new synapses throughout their life (Rodier, 1995). The brain electrical activity dependence on synapse formation is critical for proper neuronal communication (https://aopwiki.org/wiki/index.php/Relationship:358).
- Learning-induced enhancement in neuronal excitability, a measurement of neural network function, has also been shown in hippocampal neurons following classical conditioning in several experimental approaches (Saar and Barkai, 2003). Previous study with Morris water maze (MWM) test to investigate spatial learning and memory in laboratory rats also indicated that the interruption between neuronal networks rather than the brain damage of certain regions is accountable for the impairment of MWM presentation. Functional integrated neural networks that involve the coordination action of different brain regions are consequently important for spatial learning and MWM performance (https://aopwiki.org/wiki/index.php/Relationship:359, D'Hooge and De Deyn, 2001).
- Moreover, it is well accepted that alterations in synaptic transmission and plasticity contribute to deficits in cognitive function
- Concordance of dose-response relationships:
- Striatal AChE activity and extracellular ACh levels were measured in rats intracerebrally perfused with paraoxon (0, 0.03, 0.1, 1, 10 or 100 μM, 1.5 μl/min for 45 min) (https://aopwiki.org/relationships/11, Ray, 2009). In that study, ACh was below the limit of detection at the low dose of paraoxon (0.1 uM), but was transiently elevated (0.5–1.5 hr) with 10 μM paraoxon. Concentration-dependent AchE inhibition was noted but reached a plateau of about 70% at 1 μM and higher concentrations (Ray, 2009). The association among AChE inhibition and ACh accumulation at the synapse can be detected within 30 minutes after application of AChE inhibitor (Ray, 2009).
- The main proof of evidence comes from in vivo studies in rodents. Though, Colón-Ramos (2009) has recently showed that the initial developmental events that during the course of synaptogenesis in invertebrates, indicating the significance of this process in neural network formation and function.
- Because the adult hippocampus is involved in learning and memory, it is a brain region of remarkable plasticity (Johnston et al., 2009). Use-dependent synaptic plasticity is critical during brain development for synaptogenesis and fine-tuning of synaptic connectivity (Johnston et al., 2009).
- Temporal concordance among the key events and adverse effect:
- Strong evidence based on measured AChE inhibition and statistically derived acute endpoints (e.g., LC/LD50) demonstrate a correlation of increase in enzyme inhibition and decrease cognitive function. The literature includes many studies linking increases in ACh in brain tissues after exposure to an OP or carbamate pesticide with increased AChE inhibition in various taxa. As previous studies with crustacea (Reddy et al., 1990); tadpoles (Nayeemunnisa and Yasmeen, 1986); fish (Rao and Rao 1984; Verma et al., 1981); birds (Kobayashi et al., 1983); and rodents (Kobayashi et al., 1988) revealed.
- ACh is a neurotransmitter and neuromodulator that can exert either excitatory or inhibitory effects, depending on the receptor it binds to (Picciotto, 2012). ACh mediates central and peripheral functions, including somatic and autonomic functions (Picciotto, 2012). Excessive accumulation of ACh at neural synapses and at neural-muscular junctions results in increased cholinergic signaling. Clinical manifestations of an acute exposure of humans to OP insecticides include a well-defined cholinergic crisis that develops as a result of the irreversible inhibition of AChE, the enzyme that hydrolyzes the neurotransmitter ACh (Burke RD et al., 2017).
- ACh is considered to be the most important neurotransmitter involved in the regulation of cognitive functions. Once releasing from the presynaptic neuron, ACh accumulated into the synaptic cleft, followed by binding to the ACh receptors on the postsynaptic membrane, and the signal from the nerve was transmitted during the process (Schliebs R and Arendt T, 2011, Gold P. E., 2003, Wang XC, 2018). Study showed the cholinergic overstimulation once pigs exposed to dichlorvos (the AChE inhibitor), symptoms may include miosis, cyanosis, tremor, excess secretions and fasciculations. Estimation of AChE levels established that dichlorvos treatment inhibited AChE activity. (Cui, 2013).
