
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 34
Title
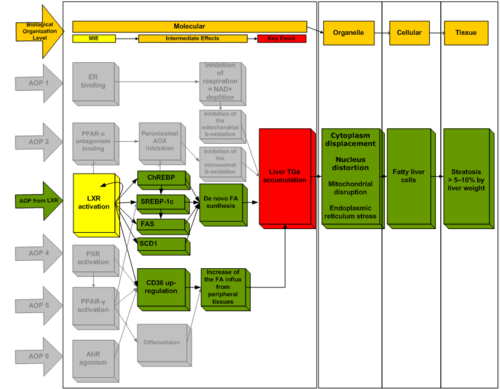
LXR activation leading to hepatic steatosis
Short name
Graphical Representation
Point of Contact
Contributors
- Agnes Aggy
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Activation, LXR | May 21, 2024 10:33 |
| Up Regulation, CD36 | March 26, 2024 10:35 |
| Increase, FA Influx | March 29, 2024 11:03 |
| Activation, ChREBP | September 16, 2017 10:14 |
| Activation, SREBP-1c | September 16, 2017 10:14 |
| Activation, FAS | September 16, 2017 10:14 |
| Activation, SCD-1 | September 16, 2017 10:14 |
| Synthesis, De Novo Fatty Acid (FA) | March 29, 2024 10:55 |
| Accumulation, Triglyceride | March 26, 2024 13:09 |
| N/A, Liver Steatosis | September 16, 2017 10:14 |
| peroxisome proliferator activated receptor promoter demethylation | September 16, 2017 10:14 |
| Mitochondrial dysfunction | April 17, 2024 08:26 |
| Activation, LXR leads to Up Regulation, CD36 | December 03, 2016 16:37 |
| Up Regulation, CD36 leads to Increase, FA Influx | March 27, 2024 10:00 |
| Activation, LXR leads to Activation, ChREBP | December 03, 2016 16:37 |
| Activation, LXR leads to Activation, SREBP-1c | December 03, 2016 16:37 |
| Activation, LXR leads to Activation, FAS | December 03, 2016 16:37 |
| Activation, LXR leads to Activation, SCD-1 | December 03, 2016 16:37 |
| Activation, SCD-1 leads to Synthesis, De Novo Fatty Acid (FA) | December 03, 2016 16:37 |
| Activation, ChREBP leads to Synthesis, De Novo Fatty Acid (FA) | December 03, 2016 16:37 |
| Activation, SREBP-1c leads to Synthesis, De Novo Fatty Acid (FA) | December 03, 2016 16:37 |
| Activation, FAS leads to Synthesis, De Novo Fatty Acid (FA) | December 03, 2016 16:37 |
| Synthesis, De Novo Fatty Acid (FA) leads to Accumulation, Triglyceride | March 29, 2024 12:11 |
| Increase, FA Influx leads to Accumulation, Triglyceride | March 29, 2024 12:08 |
| Accumulation, Triglyceride leads to Mitochondrial dysfunction | March 13, 2024 17:57 |
| demethylation, PPARg promoter leads to Up Regulation, CD36 | November 29, 2016 20:10 |
Abstract
Liver steatosis (fatty liver) is considered as one of the first manifestations of possible hepatotoxicity, however it is not regarded as an adverse effect per se and usually can be reversible. The importance of steatosis is highlighted from the fact that it is a prerequisite for the development of non-alcoholic fatty liver disease (NAFLD). NAFLD is a clinicopathological condition that comprises a wide spectrum of liver damage, ranging from steatosis alone to steatohepatitis, advanced fibrosis and cirrhosis. Non-alcoholic steatohepatitis (NASH) represents only a stage in the spectrum of NAFLD and is defined pathologically by the presence of steatosis together with necro-inflammatory activity. The clinical implications of NAFLD are derived mostly from its potential to progress to end-stage liver disease, whereas simple uncomplicated steatosis follows a relatively benign course in most patients. Steatosis is the output of the disturbance on the homeostasis of hepatic lipids which depends on the dynamic balance of several pathways including fatty acid (FA) uptake, de novo FA synthesis, β-oxidation, and very low-density lipoprotein (VLDL) secretion. It is characterized by the accumulation of lipid droplets (mainly triglycerides) in the hepatocytes. This AOP describes the linkage between hepatic steatosis triggered by nuclear receptors activation (PPAR gamma and LXR) through modulation of genes responsible for lipid homeostasis [the carbohydrate response element binding protein (ChREBP), the sterol response element binding protein 1c (SREBP-1c), the free fatty acid uptake transporter FAT/CD36, the fatty acid synthase (FAS), the stearoyl-CoA desaturase 1 (SCD1)] which subsequently leasds to in rease of de novo fatty acids/triglycerides synthesis and fat influx from the peripheral tissues to liver. The accumulation of lipid