
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 37
Title
PPARα activation leading to hepatocellular adenomas and carcinomas in rodents
Short name
Graphical Representation
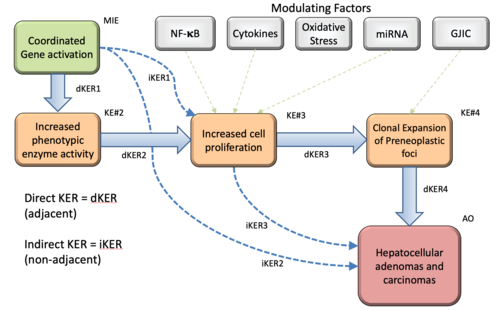
Point of Contact
Contributors
- Chris Corton
- Cataia Ives
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.17 | Under Development |
This AOP was last modified on May 26, 2024 20:39
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Activation, PPARα | December 28, 2020 12:48 |
| Increase, Phenotypic enzyme activity | December 28, 2020 12:32 |
| Increase, cell proliferation (hepatocytes) | January 06, 2021 16:21 |
| Increase, Clonal Expansion of Altered Hepatic Foci | January 06, 2021 16:15 |
| Increase, hepatocellular adenomas and carcinomas | December 26, 2020 10:09 |
| Activation, PPARα leads to Increase, Phenotypic enzyme activity | December 31, 2020 11:42 |
| Increase, Phenotypic enzyme activity leads to Increase, cell proliferation (hepatocytes) | December 31, 2020 13:00 |
| Increase, cell proliferation (hepatocytes) leads to Increase, Clonal Expansion of Altered Hepatic Foci | December 03, 2016 16:38 |
| Increase, Clonal Expansion of Altered Hepatic Foci leads to Increase, hepatocellular adenomas and carcinomas | December 03, 2016 16:38 |
| Activation, PPARα leads to Increase, cell proliferation (hepatocytes) | December 31, 2020 09:43 |
| Activation, PPARα leads to Increase, hepatocellular adenomas and carcinomas | December 31, 2020 09:46 |
| Increase, cell proliferation (hepatocytes) leads to Increase, hepatocellular adenomas and carcinomas | December 31, 2020 09:46 |
| pirinixic acid | November 29, 2016 18:42 |
| Clofibrate | November 29, 2016 18:42 |
| Bis(2-ethylhexyl) phthalate | November 29, 2016 18:42 |
| Nafenopin | November 29, 2016 18:42 |
| ciprofibrate | November 29, 2016 18:42 |
| Gemfibrozil | March 31, 2020 10:24 |
| PERFLUOROOCTANOIC ACID | November 29, 2016 18:42 |
| Bezafibrate | November 29, 2016 18:42 |
| Fenofibrate | November 29, 2016 18:42 |
Abstract
Several therapeutic agents and industrial chemicals induce liver tumors in rats and mice through the activation of the peroxisome proliferator-activated receptor alpha (PPARα). The molecular and cellular events by which PPARα activators induce rodent hepatocarcinogenesis have been extensively studied and elucidated. The weight of evidence relevant to the hypothesized AOP for PPARα activator-induced rodent hepatocarcinogenesis is summarized here. Chemical-specific and mechanistic data support concordance of temporal and dose–response relationships for the key events associated with many PPARα activators including a phthalate ester plasticizer di(2-ethylhexyl)phthalate (DEHP) and the drug gemfibrozil. The key events (KE) identified include the MIE – PPARα activation measured as a characteristic change in gene expression, KE2 – increased enzyme activation, characteristically those involved in lipid metabolism and cell cycle control, KE3 – increased cell proliferation, KE4 – selective clonal expansion of preneoplastic foci, and the AO – – increases in hepatocellular adenomas and carcinomas. Other biological factors modulate the effects of PPARα activators.These modulating events include increases in oxidative stress, activation of NF-kB, and inhibition of gap junction intercellular communication. The occurrence of hepatocellular adenomas and carcinomas is specific to mice and rats. The occurrence of the various KEs in hamsters, guinea pigs, cynomolgous monkeys are generally absent.
AOP Development Strategy
Context
During the 1970s, an increased incidence of hepatocellular adenomas and carcinomas was observed in rodents treated with a variety of seemingly disparate chemicals. The common effect was an increase in the number and size of peroxisomes and these substances were labeled ‘‘peroxisome proliferators’’ (Rao & Reddy, 1996). Peroxisomes are subcellular organelles involved in long chain fatty acid catabolism through the β-oxidation cycle (de Duve, 1996). Peroxisomes increase in number and/or size following exposure to substances that perturb fatty acid homeostasis. These substances include marketed pharmaceutical agents and drug candidates, phthalate ester plasticizers or their metabolites, herbicides , solvents and perfluorinated chemicals (Klaunig et al., 2003). Hepatocellular hypertrophy and hyperplasia, changes in apoptosis rates, Kupffer cell activation, and oxidative stress were also observed following chronic exposure of rats and mice to peroxisome proliferators (Corton, 2010).
