
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 413
Title
Oxidation and antagonism of reduced glutathione leading to mortality via acute renal failure
Short name
Graphical Representation
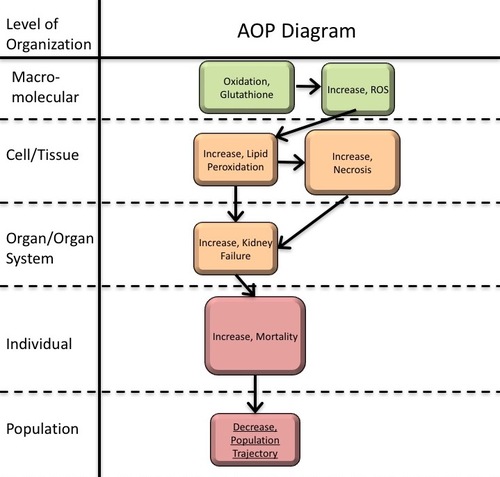
Point of Contact
Contributors
- Zarin Hossain
- Agnes Aggy
Coaches
- Knut Erik Tollefsen
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Oxidation, Glutathione (To be considered with MIE) | November 09, 2017 06:40 |
| Increased, Reactive oxygen species | January 29, 2025 12:41 |
| Increased, Kidney Failure | June 21, 2024 10:52 |
| Increase, Necrosis | March 19, 2019 09:31 |
| Increased Mortality | July 08, 2022 07:32 |
| Increased, Lipid peroxidation | July 27, 2023 10:25 |
| Oxidation, Glutathione leads to Increased, Reactive oxygen species | August 05, 2021 14:37 |
| Increased, Reactive oxygen species leads to Increased, LPO | April 11, 2024 16:24 |
| Increased, LPO leads to Increase, Necrosis | August 11, 2021 15:34 |
| Increase, Necrosis leads to Increased, Kidney Failure | August 11, 2021 15:35 |
| Increased, Kidney Failure leads to Increased Mortality | August 11, 2021 15:35 |
| Uranium | August 05, 2021 14:28 |
| Arsenic | April 27, 2021 00:15 |
| Bis(2,4,6-trimethylphenyl)-lambda~2~-germane--selenium (1/1) | August 05, 2021 16:23 |
Abstract
We are assiduously exploring mechanisms of toxicity of uranium to fish species in the context of uranium mining via an adverse outcome pathway (AOP). We have created an AOP that addresses the toxicity of aqueous concentrations of uranium. Our molecular initiating event is the oxidation of reduced glutathione, which occurs in the presence of reactive oxygen species (ROS) inducing materials such as uranium. Reduced glutathione is an antioxidant that helps cellular redox homeostasis. This oxidation prevents anti-oxidant function. The following key event - increase in ROS, is a result of both uranium promoting ROS and oxidizing glutathione, to perpetrate oxidative stress. An increase in ROS leaves major macromolecules such as DNA, proteins and membrane phospholipids susceptible to damage. Although there are multiple effects as a result of an increase in ROS, an AOP calls for one specific path. Thus, our following key event is lipid peroxidation, as it is widely observed in uranium-fish toxicity studies. Consequently, the increase in ROS is being investigated for its ability to damage lipids in the cell membranes, more specifically in the kidney for this AOP. Accordingly, our adverse outcome is the development of acute renal failure in fish species. This would be of regulatory significance as renal damage is an endpoint of concern to environmentalists and researchers in this field, as it is likely to lead to population decline.
AOP Development Strategy
Context
The biology and chemistry of uranium effluent and its effects have been well explored, especially within fish species(Cooley et al. 2000; Kelly and Janz 2009; Goulet et al. 2011; Ma et al. 2020), however it has not been clearly presented.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 926 | Oxidation, Glutathione (To be considered with MIE) | Oxidation, Glutathione |
| KE | 1115 | Increased, Reactive oxygen species | Increased, Reactive oxygen species |
| KE | 1445 | Increased, Lipid peroxidation | Increased, LPO |
| KE | 1607 | Increase, Necrosis | Increase, Necrosis |
| KE | 759 | Increased, Kidney Failure | Increased, Kidney Failure |
| AO | 351 | Increased Mortality | Increased Mortality |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Oxidation, Glutathione leads to Increased, Reactive oxygen species | adjacent | Moderate | Moderate |
| Increased, Reactive oxygen species leads to Increased, LPO | adjacent | High | Moderate |
| Increased, LPO leads to Increase, Necrosis | adjacent | Moderate | Moderate |
| Increase, Necrosis leads to Increased, Kidney Failure | adjacent | Moderate | Moderate |
| Increased, Kidney Failure leads to Increased Mortality | adjacent | High | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | Moderate |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP
The overall assesment renders this AOP: Oxidation and antagonism of reduced glutathione leading to population decline via acute renal failure, moderately plausible and applicable. The domain of applicability applies to all organisms able to experience lipid peroxidation and/or kidney failure. Key events are deemed essential, as there are likely events between events that are not accounted for or as of the same importance.