in the hepatocytes can cause cytoplasm displacement, nucleus distortion, mitochondrial toxicity and eventually necrosis and/or apoptosis. The progression of this condition can lead to tissue inflammation (steatohepatitis) and fibrosis with the involvement of other cells of the hepatic tissue like the Kupffer (inflammation) and the stellate (fibrosis) cells. This purely qualitative AOP description is plausible, the scientific data supporting the AOP are logic, coherent and consistent and there is temporal agreement between the individual KEs. Quantitative data on dose-response-relationships and temporal sequences between key events are still lacking; the provision of quantitative data will strengthen the weight of evidence and make the AOP applicable for chemical risk assessment purposes.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 167 | Activation, LXR | Activation, LXR |
| MIE | 228 | peroxisome proliferator activated receptor promoter demethylation | demethylation, PPARg promoter |
| KE | 54 | Up Regulation, CD36 | Up Regulation, CD36 |
| KE | 115 | Increase, FA Influx | Increase, FA Influx |
| KE | 66 | Activation, ChREBP | Activation, ChREBP |
| KE | 264 | Activation, SREBP-1c | Activation, SREBP-1c |
| KE | 116 | Activation, FAS | Activation, FAS |
| KE | 258 | Activation, SCD-1 | Activation, SCD-1 |
| KE | 89 | Synthesis, De Novo Fatty Acid (FA) | Synthesis, De Novo Fatty Acid (FA) |
| KE | 291 | Accumulation, Triglyceride | Accumulation, Triglyceride |
| KE | 177 | Mitochondrial dysfunction | Mitochondrial dysfunction |
| AO | 345 | N/A, Liver Steatosis | N/A, Liver Steatosis |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
Sex Applicability
Overall Assessment of the AOP
Consider the following criteria (may include references to KE Relationship pages): 1. concordance of dose-response relationships; 2. temporal concordance among the key events and adverse effect; 3. strength, consistency, and specificity of association of adverse effect and initiating event; 4. biological plausibility, coherence, and consistency of the experimental evidence; 5. alternative mechanisms that logically present themselves and the extent to which they may distract from the postulated AOP. It should be noted that alternative mechanisms of action, if supported, require a separate AOP; 6. uncertainties, inconsistencies and data gaps.
Concordance of dose-response relationships
The existing studies do not provide dose-response curves. However it may be possible in some cases to construct curves from the given numerical data and to relate the dose response for LXR activation with the dose response for TG accumulation in vitro and in vivo in a second more quantitative iteration as the next step of this AOP development.
Temporal concordance among the key events and adverse outcome
According to the available information the sequence of the events is in strong agreement and consequently the presented MoA could be considered as qualitatively accurate.
Strength, consistency, and specificity of association of adverse outcome and initiating event
The scientific evidence is presented in Scientific Evidence in support of the MoA.
Biological plausibility, coherence, and consistency of the experimental evidence
The steatogenic effect of chemicals like LXR ligands is well established in the literature (Peet 1998, Schultz et al. 2000, Horton et al. 2002) and it is well correlated with the expression of the receptor (Moya et al. 2010) the binding to it. In addition it is believed that LXR acts as a cholesterol sensor. Consistent with this role, it has been proposed that LXR induces SREBP-1c in order to generate fatty acids needed for the formation of cholesterol esters, which buffer the free cholesterol concentration (Ferré & Foufelle 2007). Further analysis of the logic, coherence and consistency along with the experimental data has already been presented in Chapter Scientific Evidence in support of the MoA.