- There is strong empirical evidence linking the key events, beginning with the molecular initiating event; AChE inhibition, followed by an increase in the ACh at synapses of muscarinic and nicotinic receptors, and subsequent physiological and biochemical response resulting in cholinergic activity (https://aopwiki.org/aops/16) , Picciotto MR et al, 2012.
- Consistency:
- (https://aopwiki.org/relationships/11) Previous study showed female ICR (Institute of Cancer Research) mice exposed to either fenobucarb or propoxur, reported a major increase in ACh in brain tissue 10 minutes after injection, simultaneously major elevation in AChE inhibition (Kobayashi et al., 1985). Sub lethal exposure to methyl parathion conclude that AChE levels in brain tissue in fish (Oreochromis mossambicus) were highly inhibited during 12-48 hrs, with inhibition increasing from 36-62% as in comparison to controls over the time elapse (Rao and Rao, 1984). The researchers found a significant increase in ACh at all-time courses measured (12-48hr) with ACh levels increasing from 33-83% as compared to controls over the same time span (Rao and Rao, 1984). A study of quail (Coturnix japonica) exposed to lethal concentrations of two OP pesticides (i.e., DDVP or fenitrothion), found significant increases in total and free ACh, and major inhibition of AChE in-comparison to controls (Kobayashi et al., 1983). Measurements (in vitro) of AChE inhibition, ACh and electrophysiological responses on the pedal ganglion of the gastropod Aplysia californica, were found to be dose-dependent, with increase in dose resulting in increased AChE inhibition, increased levels of ACh, and a decrease in the electrophysiological response (Oyama et al., 1989). Wister rats injected with a sublethal concentration of dichlorvos found a significant decrease in AChE activity, increased ACh concentrations, and enhanced contractile responses in jejunum muscle. At sublethal concentrations (56% of the LD50), researchers found a significant (18%) increase in the amount of ACh in brain tissue of Charles River rats exposed to disulfoton for 3 days and resulted in AChE inhibition of 68% with respect to controls (Stavinoha et al., 1969). An acute sublethal exposure of chlorpyrifos to Sprague-Dawley rats found significant dose and time related effects including increased inhibition of AChE, increased levels of ACh, and significant influences to motor activity (Karanth et al., 2006). Tadpoles of 20 days were treated with single sub lethal dose of the methyl parathion for 24 hrs and analysis of brain tissue found a significant inhibition in AChE activity and a concurrent increase in ACh levels, as compared to controls (https://aopwiki.org/relationships/11, Nayeemunnisa and Yasmeen 1986). Study of fourth instar Ailanthus silkworm exposed to malathion for 5 days found increased mortality, decreased AChE, and increases in ACh as compared to controls (Pant and Katiyar 1983).
- The relationship between excess ACh at synapses and nervous system dysfunction has been reviewed in Molecular Cell Biology, 4th Edition (https://aopwiki.org/relationships/456, Lodish, 2000). ACh is a neurotransmitter in most vertebrate and invertebrate species, but the mechanism of activity may differ. For example in insects, ACh acts as a neurotransmitter between sensory neurons and the central nervous system but glutamate acts as a neurotransmitter between motor neurons and skeletal muscles (https://aopwiki.org/relationships/456, Stenersen, 2004). 8-14 weeks old male quail were exposed to a single dose of either dichlorvos or fenitrothion by subcutaneous injection and brain tissue showed an 80% reduction of AChE, and a simultaneous major increase in ACh as compared to controls. With maximum doses, mortality was headed by symptoms including vigorous tremors, lacrimation, salivation, ataxia, and respiratory distress (Kobayashi, 1985). Previous study with male and female starlings of three age groups (5 days to >1 year) showed that the LD50 for nestlings was around half of the LD50 for adult birds exposed to dicrotophos. Simultaneously all birds exposed, showing impaired coordination and tremors. AChE inhibition increased in dose dependent manner for all three-age groups. There is no sex differences in LC50 or AChE inhibition were reported (Grue and Shipley, 1984). Asian stinging catfish (Heteropneustes fossilis) exposed for 40 days to sublethal concentrations of oxydemeton-methyl, had a >71% inhibition of AChe in the brain and a concurrent increase of ACh in brain (>200%) and muscles (>188%), with fish displaying violent body movements (tremors) followed by loss of equilibrium (Verma 1981).