The peroxisome proliferator-activated receptor α (PPARα) was identified after cloning from mouse DNA (Issemann & Green, 1990). PPARα along with the PPARb/d and PPARg subtypes are ligand-activated transcription factors with both DNA-binding and ligand-binding domain with variation in tissue distribution, expression during development, ligand specificity, and biological function. PPARα is expressed in metabolically active tissues, including the liver, kidney, brown fat and heart, which exhibit pleiotropic responses to peroxisome proliferators. The biological functions and role in chemical effects of PPARα has been facilitated by the PPARα-null mouse the experimental use of which revealed that a functional PPARα was required for the obseved phenotypic effects and led to the identification of the genes involved in lipid catabolism, lipid transport, peroxisome proliferation and hepatocellular adenomas and carcinomas (Corton, 2010; Lee et al., 1995).
Like other nuclear receptors, PPARα forms a heterodimer before translocating to the nucleus. with another nuclear receptor family member, retinoid X receptor (RXR), the receptor for 9-cis-retinoic acid. The PPARα-RXR heterodimer binds to peroxisome proliferator response elements (PPREs) usually found in the promoter or enhancer regions. The PPRE consensus sequence consists of the sequence 5’-AACT AGGTCA A AGGTCA-3’ with PPARα occupying the 5’ position. After agonist binding to PPARα, co-repressors dissociate from the complex leading to de-acetylation, chromatin remodeling to enable transcription (Escher & Wahli, 2000; Göttlicher et al., 1992; Moreno et al. 2010).
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 227 | Activation, PPARα | Activation, PPARα |
| KE | 1170 | Increase, Phenotypic enzyme activity | Increase, Phenotypic enzyme activity |
| KE | 716 | Increase, cell proliferation (hepatocytes) | Increase, cell proliferation (hepatocytes) |
| KE | 1171 | Increase, Clonal Expansion of Altered Hepatic Foci | Increase, Clonal Expansion of Altered Hepatic Foci |
| AO | 719 | Increase, hepatocellular adenomas and carcinomas | Increase, hepatocellular adenomas and carcinomas |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Activation, PPARα leads to Increase, Phenotypic enzyme activity | adjacent | High | High |
| Increase, Phenotypic enzyme activity leads to Increase, cell proliferation (hepatocytes) | adjacent | High | Moderate |
| Increase, cell proliferation (hepatocytes) leads to Increase, Clonal Expansion of Altered Hepatic Foci | adjacent | High | Moderate |
| Increase, Clonal Expansion of Altered Hepatic Foci leads to Increase, hepatocellular adenomas and carcinomas | adjacent | High | Moderate |
| Activation, PPARα leads to Increase, cell proliferation (hepatocytes) | non-adjacent | High | High |
| Activation, PPARα leads to Increase, hepatocellular adenomas and carcinomas | non-adjacent | High | High |
| Increase, cell proliferation (hepatocytes) leads to Increase, hepatocellular adenomas and carcinomas | non-adjacent | High | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male | High |
| Female | High |
Overall Assessment of the AOP
Molecular Initiating Event - PPARα activation
The MIE was characterized using two types of measurements: the earliest is the genomic changes representing the activation of PPARα; (Rooney et al., 2018; Corton et al., 2020; Hill et al., 2020; Lewis et al., 2020) Previously, increases in enzyme activity were used as a measure of PPARα activation . (Liu et al., 1996; David et al., 1999; Isenberg et al., 2000; Klaunig et al., 2003; Kondo et al., 2019)
Microarray profiling was used to show that alteration of gene expression following PPARα activation was almost completely abolished in PPARα-null mice s (Anderson et al., 2001, 2004a,b; Corton et al., 2004; Rosen et al.,2008a,b; Woods et al., 2007c; Ren et al., 2009, 2010; Rosen et al., 2008, 2010; Rosen et al., 2017). The genes that were dependent on PPARα were those involved in lipid homeostasis and the cell cycle.
The development of large gene expression databases TG-GATEs and DrugMatrix enabled that statistical analysis of whole genome expression profiles that form the basis of the genomic MIEs (Svoboda et al., 2019; Igarashi et al., 2015) From these data, A gene expression signature of 131 PPARα-dependent genes was built using microarray profiles from the livers of wild-type and PPARα-null mice. A quantitative measure of this expression signature is a measure of similarity/correlation between the PPARα signature and positive and negative test sets is provided by the Running Fisher test (Kupershmidt et al., 2010; Rooney et al., 2018; Corton et al., 2020).
Key Event #2 - Increased Phenotypic Enzyme Activity
Increased activity in acyl coenzyme A oxidase, peroxisomal beta-oxidase, carnitine acetyl transferase, catalase and others have been observed uniformly in rats and mice following PPARα activation (Corton et al. 2014; Cunningham et al. 2010; Klaunig et al. 2003; NTP, 2007).