Alternative mechanism(s) that logically present themselves and the extent to which they may distract from the postulated AOP.
As already mentioned abovem there are many other possible MoAs of a chemical in the development of steatosis including MoAs involving the inhibition of β-oxidation, the inhibition of oxidative phosphorylation (leading to a decrease of ATP needed for β-oxidation) and the malfunction of the mechanisms of the excretion of TG from the cell. These pathways are not covered in the presented MoA as they are not directly linked with the activation of LXR. Furthermore, as already explained LXR is not the only receptor which has been identified to be involved in fatty acid metabolism and steatogenesis. Exogenous chemicals acting as ligands for any of the following Nuclear Receptors (AhR, PXR, PPARa, PPARγ and ER) may play a role in the development of steatosis (grey elements on the AOP flow diagram, Fig. 3). There also known interactions or cross-talk between the NRs. Examples of possible interactions are related with fact that LXR is also regulated by the PPARa, the FAT/36 up-regulation from the AhR, PXR and PPARγ, the inhibition of β-oxidation from PPARa and indirectly from the ER.
It may be possible from existing literature, or further experimental work to develop MoAs taking binding to each of these receptors as the molecular initiating event and describing the converging pathways leading to steatosis. In fact this work is in progress and indicated as the grey elements on the AOP flow diagram above.
The biology of LXR function has been studied using the high affinity synthetic ligands T0901317. According to the study of Mitro (2007), T0901317 binds and activates hPXR and hLXRβ with similar affinity, and can regulate multiple PXR target genes in human cells and mice (like CD36) with similar efficacy to established PXR ligands, but significantly greater potency (Mitro 2007). The author suggested that some of the effects observed with T0901317 such as the more deleterious increase in lipogenesis and hepatic lipid accumulation (in comparison to the LXR-selective GW3965) that have been ascribed to LXR activation maybe the result of simultaneous stimulation of PXR and LXR activity and that the assumption that T0901317 behaves as an LXR-selective agonist may have led to some inaccurate conclusions regarding the effects of LXR activation in vivo.
From the data of this study it is evident that SREBP-1c, FAS and SCD-1, which are LXR but not PXR regulated genes, were significantly up-regulated by T0901317. In contrast GW3965 up-regulates less effectively the SREBP-1c, marginally the SCD-1 and not at all the FAS despite the fact that it is considered as a selective LXR agonist (Mitro 2007). The CD36 gene is considered also as a liver specific target of LXR activation (Zhou 2008). However, in the study of Mitro (2007), GW3965 did not up-regulate CD36. These findings could be explained by the lower affinity of this synthetic LXR agonist (EC50 of 0.19 and 0.03 μM for hLXRα and hLXRβ) in relation to the T0901317 (EC50 of 0.02-0.05 μM for both isoforms). Interestingly and despite the low up-regulating activity, GW3965 increases FA and TG accumulation in rat and primary human hepatocytes (Kotokorpi et al. 2007). Based on this information, it could be possible that T0901317 binding on PXR could enhance its steatogenic activity with the proposed MoA still being plausible. This plausibility, however, is clearly related with quantitative aspects.
In conclusion, the MoA described can be considered very well supported by the available scientific evidence and it is biologically plausible.
Uncertainties, inconsistencies and data gaps
The information used for the development of the present pathway is based on in-vitro and in-vivo studies. In the in-vitro studies several cell lines have been used. The expression of the LXR, the SREBP-1c and other elements on these cell lines is a key factor for the plausibility of the pathway in human. According to the study from Moya et al 2010, LXR expression (as measured from mRNA using RT-PCR) in human hepatocytes, HepG2 and HeLa cells was approximately 70%, 70% and 50% in relation to the level of expression in human liver. In addition the expression of SREBP-1c was significantly down-regulated (to less than 25% of normal levels of expression in the liver) in all 3 cell lines. Consequently positive results in relation to fat accumulation after LXR activation from studies using these cell lines may under-estimate the magnitude of effect on human liver while negative results could be interpreted as inconclusive. The assessment of the relative expression of these receptors in other cell lines would be of great importance in order to evaluate the relevance of each in vitro study result.