Domain of Applicability
This AOP is applicable to all life stages and all organisms that produce reactive oxygen species and/or experience lipid peroxidation, and/or experience kidney failure, unless otherwise stated. It is important to note that there is evidence for this AOP primarily in fish studies, however there is no indication that this AOP would not be applicable to other species or life stages. Biological plausibility is high, as much of this AOP is dependant on studies observing uranium toxicity in fish species. There is moderate emperical support, as there are studies available on these KER's but there is a lack of information on dose-response relationships in this AOP. Overall, this AOP should be considered for regulatory significance, especially for fish species exposed to uranium (possible from uranium milling effluent) because this AOP has been observed from these studies.
Essentiality of the Key Events
|
Support for Essentiality of KEs |
Defining Question Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
High (Strong) |
Moderate |
Low (Weak) |
|
Direct evidence from specifically designed experimental studies illustrating essentiality for at least one of the important KEs (e.g. stop/reversibility studies, antagonism, knock out models, etc.) |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE leading to increase in KE down or AO |
No or contradictory experimental evidence of the essentiality of any of the KEs |
||
| Oxidation, Glutathione (MIE) | High |
|
||
|
Increase, ROS Increase, Lipid Peroxidation |
High |
|
||
| Increase, Necrosis | High |
|
||
| Increase, Kidney Failure | Moderate |
|
||
|
Increase, Mortality (AO) |
High |
|
||
Evidence Assessment
Biological plausibility.
The biological plausibility for this AOP is overall moderate. It is biologically possible for each step to perpetrate one after the other and for all key events, it is well accepted that one insigates the other. However, the mechanisms and relationships between key events are not fully established or understood. It is assumed that these processes are dynamic and complicated.
|
Support for Biological Plausibility of KERs |
Defining Question |
High (Strong) |
Moderate | Low (Weak) |
|
Is there a mechanistic (i.e. structural or functional) relationship between KEup and KE down consistent with established biological knowledge? |
Extensive understanding of the KER based on extensive previous documentation and broad acceptance. |
The KER is plausible based on analogy to accepted biological relationship, but scientific understanding is not completely established. | There is empirical support for a statistical association between KES but the structural or functional relationship between them is not understood. | |
|
MIE to KE1 Glutathione Oxidation leads to Increase in ROS |
MODERATE |
Rationale: The oxidation of reduced glutathione is (at least partially) resposible for the increase in ROS. Reduced glutathione is an anti-oxidant that plays a role in homeostasis of cellular redox and eliminating free radicals/reactive oxygen species (Pizzorno 2014; Kalinina et al. 2014). When a oxidative stressor is able to bind to reduced glutathione, it is inhibited and becomes its oxidized form (oxidized glutathione). Significantly, inhibiting an anti-oxidant would increase ROS. Not only this, an imbalance between reduced and oxidized glutathione itself, is also a reason for increase in ROS and susceptibility to oxidative stress. |
||
|
KE1 to KE2 Increase in ROS leads to Increase in Lipid Peroxidation |
STRONG |
Rationale: Oxidative stress is often manifested as lipid peroxidation, thus lipid peroxidation is often used to measure oxidative stress. Lipid peroxidation occurs when reactive oxygen species attack and damage the poly unsaturated fatty acids of cellular and subcellular phospholipid membranes. This can cause structural disorder, and damage important ezymes and proteins (Ayala et al. 2014). Therefore, it was quite obvious how an increase in ROS would increase lipid peroxidation, as there are more reactive species able to attack the double-bond lipid membranes. |
||
|
KE2 to KE3 Increase in Lipid Peroxidation leads to Increase in Necrosis |
MODERATE |
Rationale: Necrosis is a common response to trauma to cells. As mentioned, lipid peroxidation can damage the structural integrity and function of phopholipid membranes; a form of trauma (Ayala et al. 2014). An increase in ROS and lipid peroxidation is commonly cited as an instgator of necrotic cell death (Kehrer 1993). Built-up damage from lipid peroxidation can cause cell and/or organ membranes to break and malfunction. Therefor, an increase in necrosis can be expected as a result of an increase in ROS and lipid peroxidation. |
||
|
KE3 to KE4 Increase in Necrosis leads to Kidney Failure |
MODERATE |
Rationale: Again, damage to lipid membranes can affects the viability/survivability of cells and organs. Necrosis and cellular damage have been primarily seen in the liver and kidneys when observing fish species exposed to uranium. There is most definitely other manifestations of an increase in necrosis, however for this AOP, kidney failure was chosen. Kidney failure is characterized by both loss of function and integrity (Lameire 2005; Hilton 2011), which can result from an increase in pathological lesions arising via necrosis. It is biologically very plausible for kidney damage to arise (partially) because of necrosis in kidney tissue, however, it is likely that several other factors regarding kidney survivability also play a role to perpetrate significant kidney failure. |