Key Event #3 - Increased Cell Proliferation
Increased cell proliferation is observed following PPARα and is measured by increases in BrdU labeling index or liver weight. great deal of work has been conducted to identify the mechanistic events that lead to alterations in cell growth by PPARα activators. Early studies focused mainly on growth factors secreted from Kupffer cells are activated by PPARα activation and may secrete tumor necrosis factor α (TNFα), interleukin-1α (IL-1α), interleukin-1β (IL-1β) and likely other cytokines (Bojes et al., 1997; Rolfe et al.,1997 Holden et al., 2000). Cell proliferation following PPARα did not occur in vivo with pretreatment with antibodies to either TNFα or TNFα receptor 1 (TNFR1) nor in PPARα null mice (West et al., 1999; Bojes et al., 1997; Rolfe et al.,1997 Anderson et al., 2001; Lawrence et al., 2001b).
PPARα reduces expression of the miRNA let-7c in the liver that, in turn, reduces the expression of the c-Myc gene. The resulting increased c-Myc expression is likely a causal factor in increased cell proliferation following PPARα activation (Shah et al., 2007; Qu et al., 2014)
Key Event #4 - Clonal Expansion of Altered Hepatic Foci
ATPase-negative foci and basophilic foci were observed following occurrence of the MIE in rats (Marsman & Popp, 1994).
PPARα activators promote the growth of chemically- and spontaneously-induced lesions through enhanced cell replication (Cattley & Popp, 1989; Cattley et al., 1991; Isenberg et al., 1997; Marsman et al., 1988). ATPase-negative foci and GGT- weakly basophilic foci were observed following occurrence of the MIE in rats but GGT+ and GST-P+ foci were not observed (Grasl-Kraupp et al. 1993c; Marsman & Popp, 1994).
PPARα activators increase cell proliferation in AHF to about half-again that of normal cells measured by labeling index (Grasl-Kraupp et al. 1993b). Continued exposure to PPARα activators causes selective increases in DNA replication within these liver foci in comparison to normal hepatocytes (Isenberg et al., 1997).
Although, apoptosis was found to be increased in both normal cells and AHF by PPARα activation, the increase in foci cell proliferation was sufficient to outweigh the increase in focal apoptosis (Isenberg et al., 1997).
Adverse Outcome - Hepatocellular tumors in mice and rats
Increases in hepatocellular adenomas and carcinomas has been observed in both rats and mice in a dose-dependent fashion after treatment with PPARα activators. Corton et al. (2014) provide a summary of the dose-response data for di-2-ethylhexyl phthalate (DEHP). Similar increases have been observed after treatement with other PPARα activators. The section on Quantitative Understanding below presents some of these data.
Domain of Applicability
Sex Differences in PPARα activators
For some PPARα activators, differences in the carcinogenic effects can be observed in both rats and mice (Astill et al., 1996; Butala et al., 1997; Kluwe et al., 1985; Gold and Zeiger, 1997; Hollander and Wiegand, 1978; Malley et al., 1995; Shirasu 1987a, 1987b; U.S. EPA, 2000a, 2000b) These differences may reflect variation in the formation of a metabolite that activates PPARα.
PPARα Activation in the Fetus, Neonate and Adult
PPARα mRNA and protein has been detected in the fetuses of both rats and mice prior to birth (Balasubramaniyan et al. 2005; Beck et al. 1992). Assembly of peroxisomal proteins into peroxisomes and peroxisomal enzyme activity are detectable in late gestation Stefanini et al. 1989; Wilson et al., 1991). Catalase and palmitoyl CoA oxidase activities were first detected in the GD15 Wistar rat fetus (Cibelli et al., 1988).
Stefanini et al. (1995) found no statistically significant differences in numerical density or volume density of peroxisomes in livers of 14-, 21-, or 35-day F344 rat neonates and no differences between neonate groups and adult females. No differences were found between 14- or 21-day neonates or between these groups and adult F344 rats in peroxisomal β-oxidation; the numerical density and volume density of liver peroxisomes were also comparable among groups (Stefanini et al., 1999). No differences were reported for the specific volume density among 7-, 8.5-, 10-, 13-, or 17-day Wistar-derived rat neonates or compared with adults (Wiebel et al., 1969; Staubli et al., 1977).
Direct exposure of the neonate to PPARα agonists results in an increase in peroxisomal enzyme activities and an increase in the numerical density or volume of peroxisomes; the increases in these parameters are comparable to those observed in young adult or adult rats (Staubli et al., 1977; Dostal et al., 1987; Yamoto et al., 1996; Yu et al., 2001).
Species Differences in PPARα Activation
Whilst mice and rats are responsive to PPARα activator-induced liver cancer and associated responses, hamsters, guinea pigs, and primates, including humans, are less sensitive (Ashby et al., 1994; Bentley et al., 1993; Cattley et al., 1998; Doull et al., 1999).
In side-by-side assays human PPARα were less sensitive than rodent PPARα to chemical activation. Hypolipidemic agents and environmental chemicals activated rat or mouse PPARα with greather potency and greater efficacy than human PPARα (Takacs & Abbott, 2007; Maloney & Waxman, 1999; Lapinskas et al., 2005)
Essentiality of the Key Events
Transgenic or knockout animals can often provide a powerful counterfactual demonstration supporting the identification of a KE (Phillips and Goodman 2006; Simon et al. 2014) Such work demonstrates the necessity of a given KE studies show that preventing a given KE also prevents the occurrence of downstream KEs. Hence, the PPARα-null mouse has provided critical evidence identifying the MIE as well as subsequent downstream KEs.