In relation to the in vivo studies which have be made mainly (if not exclusively) in rodents the relevance for humans should be addressed. LXR expression is considered adequately conserved from rodents to humans. In addition it is well known that all the other elements of the pathway are present in human liver. A good example of this is that the well-defined pathogenic role of FAT/CD36 in hepatic steatosis in rodents is also confirmed by the up-regulation in humans of the FAT/CD36 in cases of NASH, NAFLD, insulin resistance, hyperinsulinaemia, HCV and morbidly obese patients (Zhu et al. 2011, Love-Gregory & Abmurad 2011, Miquilena-Colina et al. 2011, Bechmann et al. 2010). However, there is some speculation in relation to the extent that adverse side effects observed in rodents will occur in higher species, including humans. These speculations are raised due to the different behaviour of the LXR agonist GW3965 in in vitro systems which although markedly stimulating lipogenic gene expression in primary human hepatocytes leading to significant TG accumulation at all 3 dose levels after 48 hr, produced only a very modest increase in the triglyceride content in rat cells (Kotokorpi et al. 2007), demonstrating that the use of this rat cell line could underestimate the effect in humans. FA increase was reported in both cell lines.
Another interesting finding is that in humans, total CD36 deficiency is relatively common (3–5%) in persons of African and Asian descents (Su & Abmurad 2009). Consequently the presented MoA could be affected mainly quantitatively among humans of different origin.
Induction of lipogenic enzymes from the SREBP-1c is evidenced in adult mice but not during the fetal life indicating a different role of the SREBP-1c between these two stages (Liang et al. 2002). This finding gives a strong indication that the presented pathway may be altered in other than adult life stage.
Another finding is that of the study of Hu et al. 2005 according to which administration of T0901317 in PPAR-null mice promoted a dose-dependent increase in the rate of peroxisomal β-oxidation in the liver and in relation only to the LXRα. The author suggests that this induction may serve as a counter regulatory mechanism for responding to the hypertriglyceridemia and liver steatosis that is promoted by potent LXR agonists in vivo.
T090137 was shown to up-regulate hepatic expression and plasma activity of PLTP in mice in addition to angiopoietin-like protein 3 (Angptl3), playing a critical role in LXR-induced hypertriglyceridemia. However it should be noted that hypertriglyceridemic effect of LXR agonists is usually transient and limited to the first few days of the treatment likely due to enhanced VLDL-triglyceride hydrolysis resulting from increased expression of hepatic LPL (Baranowski 2008).
Some studies have demonstrated absence of triglyceride accumulation on SREBP-1c (-/-) mice suggesting that SREBP-1c is a crucial element of the present MoA (Liang et al. 2002, Schultz et al. 2000, Horton et al. 2002, Shimano et al. 1999). In another study in FAT/CD36 knockout mice the effect of LXR agonists on increasing hepatic and circulating levels of triglycerides and free fatty acids (FFAs) was largely abolished suggesting that intact expression and/or activation of FAT/CD36 is required for the steatotic effect of LXR agonists (Febbraio et al. 1999). These two findings together and considering that they are constant and not related with specific experimental conditions could lead one to the hypothesis that both SREBP-1c and CD36 are imperative elements for the cause of steatosis. This hypothesis could be further examined.
The present MoA could also be affected by factors related to the formation of steatosis such as trends in adipose tissue (AT) deposition, the total body fat, the visceral AT and the subcutaneous AT which vary among different life stages such as childhood, puberty and adolescence, between sexes and among humans of different origin (Staiano 2012).
Assessment of the quantitative understanding of the AOP
In the present study only qualitative assessment of the proposed MoA was performed. In the studies used there are numerical data mainly to support the expression and up-regulation of the different elements of the pathway. However, further analysis of these numerical data is suggested in following steps.
Interestingly, the existence of many network motifs along the pathways was noted during the analysis of the literature, e.g. the positive feed forward LXR up-regulation. This information could be used in the future for the quantitative interpretation of dose response curves and the development of quantitative prediction models of the adverse outcome following the activation of the LXR.