- Single injection of methylmercury (8 mg/kg by gavage) at gestational day 15. Offsprings examined at the age of 14, 21, and 60 days showed a reduction in the number of muscarinic receptors at 14 and 21 days and a decline in avoidance latency at 60 days, demonstrating learning and memory deficits (Zanoli et al., 1994), (https://aopwiki.org/relationships/359, Rice, 1992).
- Uncertainties, inconsistencies, and data gaps:
- No known qualitative inconsistencies or uncertainties associated between AChE inhibition and ACh accumulation at the synapse as well as ACh accumulation at the synapse to cholinergic signaling.
- The exact mechanism by which a change in cell number, reduced dendritric arborization and synaptogenesis may lead to decreased neuronal network function has not been fully elucidated.
The direct relationship of alterations in neural network function and specific cognitive deficits is difficult to ascertain given the many forms that learning and memory can take and the complexity of synaptic interactions in even the simplest brain circuit. Linking of neurophysiological assessments to learning and memory processes have been made across simple monosynaptic connections and largely focused on the hippocampus. Changes in synaptic function have been noticed even in the lack of any behavioral losses. (https://aopwiki.org/relationships/359).
Known Modulating Factors
Quantitative Understanding
Exposure to organophosphate (OP) pesticides, which inhibit acetylcholinesterase, increases the risk of neurological disorders (Voorhees, J.R. et al., 2019). Recent study suggested that organophosphate pesticides may cause cognitive impairment. Mild cognitive impairment are dominating with symptoms like decreased attention or vigilance, narrowed information processing speed and memory impairment (Zhang HY et al., 2021). Mild cognitive impairment is largely being ignored for a long periods of time. Though, it might have huge impact on patients' life and work, and even progress to irreversible neurodegenerative disorder (Zhang HY et al., 2021). On cognitive tasks of learning and memory, male TgF344-AD rats displayed chlorpyrifos- an OP pesticide dependent deficits that were not seen in WT males or females of either genotype (Voorhees, J.R. et al., 2019). Previous study suggested that lower cognitive performances with huge decline in performances in vine workers is linked with pesticides exposure (Audrey Blanc-Lapierre et al., 2013). Thus this AOP attempt to establish the quantitative relationship between organophosphate pesticides and cognitive defects. Understanding the underlying mechanisms, this AOP can provide new means to avoid or neutralize the pesticide exposure risk. The stepwise relationships between consecutive key events is as follow. Existing key event relationship number 11, define inhibition of acetylcholinesterase (MIE) leads to synaptic accumulation of acetylcholine (KE1). Simultaneously key event relationship number 456 explained synaptic accumulation of acetylcholine (KE1) increases cholinergic signaling (KE2). Although the exact mechanism for increase cholinergic signaling lead to decreased neuronal network function (KE 3) has not been fully elucidated. But it’s well-known that the ability of a neuron to communicate is based on neural network formation that relies on functional synapse establishment by cholinergic neuron (Colón-Ramos, 2009). The direct relationship of alterations in neural network function (KE 3) and specific cognitive deficits (AO) is difficult to ascertain given the many forms that learning and memory can take and the complexity of synaptic interactions in even the simplest brain circuit (https://aopwiki.org/relationships/359). Though the AOP 13 advocate that, damage of neurons during development when they are in the process of formation of neural networks (KE3), setting the stage for subsequent impairment of learning and memory as sign of cognitive defects (AO). Thus above evidence supporting the development of this AOP.