Global gene expression profiling revealed that alteration of gene expression following PPARα-activation was almost completely abolished in PPARα-null mice at multiple time points (Anderson et al., 2004a,b; Corton et al., 2004; Li et al. 2018; Woods et al., 2007; Rosen et al., 2008. 2010, 2017; Sanderson et al. 2008; Ren et al., 2010; Wang et al. 2020). Following PPARα activation, wild-type mice exhibited increased hepatocyte proliferation compared to untreated controls and no increase was observed in PPARα-null mice (Peters et al., 1997, 1998; Valles et al., 2003; Laughter et al., 2004; Wolf et al., 2008). Apoptosis was suppressed in hepatocytes from wild-type mice but not in those from the knockouts (Hasmall et al., 2000). Finally, chronic treatment with PPARα activators resulted in 100% incidence of hepatocellular tumor in wild-type mice but the knockouts remained unaffected (Peters et al., 1997; Hays et al., 2005).
KE#2, increased enzyme activity, KE#3, increased cell proliferation and KE#4, clonal expansion of preneoplastic lesions have been recognized as KEs in many carcinogenic modes of action and are not specific to PPARα activation.
Finally, in an initiation-promotion study, 25% of wild-type mice receiving only diethylnitrosamine (DEN) developed tumors (25%) whereas 63% of wild-type mice receiving both DEN and a PPARα activator developed tumors. (Glauert et al., 2006).
Evidence Assessment
Evidence supporting PPARα activation as the MIE
- All PPARα activators exhibit a characteristic genomic signature that was used to characterize the MIE (Corton et al. 2020; Hill et al. 2020; Rooney et al. (2018).
- The potency of PPARα activation is roughly proportional to the potency of the chemical as an inducer of the liver tumor response (Klaunig et al., 2003).
- Characteristic gene expression changes do not occur in PPARα-null mice (Anderson et al., 2004a,b; Corton et al., 2004; Rosen et al.,2008, 2017; Ren et al. 2009, 2010; Woods et al., 2007c).
Evidence supporing KE2 - Increased enzyme activity
- Increased activity of lipid metabolizing enzymes following PPARα has been repeated demonstrated (Amacher et al 1997; Klaunig et al. 2003; Belury et al. 1998; Corton et al. 2000).
Weaknesses in the evidence
- Hepatocytes from PPARα-null mice can respond PPARα activators if adjacent to wild-type hepatocytes (Weglarz & Sandgren, 2004).
Evidence supporting KE3 - Increased Cell Proliferation
- PPARα activators increase cell proliferation.
- Increases in cell proliferation after exposure to a number of PPARα activators were abolished in PPARα-null mice.
Evidence supporting KE4 - Clonal Expansion of altered foci
- Clonal expansion is consistently induced in mice and rats by diverse PPARα activators (Marsman & Popp 1994).
- Clonal expansion and the appearance of foci and tumors by PPARα activators were not observed in PPARα-null mice after exposure to PPARα activators (Hays et al. 2005; Peters et al. 1997).
Evidence supporting the AO - Hepatocellular Adenomas and Carcinomas
- Chronic treatment with PPARα activators produced hepatocellular neoplasia in 100% of wild-type mice and not in PPARα-null mice (Hays et al., 2005; Peters et al., 1997).
Strength and Specificity of the Evidence
The activation of PPARα is specific this AOP, (AOP 37) whereas the downstream key events are common to the neoplastic process in the rodent liver, e.g AOP 107 and AOP 41. Data supporting each key event were determined to be strong in that several studies support that key event as part of the AOP using multiple PPARα activators from multiple laboratories with no or limited evidence of contradiction.
Known Modulating Factors
Quantitative Understanding
To understand the dose-response of the PPARα genomic MIE over time, the level of MIE activation of seven most active substances from the TG-GATEs database were fit to a gain-loss model used a dose range normalized over the range of zero to one at each of the time-points, i.e., 3, 6 and 9 hours and 1, 3, 7, 14 and 28 days. The gain-loss model is shown below as Eq. 1 (Watt and Judson, 2018).

The gain-loss model was chosen because the response variable -log(p-value) from the Running Fisher test did not increase monotonically with dose. This model provided a flexible means of fitting all dose responses for the PPARα agonists in the TG-GATES database at all times. The fits were compared to the data and both visual comparisons and calculated AICs revealed very good fits. The fitted curves were plotted against both dose and time in both 2D and 3D surface plots.

As noted, for all substances, MIE activation does not rise monotonically over dose or time. These fluctuations are likely due to variations in cofactor availability or access to the site of transcription (Gaillard et al., 2006; Koppen et al., 2009; Kupershmidt et al., 2010; Ong et al., 2010; Chow et al., 2011; De Vos et al., 2011; Simon et al., 2015).
A more extensive analysis was conducted for the three PPARalpha agonists shown above to understand the KERs between the genomic MIE and downstream KEs. For all these analyses, the MIE response at 7 days was chosen as the most representative due to the generally steeper response. The 7 day response was also steepest for the PPARalpha activators fenofibrate, benzbromarone, benzodiarone and simvastatin (not shown).