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Confidence in the AOP
Information from this section should be moved to the Key Event Relationship pages! Elaborate on the domains of applicability listed in the summary section above. Specifically, provide the literature supporting, or excluding, certain domains.
How well characterised is the AOP?
Liver steatosis is a well understood adverse outcome. A great number of publications from in vitro, in vivo, mechanistic, clinical and epidemiological studies exist for the qualitative assessment of steatosis. However, the quantitative analysis of the role of a specific exogenous chemical in an adverse outcome in human is a very challenging task due to the involvement of a large number of inter-related factors following the MIE. In fact one chemical may bind to more than one receptor and consequently have different impacts either quantitatively or qualitatively on the downstream events.
How well are the initiating and other key events causally linked to the outcome?
LXR agonists such T0901317 have been shown to produce LXR activation, as well as triglyceride accumulation, which has been demonstrated in rodent (mouse and rat) and human liver cell lines in vitro. The same chemicals shown to be LXR agonists in the in vitro assays have shown triglyceride accumulation in the liver leading to steatosis in animals and humans through steps of the reported MoA.
What are the limitations in the evidence in support of the AOP?
Disagreement in the scientific evidence supporting the presented AOP was not found. In relation to data gaps in addition to lack of quantitative information as discussed above there is also lack of specific information in relation to the role of other target genes expressed after the LXR activation.
Is the AOP specific to certain tissues, life stages / age classes?
There is evidence of different levels of expression of CD36 in different ethnic groups which may be expected to alter the sensitivity to development of steatosis. There may also be differences in expression and role of the same proteins/enzymes in foetal life but this has not been fully elucidated. Further information can be found in the Chapter Uncertainties, inconsistencies and data gaps.
Are the initiating and key events expected to be conserved across taxa?
From the analysis of the available information from experimental studies using rodents the elements of the MoA appeared to be well conserved between mice and rats. Some concerns in relation to the relevance of the in vivo studies to human are raised mainly due to the different behaviour of the LXR agonist GW3965 which while stimulating lipogenic gene expression in human hepatocytes, causes only a slight increase in TGs in rats (Kotokorpi et al. 2007). Some more differences were also reported between hamsters and monkeys in relation to hypertriglyceridemia (Groot et al. 2005).
Scientific Evidence to Support AOP
Information from this section should be moved to the Key Event Relationship pages! Detailed Description
Old format, potemtially to be migrated to table above and underlying articles
| Events | Scientific Support | Strength of Evidence |
|---|---|---|
| LXR binding and activation
(Molecular initiating event) receptor |
Very Strong | |
| Binding in the LXREs | [11] | Very Strong |
| Target genes transcription | [12] | Very Strong |
| Auto-regulation of the LXRa | Very Strong | |
| Increase in expression and activity of the carbohydrate response element-binding protein (ChREBP) | Strong
According to the study of Denechaud increase in the glucose flux in the cell is a prerequisite for ChREBP activation from T0901317 in mice |
|
| Increase in expression of the SREBP-1c from LXR | Very Strong
However, there are many studies supporting a different behaviour between LXRα and LXRβ, suggesting that SREBP-1c up-regulation is only due to LXRα |
|
| Increase in expression of the SREBP-1c from the ChREBP | [23] | Well established |
| Induction of lipogenic enzymes from the SREBP-1c (FAS, ACC, GK) | Very Strong
However there is evidence that this effect is not induced in the embryonic state indicating a different role of the SREBP-1c between embryonic and adult life (Liang et al. 2002). It is also suggested that for lipogenic genes, SREBP-1c acts together with ChREBP (Ishii et al. 2004). |
|
| Direct induction of the fatty acid synthase (FAS) | Very Strong | |
| Direct induction of the stearoyl-CoA desaturase 1 (SCD1) | Very Strong | |
| Up-regulation of the free fatty acid uptake transporter FAT/CD36 | Very Strong | |
| De novo fatty acids and triglyceride synthesis | [43] | Very Strong |
| Fat influx from the peripheral tissues | Very Strong | |
| Steatosis | Very Strong |
Cite error: <ref> tags exist, but no <references/> tag was found