
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 430
Title
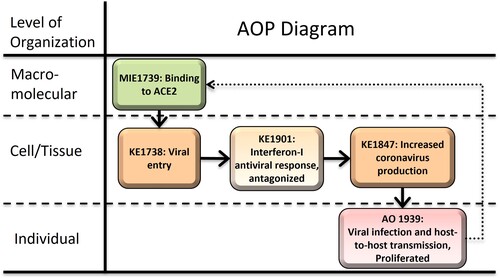
Binding of SARS-CoV-2 to ACE2 leads to viral infection proliferation
Short name
Graphical Representation
Point of Contact
Contributors
- Sally Mayasich
- Laure-Alix Clerbaux
- Maria Joao Amorim
- Arthur Author
Coaches
- Cinzia La Rocca
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.96 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Binding to ACE2 | August 30, 2023 20:36 |
| SARS-CoV-2 cell entry | April 04, 2023 07:39 |
| Interferon-I antiviral response, antagonized by SARS-CoV-2 | December 15, 2023 14:27 |
| Increased SARS-CoV-2 production | June 14, 2022 08:49 |
| Viral infection and host-to-host transmission, proliferated | December 12, 2023 14:48 |
| Binding to ACE2 leads to SARS-CoV-2 cell entry | February 07, 2023 23:24 |
| SARS-CoV-2 cell entry leads to IFN-I response, antagonized | December 12, 2023 15:15 |
| IFN-I response, antagonized leads to SARS-CoV-2 production | December 14, 2023 15:23 |
| SARS-CoV-2 production leads to Viral infection, proliferated | March 31, 2023 17:06 |
| SARS-CoV | March 01, 2020 10:42 |
| Sars-CoV-2 | September 09, 2022 05:09 |
| HCoV-NL63 | February 07, 2021 07:01 |
Abstract
Severe accute respiratory syndrome (SARS) and SARS-CoV-2 coronoviruses enter the cell through interaction with the ACE2 receptor. The first event upon cell entry after uncoating is the primary translation of the ORF1a and ORF1b genomic RNA to produce non-structural proteins (nsps). The nsps structural proteins, and accessory proteins, are encoded by 10 ORFs in the SARS-CoV-2 RNA genome. They may have multiple functions during viral replication as well as in evasion of the host innate immune response, thus augmenting viral replication and spread. The early innate immune system evasion proteins produced in the sub-genomic translation after viral genome replication and transcription within the infected cell suppress the Interferon-I antiviral response to increase viral load. Beyond potentially contributing to the severity of clinical symptoms and adverse disease outcome in individuals, increase in viral load can lead to proliferation from person-to-person and across species, also increasing the likelihood of mutations that result in more infective or virulant strains.
AOP Development Strategy
Context
This AOP was developed in the context of other COVID-19 AOPs through the work of a larger international effort to model the pathogenesis of COVID-19 using the AOP framework (the CIAO project, https://www.ciao-covid.net/about-us), initiated by the European Commission-Joint Research Centre (EC-JRC), and supported by the Society for the Advancement of Adverse Outcome Pathways (SAAOP). More than 80 scientists from 50 institutions contributed to the fifteen AOPs connected to the molecular initiating event (1739) SARS-CoV-2 binding to ACE2, and other COVID-19-related AOPs. AOP 430 serves as a hub of early key events leading to viral transmission (AO 1939) and the severe disease outcomes described in the networked COVID-19 AOPs.
Strategy
At the time the CIAO project was initiated in mid- to late 2020, the molecular initiating event of the SARS-CoV-2 spike protein binding to the host ACE2 receptor was known, and many of the severe disease outcomes required binding, cell entry and viral replication to spread to other tissues and organs from the point of infection. With the strategy of reuse of key events in the AOP-Wiki, AOP 430 was developed as a hub containing the MIE and early KEs of viral entry and replication. Through early literature and understanding of the virus, it was determined that evasion of the innate immune interferon-I (IFN-I) antiviral response is a key event to allow the virus to replicate and produce new virions to spread from cell to cell, and to generate a viral load for host-to-host transmission, including transmission across species (zoonotic spillover) and was included in the hub. The contributors are experts in their fields, including Dr. Young Jun Kim, who has conducted ACE2 binding research; Dr. Maria João Amorim, virologist; Dr. Laure-Alix Clerbaux, EU-JRC researcher and CIAO coordinator with other EU-JRC researchers Dr. Nikolaos Parissis and Dr. Alicia Paini; Dr. Penny Nymark, researcher with a background in genetics, cancer, toxicology and AOP development, and member of the CIAO coordination team; and Dr. Sally Mayasich, researcher with a background in molecular biology of endocrine and immune interactions, bioinformatics, toxicology and risk assessment. These researchers developed the AOP through focused literature searches with peer-reviewed literature preferred, but in some cases pre-prints are referenced due to the short timeframe for emerging information during the rapidly evolving COVID-19 pandemic. The COVID-19 AOPs were developed within a collaborative exchange of information through online platforms and frequent virtual meetings and workshops which also enlisted the guidance of CIAO members experienced as AOP developers and coaches. Dr. Carlie LaLone, bioinformatician, toxicologist and AOP developer and coach, US Environmental Protection Agency, contributed to later stages of development of the AOP.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1739 | Binding to ACE2 | Binding to ACE2 |