Relationship between the Genomic MIE and Tumor Frequency
To assess this relationship, the dose-response of all three PPARalpha activation at all times measured in the TG-GATES database were plotted along with the corresponding tumor frequency.
The apparent potency of Gemfibrozil (middle plot) increased with time. This trend was weaker for Clofibrate. For gemfibrozil, the potency was higher than the other two PPARalpha activators at all time points. For all chemicals, the efficacy of the MIE response at the 7-day time point is generally proportional to the tumor frequency at the highest dose and is apparent in the table below. The value for efficacy was tp in the gain-loss equation
| Time point | Clofibrate | Gemfibrozil | WY-14,643 |
|---|---|---|---|
| 3h | 6.36 | 3.73 | 37.7 |
| 6h | 11.4 | 4.49 | 42.8 |
| 9h | 18.9 | 24.6 | 48.6 |
| 24h | 20.8 | 29.7 | 51.4 |
| 3d | 20.7 | 19.4 | 50.2 |
| 7d | 23.6 | 20.3 | 62.0 |
| 14d | 23.2 | 35.0 | 54.7 |
| 28d | 40.4 | 18.4 | 53.5 |
| Max. Tumor Freq. | 0.26 | 0.36 | 1.0 |
KERs for PPARalpha Activation by Clofibrate
The figure below displays the analysis.
The KER between the MIE and the AO is revealed by plots B and the bottom panel in D: tumors don't occur until the MIE is sustained at an MIE level of between 10 and 15. Fatty acid CoA oxidase is a lipid metabolizing enzyme and thus a measure of KE#1 and rises more steeply in female rats. The two responses measuring KE#2 also show male-female differences. Labeling index is a measure of cell proliferation and rises more steeply than in females with increasing MIE levels. Liver weight, another measure of KE#2 changes little in females and more in males, consistent with labeling index, which is a more direct measure of cell proliferation. The tumor response in both males and females trends upward somewhere between and MIE level of 10 to 15. Specifying a response level related to tumors would require a bioassay with more doses.
KERs for PPARalpha Activation by Gemfibrozil
The figure below displays the analysis.
The KER between the MIE and the AO is revealed by plots B and the bottom panel in D: the MIE reaches a plateau at an MIE level of 20 corresponding to a dose of 30 mg/kg/d; an increase in the frequency of neoplastic nodules to 4% occurs at this dose and MIE level; an increase in the frequency of carcinomas to 8% also occurs at this dose level. At a dose of 100 mg/kg/d corresponding to the plateau value of the MIE at 20, the frequency of neoplastic nodules rises to 36% and for carcinomas to 10%.
The steepness in the rise of the MIE between doses of 0 and 30 mg/kg/d occurs along with any level of increase in downstream KEs. In the four graphs in D, Acyl CoA oxidase is a lipid metabolizing enzyme and thus a measure of KE#1 and continues to rise at the plateau level of the MIE. Labeling index as a measure of KE#2 continued to rise from 4% in controls to 26% at a dose of 510 mg/kg/d. At the highest dose of 1300 mg/kg/d, the MIE was at the plateau level of 20 and Labeling Index was 7%. Relative Liver weight, a less direct measure of KE#2, rose to between 55 and 60 mg liver / g BW and remained at the level at the three highest doses with the same MIE level of 20 (Cunningham et al. 2010)
The tumor response was evident only in male rats. (Fitzgerald et al. 1981). Specifying a range of the MIE corresponding to an increase in tumors because of the apparently steep dose-response curve of the MIE [B], and the lack of any doses between 0 and 30 in either the TG-GATES database or the bioassay (Corton et al. 2020; Fitzgerald et al. 1981; Hill et al. 2020; Rooney et al. 2018).
KERs for PPARalpha Activation by WY-14,643
The figure below displays the analysis.
The combined tumor response was obtained from the carcdb at https://carcdb.lhasalimited.org/carcdb-frontend/. Because the studies used no more than three dose levels (and a single dose in some), the results from four of the studies were combined. The table below shows the dose response and primary sources.
|
Dose (mg/kg/d) |
N |
Incidence |
ftumor |
LCL |
UCL |
Sources |
|
0 |
156 |
0 |
0.00 |
0.00 |
0.02 |
Hayashi et al. 1994; Lalwani et al. 1981; Marsman & Popp 1984; Reddy et al. 1979 |
|
14.2 |
20 |
3 |
0.15 |
0.03 |
0.38 |
|
|
20 |
20 |
11 |
0.55 |
0.32 |
0.77 |
|
|
40 |
37 |
35 |
0.95 |
0.82 |
0.99 |
|
|
80 |
14 |
14 |
1.00 |
0.77 |
1.00 |
The MIE rises steeply and falls less steeply with increasing dose (A). The KER between the MIE and the AO is revealed by plots B and the bottom panel in D: the MIE reaches a plateau at an MIE level of over 30 corresponding to a dose of 10 mg/kg/d; an increase in the frequency of combined liver tumors to 15% occurs a dose of 14 mg/kg/d. Tumor frequency increases monotonically with dose and the MIE falls slightly to about 26 over the dose range of 10 to 100.