||
|
KE4 to AO Kidney Failure leads to Increase in Mortality |
HIGH |
Rationale: Significant kidney failure can lead to death (Cooley et al. 2000). This has been seen in humans, and many other species. It is well accepted. It is likely that kidney failure may not be the only reason for mortality from this pathway, however it is positive that the drecreased function of the kidney plays a role in survivability. Therefore, it is logical to hypothesize that kindey failure can lead to death in this AOP. |
||
Empirical Support
The empirical support available for this AOP is moderate. Clear evidence between dose response relationships is not present. However, there are several in-vivo and in-vitro experiments showing these key events respectively causing each other. Only one stressor, uranium, is considered for the empirical support.
|
Empirical support for KERs |
Defining Question Does the empirical evidence support that a change in the KEup leads to an appropriate change in the KE down? Does KEup occur at lower doses and earlier time points than KE down and is the incidence of KEup higher than that for KE down? Are inconsistencies in empirical support cross taxa, species and stressors that don’t align with expected pattern of hypothesized AOP? |
High (Strong) |
Moderate |
Low(Weak) |
|
Multiple studies showing dependent change in both exposure to a wide range of specific stressors (extensive evidence for temporal, dose-response and incidence concordance) and no or few critical data gaps or conflicting data. |
Demonstrated dependent change in both events following exposure to a small number of specific stressors and some evidence inconsistent with expected pattern that can be explained by factors such as experimental design, technical considerations, differences among laboratories, etc. |
Limited or no studies reporting dependent change in both events following exposure to a specific stressor (ie endpoints never measured in the same study or not at all); and/or significant inconsistencies in empirical support across taxa and species that don’t align with expected pattern for hypothesized AOP |
||
|
MIE to KE1 Glutathione Oxidationleads to Increase in ROS |
MODERATE |
Rationale: Kelly and Janz (2009) demonstrated that low exposure of uranium had significantly increased oxidized glutathione overall and compared to reduced glutathione. Uranium exposure has also shown an increase in genes playing a role in glutathione homeostasis (Lerebours et al. 2009; Barillet et al. 2011; Song et al. 2012.). |
||
|
KE1 to KE2 Increase in ROS leads to Increase in Lipid Peroxidation |
MODERATE |
Rationale: An increase in ROS was determined as the cause of significant lipid peroxidation in juvenile northern pike inhabiting lakes downstream of uranium milling facilities, compared to a control (Kelly and Janz 2009). |
||
|
KE2 to KE3 Increase in Lipid Peroxidation leads to Increase in Necrosis |
MODERATE |
Rationale: In whitefish exposed to uranium via diet, damaged lesions were observed the most in the liver where necrotic cell death was also highly observed. This necrotic cell death was accredited to oxidative stress and an increase in lipid peroxidation. The authors also suggested that the increase in necrosis was dose-dependant on uranium exposure (Cooley et al. 2000). |
||
|
KE3 to KE4 Increase in Necrosis leads to Kidney Failure |
LOW |
Rationale: lesions have been especially observed in the kidney of fish species as a result of exposure to uranium exhibiting the KE's listed above, including in the the proximal and distal convoluted tubes (Kelly and Janz 2009). There is no/little quantitative analysis confirming this KER. However, no evidence suggests against necrotic lesions instigating kidney damage or failure. It is biologically plausible and thus appropriate. |
||
|
KE4 to AO Kidney Failure leads to Increase in Mortality |
HIGH |
Rationale: Stressors responsible for inducing this AOP (such as uranium) have been observed to decrease egg hatchability and reproductive success, decrease survivability, hinder growth and increase mortality overall (Pyle et al. 2002; Kelly and Janz 2009; Goertzen et al. 2011; Simon et al. 2011). There is little quantitative data available for direct evidence showing that kidney failure will lead to mortality, however it is widely accepted that severe cases have high probability of death. Again, there are additional manifestations of uranium toxicity (not outlined in this AOP) that can contribute to death. It is therefore, very difficult to measure and conclude the "dose"-response relationship between these key events ie. how much kidney failure causes mortality. Though, because this phenomenon is widely accepted (in it's inconclusive form), we suggest the emperical support is high. |
||
Uncertainties and Inconsistencies
There are a few uncertainties for consideration:
- The increase in reactive oxygen species could be the molecular initiating event - while this is true and an increase is ROS is a major contributor and key event to this AOP, proceeding is the event of oxidation of glutathione. The increase is ROS is at least partially due to the oxidation of glutathione but is also be at the cause of other events not mentioned in this AOP. ROS production occurs in multiple ways and is quite complex, however the oxidation of glutathione was pertinent when looking at fish species in which this AOP was based.