| KE | 1738 | SARS-CoV-2 cell entry | SARS-CoV-2 cell entry |
| KE | 1901 | Interferon-I antiviral response, antagonized by SARS-CoV-2 | IFN-I response, antagonized |
| KE | 1847 | Increased SARS-CoV-2 production | SARS-CoV-2 production |
| AO | 1939 | Viral infection and host-to-host transmission, proliferated | Viral infection, proliferated |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Binding to ACE2 leads to SARS-CoV-2 cell entry | adjacent | High | Moderate |
| SARS-CoV-2 cell entry leads to IFN-I response, antagonized | adjacent | High | Not Specified |
| IFN-I response, antagonized leads to SARS-CoV-2 production | adjacent | High | Not Specified |
| SARS-CoV-2 production leads to Viral infection, proliferated | adjacent | High | Not Specified |
Network View
Prototypical Stressors
| Name |
|---|
| SARS-CoV |
| Sars-CoV-2 |
| HCoV-NL63 |
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| mink | Mustela lutreola | High | NCBI |
| ferret | Mustela putorius furo | High | NCBI |
| cat | Felis catus | High | NCBI |
| dog | Canis lupus familiaris | High | NCBI |
| Syrian golden hamster | Mesocricetus auratus | High | NCBI |
| rhesus macaque | Macaca mulatta | High | NCBI |
| lowland gorilla | Gorilla gorilla gorilla | High | NCBI |
| crab eating macaque | Macaca fascicularis | High | NCBI |
| African green monkeys | Chlorocebus aethiops | High | NCBI |
| humans | Homo sapiens | High | NCBI |
| Hippopotamus amphibius | Hippopotamus amphibius | High | NCBI |
| bank vole | Myodes glareolus | High | NCBI |
| Lynx canadensis | Lynx canadensis | High | NCBI |
| Puma concolor | Puma concolor | High | NCBI |
| Panthera tigris jacksoni | Panthera tigris jacksoni | High | NCBI |
| Panthera uncia | Uncia uncia | High | NCBI |
| Prionailurus viverrinus | Prionailurus viverrinus | High | NCBI |
| Crocuta crocuta | Crocuta crocuta | High | NCBI |
| Arctictis binturong | Arctictis binturong | High | NCBI |
| Odocoileus virginianus | Odocoileus virginianus | High | NCBI |
| American mink | Neovison vison | High | NCBI |
| Nasua nasua | Nasua nasua | High | NCBI |
| Panthera leo | Panthera leo | High | NCBI |
| Sus scrofa | Sus scrofa | High | NCBI |
| European rabbit | Oryctolagus cuniculus | High | NCBI |
| Castor fiber | Castor fiber | High | NCBI |
| Aonyx cinereus | Aonyx cinerea | High | NCBI |
| Vulpes vulpes | Vulpes vulpes | High | NCBI |
| Nyctereutes procyonoides | Nyctereutes procyonoides | High | NCBI |
| Tupaia belangeri | Tupaia belangeri | High | NCBI |
| Bos taurus | Bos taurus | High | NCBI |
| Odocoileus hemionus | Odocoileus hemionus | High | NCBI |
| Peromyscus maniculatus bairdii | Peromyscus maniculatus bairdii | High | NCBI |
| Cynopterus brachyotis | Cynopterus brachyotis | High | NCBI |
| common marmoset | Callithrix jacchus | High | NCBI |
| baboon | Papio anubis | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | Not Specified |
Overall Assessment of the AOP
See details below.
Domain of Applicability
Life stage and sex
Although COVID-19 has shown to be a more severe illness in older than in young people, there is evidence that viral load was not influenced by age or sex (Challenger et al., 2022), and infection rate and viral load did not differ by sex (Arnold et al., 2022; Qi et al., 2021; Cheemarla et al., 2021). Therefore, this AOP is applicable to all life stages and both sexes.
Taxonomic domain
No non-mammals have been found to be infected by SARS-CoV-2. Mammals listed in the Taxonomic Applicability table were either experimentally or naturally infected, as confirmed by polymerase chain reaction (PCR) or antibody assays, hence evidence is high for these species. Other mammalian species are likely also susceptible, but some mammals experimentally exposed to the virus did not become infected (Bosco-Lauth et al., 2021). The AOP is therefore applicable to humans and other mammals. Infections in non-human mammals is important in the potential for zoonotic spillover and is discussed in more detail in the adverse outcome (AO 1939), with species-specific references.
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|