The KER between the MIE and the AO is revealed by plots B and the bottom panel in D: the MIE reaches a plateau at an MIE level of over 30 corresponding to a dose of 10 mg/kg/d; an increase in the frequency of combined liver tumors to 15% occurs a dose of 14 mg/kg/d. Tumor frequency increases monotonically with dose and the MIE falls slightly to about 26 over the dose range of 10 to 100. At a dose of 100 mg/kg/d corresponding to the plateau value of the MIE at 20, the frequency of neoplastic nodules rises to 36% and for carcinomas to 10%.
Similar to the KERs for Gemfibrozil, increases in KE#1 were not observed until the peak of the MIE response and continued as dose continued to rise (D, upper plot).
Considerations for Potential Applications of the AOP (optional)
This AOP has no potential application for human risk assessment save to provide a case example of the mode of action of a type of non-genotoxic carcinogen (Wolf et al. 2019; Doe et al. 2019; Cohen et al. 2019). The application to ecological risk assessment is also unlikely. Although some PPARα activators occur in the enviroment, e.g. perfluorinated chemicals, the levels may not be sufficient to produce effects in wild rodent populations.
Several large retrospective epidemiological studies observed no elevated risk of death from liver cancer associated with chronic treatment with PPARα activatorsr (Peters et al., 2005). Over a decade of chronic use of these pharmaceuticals was not associated with liver tumors in large human cohorts (Frick et al., 1987; Huttunen et al., 1994).
PPARα activators do not induce cell proliferation or suppress apoptosis in human hepatocytes cultured in vitro (Goll et al., 1999; Hasmall et al., 1999, 2000b; Perrone et al., 1998; Williams & Perrone, 1995). In non-human primates, PPARα activators did not induce cell proliferation in vitro or in vivo (Doull et al., 1999).
In summary, humans are not responsive to the effects of PPARα activators as are mice and rats.
References
Amacher, D. E., Beck, R., Schomaker, S. J., & Kenny, C. V. (1997). Hepatic microsomal enzyme induction, beta-oxidation, and cell proliferation following administration of clofibrate, gemfibrozil, or bezafibrate in the CD rat. Toxicol Appl Pharmacol, 142(1), 143-150. https://doi.org/10.1006/taap.1996.8007
Anderson, S. P., Dunn, C., Laughter, A., Yoon, L., Swanson, C., Stulnig, T. M., Steffensen, K. R., Chandraratna, R. A., Gustafsson, J. A., & Corton, J. C. (2004a). Overlapping transcriptional programs regulated by the nuclear receptors peroxisome proliferator-activated receptor alpha, retinoid X receptor, and liver X receptor in mouse liver. Mol Pharmacol, 66(6), 1440-1452. https://doi.org/10.1124/mol.104.005496
Anderson, S. P., Dunn, C. S., Cattley, R. C., & Corton, J. C. (2001a). Hepatocellular proliferation in response to a peroxisome proliferator does not require TNFalpha signaling. Carcinogenesis, 22(11), 1843-1851. https://doi.org/10.1093/carcin/22.11.1843
Anderson, S. P., Dunn, C. S., Cattley, R. C., & Corton, J. C. (2001b). Hepatocellular proliferation in response to a peroxisome proliferator does not require TNFalpha signaling. Carcinogenesis, 22(11), 1843-1851. https://doi.org/10.1093/carcin/22.11.1843
Anderson, S. P., Howroyd, P., Liu, J., Qian, X., Bahnemann, R., Swanson, C., Kwak, M. K., Kensler, T. W., & Corton, J. C. (2004b). The transcriptional response to a peroxisome proliferator-activated receptor alpha agonist includes increased expression of proteome maintenance genes. J Biol Chem, 279(50), 52390-52398. https://doi.org/10.1074/jbc.M409347200
Ashby, J., Brady, A., Elcombe, C. R., Elliott, B. M., Ishmael, J., Odum, J., Tugwood, J. D., Kettle, S., & Purchase, I. F. (1994). Mechanistically-based human hazard assessment of peroxisome proliferator-induced hepatocarcinogenesis. Hum Exp Toxicol, 13 Suppl 2, S1-117. https://doi.org/10.1177/096032719401300201
Balasubramaniyan, N., Shahid, M., Suchy, F. J., & Ananthanarayanan, M. (2005). Multiple mechanisms of ontogenic regulation of nuclear receptors during rat liver development. Am J Physiol Gastrointest Liver Physiol, 288(2), G251-60. https://doi.org/10.1152/ajpgi.00351.2004