Considerations for Potential Applications of the AOP (optional)
Although the present AOP may require supplementary conditions to fully establish the neurotoxicity potential of OP pesticide and their mode of action. This AOP is an attempt to establish the mechanism of organophosphorus (OP) pesticides induced cognitive defects via cholinergic signaling. It can also be applied to risk assessment in predictive modeling of OP pesticide toxicity.
References
Agency for Toxic Substances and Disease Registry (ATSDR). 2001. Toxicological profile for methyl parathion. Update. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service.
Alkondon M, Pereira EF, Barbosa CT, Albuquerque EX. Neuronal nicotinic acetylcholine receptor activation modulates gamma-aminobutyric acid release from CA1 neurons of rat hippocampal slices. J Pharmacol Exp Ther. 1997; 283:1396–1411. [PubMed: 9400016]
Aloizou AM, Siokas V, Vogiatzi C, Peristeri E, Docea AO, Petrakis D, et al. Pesticides, cognitive functions and dementia: A review. Toxicology Letters. 2020; 326:31-51.
Audrey Blanc-Lapierre, Ghislaine Bouvier, Anne Gruber, Karen Leffondré, Pierre Lebailly, Colette Fabrigoule, Isabelle Baldi, Cognitive Disorders and Occupational Exposure to Organophosphates: Results from the PHYTONER Study, American Journal of Epidemiology, Volume 177, Issue 10, 15 May 2013, Pages 1086–1096, https://doi.org/10.1093/aje/kws346
Berman JA, Talmage DA, Role LW. Cholinergic circuits and signaling in the pathophysiology of schizophrenia. Int Rev Neurobiol. 2007; 78:193-223. doi:10.1016/S0074-7742(06)78007-2
Burgess N (2002). The hippocampus, space, and viewpoints in episodic memory. Q J Exp Psychol A 55:1057-1080.
Burke RD, Todd SW, Lumsden E, et al. Developmental neurotoxicity of the organophosphorus insecticide chlorpyrifos: from clinical findings to preclinical models and potential mechanisms. J Neurochem. 2017; 142 Suppl 2(Suppl 2):162-177. doi:10.1111/jnc.14077
Centers for Disease Control and Prevention (CDC) 2009,https://www.cdc.gov/biomonitoring/Methyl_Eethyl_Parathion_BiomonitoringSummary.html
Colangelo C, Shichkova P, Keller D, Markram H, Ramaswamy S. Cellular, Synaptic and Network Effects of Acetylcholine in the Neocortex. Front Neural Circuits. 2019; 13:24. Published 2019 Apr 12. doi:10.3389/fncir.2019.00024
Colón-Ramos DA. (2009). Synapse formation in developing neural circuits. Curr Top Dev Biol. 87: 53-79
Corral SA, de Angel V, Salas N, Zúñiga-Venegas L, Gaspar PA, Pancetti F. Cognitive impairment in agricultural workers and nearby residents exposed to pesticides in the Coquimbo Region of Chile. Neurotoxicology and Teratology. 2017; 62:13-9..
Cragg SJ. Meaningful silences: How dopamine listens to the ACh pause. Trends Neurosci. 2006; 29:125–131
Cui J, Li CS, He XH, Song YG. Protective effects of penehyclidine hydrochloride on acute lung injury caused by severe dichlorvos poisoning in swine. Chin Med J (Engl). 2013; 126(24):4764-70.
Deshpande LS, Phillips K, Huang B, DeLorenzo RJ (September 2014). "Chronic behavioral and cognitive deficits in a rat survival model of paraoxon toxicity". Neurotoxicology. 44: 352–7.