- At high concentrations of the stressor, it may be expected that reduced glutathione is found at a higher concentration in exposed subjects compared to a control (Kelly and Janz 2009). This is confusing at first, but may be appropriate. Because there is an increase in ROS, a feedback loop exists where the increase in ROS causes an increase in production of reduced glutathione to combat the increased ROS production. There is still oxidation of glutathione occuring, however, an increase in reduced glutathione may also be an indicator or an increase in ROS. At low concentrations, the expected result can be seen where there is a decrease in reduced glutathione or increase in oxidized glutathione compared to a control (Song et al. 2012). Theoretically, in propagating this AOP, oxidized glutathione should typically be in a higher ratio compared to reduced glutathione. Yet, these processes are quite complex and also may very from species to species or organism to organism. They key indicator is an imabalance in the anti-oxidant mechanisms existing with (reduced/oxidized) glutathione.
Acknowledging these inconcistencies is important to understand the decisions made in this AOP. Although these inconcistencies are present, they do not hinder the validity of the AOP in its biological plausibility or empirical analsysis. Please take these notes into consideration when evaluating the AOP.
Known Modulating Factors
Quantitative Understanding
The WOE analysis indicates that many KEs and KERs lack some experimental evidence, especially that showing that without one KE, the following KE will be affected or non-existent. However, the overall analysis supports the AOP. Because there is a lack of experimentation on dose-response relationships within this AOP, we propose that it is a qualitative AOP. This is appropritate because the overall pathway is partially based on several studies observing uranium toxicity in fish species. This provides strong evidence and biological plausability in the development of this AOP. There is a large degree of biological plausibility as many of these phenomenons are widly accepted and are logically presented in regards to the sequence of key events. The lack of specific experimental data and uncertainty in quantitative experimentation is difficult for this AOP, as many factors can simultaneously cause key events (such as an increase in ROS or kidney failure). Especially when regarding fish studies, there are several external factors to consider in the environment that may affect these key events. To some degree, many studies have assumed the relationship between exposure and toxic response is dose dependant, however it is yet to be quantified and accepted. In-vitro studies may provide more insight on the dose-response relationship between the stressor and key events, thus providing a better quantitative understanding. A future need for this AOP is a vigorous and thorough experiment model system to evaluate dose-response relationships, conentration-response relationships and exposure-effect relationships within the framework of this AOP.
Considerations for Potential Applications of the AOP (optional)
The establishment of this AOP will allow for the understanding of uranium toxicity to fish species and predictive toxicology with stressors inducing glutathione oxidation. This may especially play a part in understanding the environmental toxicity of heavy metals. It is possible to expand this AOP and explore tritium as another toxic metal that is potentially of threat to fish species. With the prospective overlap of key events between the two stressors, this connection may be the beginning of an AOP web.