Beck, F., Plummer, S., Senior, P. V., Byrne, S., Green, S., & Brammar, W. J. (1992). The ontogeny of peroxisome-proliferator-activated receptor gene expression in the mouse and rat. Proc Biol Sci, 247(1319), 83-87. https://doi.org/10.1098/rspb.1992.0012
Belury, M. A., Moya-Camarena, S. Y., Sun, H., Snyder, E., Davis, J. W., Cunningham, M. L., & Vanden Heuvel, J. P. (1998). Comparison of dose-response relationships for induction of lipid metabolizing and growth regulatory genes by peroxisome proliferators in rat liver. Toxicol Appl Pharmacol, 151(2), 254-261. https://doi.org/10.1006/taap.1998.8443
Bentley, P., Calder, I., Elcombe, C., Grasso, P., Stringer, D., & Wiegand, H. J. (1993). Hepatic peroxisome proliferation in rodents and its significance for humans. Food Chem Toxicol, 31(11), 857-907. https://doi.org/10.1016/0278-6915(93)90225-n
Bojes, H. K., Germolec, D. R., Simeonova, P., Bruccoleri, A., Schoonhoven, R., Luster, M. I., & Thurman, R. G. (1997). Antibodies to tumor necrosis factor alpha prevent increases in cell replication in liver due to the potent peroxisome proliferator, WY-14,643. Carcinogenesis, 18(4), 669-674. https://doi.org/10.1093/carcin/18.4.669
Cattley, R. C., DeLuca, J., Elcombe, C., Fenner-Crisp, P., Lake, B. G., Marsman, D. S., Pastoor, T. A., Popp, J. A., Robinson, D. E., Schwetz, B., Tugwood, J., & Wahli, W. (1998). Do peroxisome proliferating compounds pose a hepatocarcinogenic hazard to humans. Regul Toxicol Pharmacol, 27(1 Pt 1), 47-60. https://www.ncbi.nlm.nih.gov/pubmed/9629596
Cattley, R. C., Marsman, D. S., & Popp, J. A. (1991). Age-related susceptibility to the carcinogenic effect of the peroxisome proliferator WY-14,643 in rat liver. Carcinogenesis, 12(3), 469-473. https://doi.org/10.1093/carcin/12.3.469
Cattley, R. C., & Popp, J. A. (1989). Differences between the promoting activities of the peroxisome proliferator WY-14,643 and phenobarbital in rat liver. Cancer Res, 49(12), 3246-3251. https://pubmed.ncbi.nlm.nih.gov/2566380
Chow, C. C., Ong, K. M., Dougherty, E. J., & Simons, S. S. (2011). Inferring mechanisms from dose-response curves. Methods Enzymol, 487, 465-483. https://doi.org/10.1016/B978-0-12-381270-4.00016-0
Corton, J. C., Apte, U., Anderson, S. P., Limaye, P., Yoon, L., Latendresse, J., Dunn, C., Everitt, J. I., Voss, K. A., Swanson, C., Kimbrough, C., Wong, J. S., Gill, S. S., Chandraratna, R. A., Kwak, M. K., Kensler, T. W., Stulnig, T. M., Steffensen, K. R., Gustafsson, J. A., . . . Mehendale, H. M. (2004). Mimetics of caloric restriction include agonists of lipid-activated nuclear receptors. J Biol Chem, 279(44), 46204-46212. https://doi.org/10.1074/jbc.M406739200
Corton, J. C., Cunningham, M. L., Hummer, T. B., Lau, C, Meek, B, Peters, JM, Popp, JA, Rhomberg, L, Seed, J., & Klaunig, J. E. (2014). Mode of action framework analysis for receptor-mediated toxicity: The peroxisome proliferator-activated receptor alpha (PPARα) as a case study. Crit Rev Toxicol, 44(1), 1-49. https://doi.org/10.3109/10408444.2013.835784
Corton, J. C., Hill, T., Sutherland, J. J., Stevens, J. L., & Rooney, J. (2020). A Set of Six Gene Expression Biomarkers Identify Rat Liver Tumorigens in Short-Term Assays. Toxicol Sci. https://doi.org/10.1093/toxsci/kfaa101
Corton, J. C., Lapinskas, P. J., & Gonzalez, F. J. (2000). Central role of PPARalpha in the mechanism of action of hepatocarcinogenic peroxisome proliferators. Mutat Res, 448(2), 139-151. https://doi.org/10.1016/s0027-5107(99)00232-8
Corton JC. (2010). Mode of action analysis and human relevance of liver tumors induced by PPARα activation. In: Hsu C-H, Stedeford T, eds. Cancer risk assessment: chemical carcinogenesis, hazard evaluation, and risk quantification. Hoboken (NJ): John Wiley & Sons, Inc, 438—81
Cunningham, M. L., Collins, B. J., Hejtmancik, M. R., Herbert, R. A., Travlos, G. S., Vallant, M. K., & Stout, M. D. (2010). Effects of the PPARα Agonist and Widely Used Antihyperlipidemic Drug Gemfibrozil on Hepatic Toxicity and Lipid Metabolism. PPAR Res, 2010. https://doi.org/10.1155/2010/681963