D'Hooge R, De Deyn PP. (2001) Applications of the Morris water maze in the study of learning and memory. Brain Res Brain Res Rev. 36: 60-90
Farizatto KLG, Bahr BA. Paraoxon: An Anticholinesterase That Triggers an Excitotoxic Cascade of Oxidative Stress, Adhesion Responses, and Synaptic Compromise. Eur Sci J. 2017; 13:29-37. doi:10.19044/esj.2017.c1p4
Fukuto, TR. 1990. Mechanism of action of organophosphorus and carbamate insecticides. Environ Health Perspect. 87:245-254.
Furlong CE. Genetic variability in the cytochrome P450-paraoxonase 1 (PON1) pathway for detoxication of organophosphorus compounds. J Biochem Mol Toxicol. 2007; 21(4):197-205. doi: 10.1002/jbt.20181. Erratum in: J Biochem Mol Toxicol. 2007; 21(5):323. PMID: 17936934.
Garcia S , Abu-Qare A, Meeker-O'Connell W, Borton A & Abou-Donia M (2003) Methyl Parathion: A Review of Health Effects, Journal of Toxicology and Environmental Health Part B: Critical Reviews, 6:2, 185-210, DOI: 10.1080/10937400306471.
Gold PE, “Acetylcholine: Cognitive and brain functions,” Neurobiology of Learning and Memory, vol. 80, no. 3, p. 177, 2003.
Groleau M., Kang J. I., Huppé-Gourgues F., Vaucher E. (2015). Distribution and effects of the muscarinic receptor subtypes in the primary visual cortex. Front. Synaptic Neurosci. 7:10. 10.3389/fnsyn.2015.00010.
Grue CE, Shipley BK. 1984. Sensitivity of nestling and adult starling to dicrotophos, an organophosphate pesticide. Environ Res 35:454–465.
Hill RH Jr, Alley CC, Ashley DL, Cline RE, Head SL, Needham LL, et al. Laboratory investigation of a poisoning epidemic in Sierra Leone. J Anal Toxicol 1990; 14(4):213-216.
Jaga K, Dharmani C. Methyl parathion: an organophosphate insecticide not quite forgotten. Rev Environ Health 2006; 21(1):57-67.
Johnson FO, Chambers JE, Nail CA, Givaruangsawat S, Carr RL. Developmental chlorpyrifos and methyl parathion exposure alters radial-arm maze performance in juvenile and adult rats. Toxicol Sci. 2009; 109(1):132-142. doi:10.1093/toxsci/kfp053.
Johnston MV, Ishida A, Ishida WN, Matsushita HB, Nishimura A, Tsuji M. (2009) Plasticity and injury in the developing brain. Brain Dev. 31:1-10.
Karanth, S., Liu, J., Mirajkar, N., and Pope, C. 2006. Effects of Acute Chlorpyrifos Exposure on In Vivo Acetylcholine Accumulation in Rat Striatum. Toxicol.Appl.Pharmacol. 216[1], 150-156.
Kilgard, M. P., and Merzenich, M. M. (1998). Cortical map reorganization enabled by nucleus basalis activity. Science 279, 1714–1718. doi: 10.1126/science.279. 5357.1714
Klinkenberg, I., Sambeth, A., and Blokland, A. (2011). Acetylcholine and attention. Behav. Brain Res. 221, 430–442. doi: 10.1016/j.bbr.2010.11.033
Kobayashi, H., Yuyama, A., Kudo, M., and Matsusaka, N. 1983. Effects of Organophosphorus Compounds, O, O-Dimethyl O-(2, 2-Dichlorovinyl) Phosphate (DDVP) and O, O-Dimethyl O-(3-Methyl 4-Nitrophenyl)Phosphorothioate (Fenitrothion), on Brain Acetylcholine Content and Acetylcholinesterase Activity in Japanese Quail. Toxicology 28[3], 219-227.