References
Ayala A, Muñoz MF, Argüelles S. 2014. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxid Med Cell Longev. 2014:360438. https://doi.org/10.1155/2014/360438
Barillet S, Adam-Guillermin C, Palluel O, Porcher J-M, Devaux A. 2011. Uranium bioaccumulation and biological disorders induced in zebrafish (Danio rerio) after a depleted uranium waterborne exposure. Environ Pollut. 159(2):495–502. https://doi.org/10.1016/j.envpol.2010.10.013
Cooley HM, Evans RE, Klaverkamp JF. 2000. Toxicology of dietary uranium in lake whitefish (Coregonus clupeaformis). Aquatic Toxicology. 48(4):495–515. https://doi.org/10.1016/S0166-445X(99)00057-0
Cooley HM, Klaverkamp JF. 2000. Accumulation and distribution of dietary uranium in lake whitefish (Coregonus clupeaformis). Aquatic Toxicology. 48(4):477–494. https://doi.org/10.1016/S0166-445X(99)00058-2
Goertzen MM, Driessnack MK, Janz DM, Weber LP. 2011. Swimming performance and energy homeostasis in juvenile laboratory raised fathead minnow (Pimephales promelas) exposed to uranium mill effluent. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology. 154(4):420–426. https://doi.org/10.1016/j.cbpc.2011.07.012
Goulet RR, Fortin C, J. Spry D. 2011. Fish Physiology: Homeostasis and Toxicology of Non-Essential Metals. [place unknown]: Academic Press.
Hilton R. 2011. Defining acute renal failure. CMAJ. 183(10):1167–1169. https://doi.org/10.1503/cmaj.081170
Kalinina EV, Chernov NN, Novichkova MD. 2014. Role of glutathione, glutathione transferase, and glutaredoxin in regulation of redox-dependent processes. Biochemistry (Mosc). 79(13):1562–1583. https://doi.org/10.1134/S0006297914130082
Kehrer JP. 1993. Free radicals as mediators of tissue injury and disease. Crit Rev Toxicol. 23(1):21–48. https://doi.org/10.3109/10408449309104073
Kelly JM, Janz DM. 2009. Assessment of oxidative stress and histopathology in juvenile northern pike (Esox lucius) inhabiting lakes downstream of a uranium mill. Aquatic Toxicology. 92(4):240–249. https://doi.org/10.1016/j.aquatox.2009.02.007
Lameire N. 2005. The Pathophysiology of Acute Renal Failure. Critical Care Clinics. 21(2):197–210. https://doi.org/10.1016/j.ccc.2005.01.001
Lerebours A, Gonzalez P, Adam C, Camilleri V, Bourdineaud J-P, Garnier-Laplace J. 2009. Comparative analysis of gene expression in brain, liver, skeletal muscles, and gills of zebrafish (Danio rerio) exposed to environmentally relevant waterborne uranium concentrations. Environmental Toxicology and Chemistry. 28(6):1271–1278. https://doi.org/10.1897/08-357.1
Ma M, Wang R, Xu L, Xu M, Liu S. 2020. Emerging health risks and underlying toxicological mechanisms of uranium contamination: Lessons from the past two decades. Environment International. 145:106107. https://doi.org/10.1016/j.envint.2020.106107
Pizzorno J. 2014. Glutathione! Integr Med (Encinitas). 13(1):8–12.
Proskuryakov SY a, Konoplyannikov AG, Gabai VL. 2003. Necrosis: a specific form of programmed cell death? Experimental Cell Research. 283(1):1–16. https://doi.org/10.1016/S0014-4827(02)00027-7
Pyle GG, Swanson SM, Lehmkuhl DM. 2002. Toxicity of uranium mine receiving waters to early life stage fathead minnows (Pimephales promelas) in the laboratory. Environmental Pollution. 116(2):243–255. https://doi.org/10.1016/S0269-7491(01)00130-0
Simon O, Mottin E, Geffroy B, Hinton T. 2011. Effects of dietary uranium on reproductive endpoints—fecundity, survival, reproductive success—of the fish Danio rerio. Environmental Toxicology and Chemistry. 30(1):220–225. https://doi.org/10.1002/etc.381
Song Y, Salbu B, Heier LS, Teien H-C, Lind O-C, Oughton D, Petersen K, Rosseland BO, Skipperud L, Tollefsen KE. 2012. Early stress responses in Atlantic salmon (Salmo salar) exposed to environmentally relevant concentrations of uranium. Aquatic Toxicology. 112–113:62–71. https://doi.org/10.1016/j.aquatox.2012.01.019
Song Y, Salbu B, Teien H-C, Sørlie Heier L, Olav Rosseland B, Høgåsen T, Erik Tollefsen K. 2014. Hepatic transcriptomic profiling reveals early toxicological mechanisms of uranium in Atlantic salmon (Salmo salar). BMC Genomics. 15(1):694. https://doi.org/10.1186/1471-2164-15-694