Kobayashi, H., Yuyama, A., Ohkawa, T., and Kajita, T. 1988. Effect of Single or Chronic Injection with a Carbamate, Propoxur, on the Brain Cholinergic System and Behavior of Mice. Jpn.J.Pharmacol. 47[1], 21-27.
Kobayashi,H., A. Yuyama, T. Kajita, K. Shimura, T. Ohkawa, and K. Satoh. 1985. Effects of Insecticidal Carbamates on Brain Acetylcholine Content, Acetylcholinesterase Activity and Behavior in Mice. Toxicol. Lett.29 (2-3): 153-159.
Laxmikant S. Deshpande, Dawn S. Carter, Kristin F. Phillips, Robert E. Blair, Robert J. DeLorenzo, Development of status epilepticus, sustained calcium elevations and neuronal injury in a rat survival model of lethal paraoxon intoxication, NeuroToxicology (2014), Volume 44,2014,Pages 17-26
Leslie, F. M., Mojica, C. Y., and Reynaga, D. D. (2013). Nicotinic receptors in addiction pathways. Mol. Pharmacol. 83, 753–758. doi: 10.1124/mol.112. 083659
Lodish H, Berk A, Zipursky SL, et al. Molecular Cell Biology. 4th edition. New York: W. H. Freeman; 2000. Section 21.4, Neurotransmitters, Synapses, and Impulse Transmission. Available from: https://www.ncbi.nlm.nih.gov/books/NBK21521/
Loewenherz C et al. Biological monitoring of organophosphorus pesticide exposure among children of agricultural workers in Central Washington State. Environ Health Persp 1997, 105:1344
Lores EM, Bradway DE, Moseman RF. Organophosphorus pesticide poisonings in humans: determination of residues and metabolites in tissues and urine. Arch Environ Health 1978; 33(5):270-276.
Luchicchi A, Bloem B, Viaña JNM, Mansvelder HD, Role LW. Illuminating the role of cholinergic signaling in circuits of attention and emotionally salient behaviors. Frontiers in Synaptic Neuroscience. 2014; 6(24).
Ludke JL, Hill EF, Dieter MP. 1975. Cholinesterase (ChE) response and related mortality among birds fed ChE inhibitors. Arch Environ ContamToxicol 3:1–21.
Lushington, G.H., J-X. Guo, and M.M. Hurley. 2006. Acetylcholinesterase: Molecular modeling with the whole toolkit. Curr. Topics Medic. Chem. 6: 57-73
Mayford M, Siegelbaum SA, Kandel ER., Synapses and memory storage. Cold Spring Harb Perspect Biol., 2012:4(6). pii: a005751
McHardy SF, Wang HL, McCowen SV, Valdez MC. Recent advances in acetylcholinesterase Inhibitors and Reactivators: an update on the patent literature (2012-2015). Expert Opin Ther Pat. 2017 Apr; 27(4):455-476.
Moser VC, Phillips PM, McDaniel KL. (2015) Assessment of biochemical and behavioral effects of carbaryl and methomyl in Brown-Norway rats from preweaning to senescence. Toxicology 331, 1-13.
Nayeemunnisa and Yasmeen, N. 1986. On the Presence of Calmodulin in the Brain of Control and Methyl Parathion-Exposed Developing Tadpoles of Frog, Rana cyanophlictis. Curr.Sci. (Bangalore) 55[11], 546-548.
Nijholt I, Farchi N, Kye M, Sklan EH, Shoham S, Verbeure B, Owen D, Hochner B, Spiess J, Soreq H, et al. Stress-induced alternative splicing of acetylcholinesterase results in enhanced fear memory and long-term potentiation. Mol Psychiatry. 2004; 9:174–183. [PubMed: 14581933]
Overstreet DH. The Flinders sensitive line rats: a genetic animal model of depression. Neurosci Biobehav Rev. 1993; 17:51–68. [PubMed: 8455816]
Oyama, Y., N. Hori, M.L. Evans, C.N. Allen, and D.O. Carpenter. 1989. Electrophysiological estimation of the actions of acetylcholinesterase inhibitors on acetylcholine receptor and cholinesterase in physically isolated Aplysia neurons. Br. J. Pharmacol. 96:573-582.
Pant R, and Katiyar S K. 1983. “Effect of Malathion and Acetylcholine on the Developing Larvae of Philosamia Ricini (Lepidoptera: Saturniidae).” Journal of Biosciences 5 (1): 89–95. https://doi.org/10.1007/BF02702598.
Picciotto MR, Higley MJ, Mineur YS. Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron. 2012; 76(1):116-129. doi:10.1016/j.neuron.2012.08.036
Pope CN. Organophosphorus pesticides: do they all have the same mechanism of toxicity? J Toxicol Environ Health B Crit Rev. 1999 Apr-Jun; 2(2):161-81. doi: 10.1080/109374099281205. PMID: 10230392
Prado, MAM, Marchot, P, Silman, I. Preface: Cholinergic Mechanisms. J Neurochem. 2017 Aug; 142 Suppl 2:3-6. doi: 10.1111/jnc.14027.
Rao, K S P and Rao, KV R. 1984. Impact of Methyl Parathion Toxicity and Eserine Inhibition on Acetylcholinesterase Activity in Tissues of the Teleost (Tilapia mossambica) - a Correlative Study. Toxicol.Lett. 22, 351-356.
Rattner, B.A. and Fairbrother A. 1991. Biological variability and the influence of stress on cholinesterase activity. In: P. Mineau (Ed.), Cholinesterase-inhibiting Insecticides: Their Impact on Wildlife and the Environment. (pp. 89-107). Elsevier, Amsterdam, The Netherlands.
Ray, A., J. Liu, S. Karanth, Y. Gao, S. Brimijoin, and C. Pope. 2009. “Cholinesterase inhibition and acetylcholine accumulation following intracerebral administration of paraoxon in rats.” Toxicology and Applied Pharmacology 236 (3): 341–47. https://doi.org/10.1016/j.taap.2009.02.022.
Reddy, M. S., Jayaprada, P., and Rao, K. V. R. 1990. Impact of Methylparathion and Malathion on Cholinergic and Non-Cholinergic Enzyme Systems of Penaeid Prawn, Metapenaeus monoceros. Biochem.Int. 22[4], 769-780.
Rice, D. C. (1992). "Effects of pre- plus postnatal exposure to methylmercury in the monkey on fixed interval and discrimination reversal performance." Neurotoxicology 13(2): 443-452.
Rodier PM. (1995) Developing brain as a target of toxicity. Environ. Health Perspect. 103: 73-76.
Roldan-Tapia, L., Nieto-Escamez, F. A., del Aguila, E. M., Laynez, F., Parron, T., and Sanchez-Santed, F. (2006). Neuropsychological Sequelae from Acute Poisoning and Long-Term Exposure to Carbamate and Organophosphate Pesticides. Neurotoxicology and Teratology 28 (6), 694–703. doi:10.1016/j.ntt.2006.07.004
Saar D, Barkai E. (2003) Long-term modifications in intrinsic neuronal properties and rule learning in rats. Eur J Neurosci. 17: 2727-2734.
Sarter M, Parikh V, Howe WM. Phasic acetylcholine release and the volume transmission hypothesis: time to move on. Nature Rev Neuroscience. 2009; 10:383–390.
Sarter M, Parikh V. Choline transporters, cholinergic transmission and cognition. Nat Rev Neurosci. 2005; 6:48–56
Schliebs R and Arendt T, “The cholinergic system in aging and neuronal degeneration,” Behavioural Brain Research, vol. 221, no. 2, pp. 555–563, 2011
Schuurmann G. 1992. Ecotoxicology and structure-activity studies of organophosphorus compounds. Rational Approaches to Structure, Activity, and Ecotoxicology of Agrochemicals, CRC Press, Boca Raton, FL, USA pp 485-541
Sogob MA, Vilanova E. 2002. Enzymes involved in the detoxification of organophosphorus, carbamate and pyrethroid insecticides through hydrolysis. Toxicol Lett 128:215-228.
Soreq, H. and S. Seidman. 2001. Acetylcholinesterase – new roles for an old actor. Nat. Rev. Neurosci. 2: 294-302.
Stavinoha, W. B., Ryan, L. C., and Smith, P. W. 1969. Biochemical Effects of an Organophosphorus Cholinesterase Inhibitor on the Rat Brain. Ann.N.Y.Acad.Sci. 160[1], 378-382.
Stenersen J. 2004. Specific enzyme inhibitors. Chemical Pesticides: Mode of action and toxicology, CRC Press, Boca Raton, FL, USA.
Straus, D. L., Schlenk, D., and Chambers, J. E. 2000. Hepatic microsomal desulfuration and dearylation of chlorpyrifos and parathion in fingerling channel catfish: Lack of effect from Aroclor 1254. Aquat. Toxicol. 50:141–149.
Taylor, P. 2011. Anticholinesterase agents. In: L.J. Brunton (Ed.), Goodman and Gilman’s The Pharmacological Basis of Therapeutics; 12th Edition. (pp. 255-276). McGraw Hill, New York, NY. (Accessed from the web: http://accessmedicine.com/resourceTOC.aspx?resourceID=651).
Thapa S, Lv M, Xu H. Acetylcholinesterase: A Primary Target for Drugs and Insecticides. Mini Rev Med Chem. 2017; 17(17):1665-1676. doi: 10.2174/1389557517666170120153930. PMID: 28117022
U.S. Environmental Protection Agency (U.S. EPA). R.E.D. Facts. Ethyl parathion. September 2000. EPA-738-FOO-009. Available at URL: https://www.epa.gov/oppsrrd1/REDs/factsheets/0155fct.pdfpdf iconexternal icon.1/24/13
Verma, S. R., Tonk, I. P., Gupta, A. K., and Dalela, R. C. 1981. In Vivo Enzymatic Alterations in Certain Tissues of Saccobranchus fossilis Following Exposure to Four Toxic Substances. Environ.Pollut.A. 26[2], 121-127.
Voorhees, J.R., Remy, M.T., Erickson, C.M. et al. Occupational-like organophosphate exposure disrupts microglia and accelerates deficits in a rat model of Alzheimer’s disease. npj Aging Mech Dis 5, 3 (2019). https://doi.org/10.1038/s41514-018-0033-3
Wang XC, Xu YM, Li HY, Wu CY, Xu TT, Luo NC, Zhang SJ, Wang Q, Quan SJ. Jiao-Tai-Wan Improves Cognitive Dysfunctions through Cholinergic Pathway in Scopolamine-Treated Mice. Biomed Res Int. 2018 Jun 27; 2018:3538763. doi: 10.1155/2018/3538763. PMID: 30050927; PMCID: PMC6040267.
Wilson BW. 2010. Cholinesterases. Hayes’ Handbook of Pesticide Toxicology, 3rd ed, Vol 2. Elsevier, Amsterdam, The Netherlands, pp 1457-1478.
Wonnacott S, Sidhpura N, Balfour DJ. Nicotine: From molecular mechanisms to behaviour. Curr Opin Pharmacol. 2005; 5:53–59.
Zanoli, P., et al. (1994). "Methyl mercury during late gestation affects temporarily the development of cortical muscarinic receptors in rat offspring." Pharmacol Toxicol 75(5): 261-264.
Zhang HY, Wang C, Li HS. [Effect of organophosphate pesticides poisoning on cognitive impairment]. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi. 2021 Apr 20; 39(4):313-316. Chinese. doi: 10.3760/cma.j.cn121094-20200325-00161. PMID: 33910299.