David, R. M., Moore, M. R., Cifone, M. A., Finney, D. C., & Guest, D. (1999). Chronic peroxisome proliferation and hepatomegaly associated with the hepatocellular tumorigenesis of di(2-ethylhexyl)phthalate and the effects of recovery. Toxicol Sci, 50(2), 195-205.https://doi.org/10.1093/toxsci/50.2.195
de Duve, C. (1996). The peroxisome in retrospect. Ann N Y Acad Sci, 804, 1-10. https://doi.org/10.1111/j.1749-6632.1996.tb18603.x
De Vos, D., Bruggeman, F. J., Westerhoff, H. V., & Bakker, B. M. (2011). How molecular competition influences fluxes in gene expression networks. PLoS One, 6(12), e28494. https://doi.org/10.1371/journal.pone.0028494
Dostal, L. A., Jenkins, W. L., & Schwetz, B. A. (1987). Hepatic peroxisome proliferation and hypolipidemic effects of di(2-ethylhexyl)phthalate in neonatal and adult rats. Toxicol Appl Pharmacol, 87(1), 81-90. https://doi.org/10.1016/0041-008x(87)90086-x
Doull, J., Cattley, R., Elcombe, C., Lake, B. G., Swenberg, J., Wilkinson, C., Williams, G., & van Gemert, M. (1999). A cancer risk assessment of di(2-ethylhexyl)phthalate: application of the new U.S. EPA Risk Assessment Guidelines. Regul Toxicol Pharmacol, 29(3), 327-357. https://doi.org/10.1006/rtph.1999.1296
Escher, P., & Wahli, W. (2000). Peroxisome proliferator-activated receptors: insight into multiple cellular functions. Mutat Res, 448(2), 121-138. https://doi.org/10.1016/s0027-5107(99)00231-6
Fitzgerald, J. E., Sanyer, J. L., Schardein, J. L., Lake, R. S., McGuire, E. J., & de la Iglesia, F. A. (1981). Carcinogen bioassay and mutagenicity studies with the hypolipidemic agent gemfibrozil. J Natl Cancer Inst, 67(5), 1105-1116. https://pubmed.ncbi.nlm.nih.gov/7029098
Gaillard, S., Grasfeder, L. L., Haeffele, C. L., Lobenhofer, E. K., Chu, T. M., Wolfinger, R., Kazmin, D., Koves, T. R., Muoio, D. M., Chang, C. Y., & McDonnell, D. P. (2006). Receptor-selective coactivators as tools to define the biology of specific receptor-coactivator pairs. Mol Cell, 24(5), 797-803. https://doi.org/10.1016/j.molcel.2006.10.012
Glauert, H. P., Eyigor, A., Tharappel, J. C., Cooper, S., Lee, E. Y., & Spear, B. T. (2006). Inhibition of hepatocarcinogenesis by the deletion of the p50 subunit of NF-kappaB in mice administered the peroxisome proliferator Wy-14,643. Toxicol Sci, 90(2), 331-336. https://doi.org/10.1093/toxsci/kfj116
Göttlicher, M., Widmark, E., Li, Q., & Gustafsson, J. A. (1992). Fatty acids activate a chimera of the clofibric acid-activated receptor and the glucocorticoid receptor. Proc Natl Acad Sci U S A, 89(10), 4653-4657. https://doi.org/10.1073/pnas.89.10.4653
Grasl-Kraupp, B., Huber, W., Timmermann-Trosiener, I., & Schulte-Hermann, R. (1993a). Peroxisomal enzyme induction uncoupled from enhanced DNA synthesis in putative preneoplastic liver foci of rats treated with a single dose of the peroxisome proliferator nafenopin. Carcinogenesis, 14(11), 2435-2437. https://doi.org/10.1093/carcin/14.11.2435
Grasl-Kraupp, B., Waldhör, T., Huber, W., & Schulte-Hermann, R. (1993b). Glutathione S-transferase isoenzyme patterns in different subtypes of enzyme-altered rat liver foci treated with the peroxisome proliferator nafenopin or with phenobarbital. Carcinogenesis, 14(11), 2407-2412. https://doi.org/10.1093/carcin/14.11.2407
Hartig, F., Stegmeier, K., Hebold, G., Özel, M., & Fahimi, H. D. (1982). Study of liver enzymes: peroxisome proliferation and tumor rates in rats at the end of carcinogenicity studies with bezafibrate and clofibrate. Annals of the New York Academy of Sciences, 386(1), 464-467. https://nyaspubs.onlinelibrary.wiley.com/doi/abs/10.1111/j.1749-6632.1982.tb21453.x
Hasmall, S. C., James, N. H., Macdonald, N., Gonzalez, F. J., Peters, J. M., & Roberts, R. A. (2000). Suppression of mouse hepatocyte apoptosis by peroxisome proliferators: role of PPARalpha and TNFalpha. Mutat Res, 448(2), 193-200. https://doi.org/10.1016/s0027-5107(99)00236-5
Hayashi, F., Tamura, H., Yamada, J., Kasai, H., & Suga, T. (1994). Characteristics of the hepatocarcinogenesis caused by dehydroepiandrosterone, a peroxisome proliferator, in male F-344 rats. Carcinogenesis, 15(10), 2215-2219. https://doi.org/10.1093/carcin/15.10.2215
Hays, T., Rusyn, I., Burns, A. M., Kennett, M. J., Ward, J. M., Gonzalez, F. J., & Peters, J. M. (2005). Role of peroxisome proliferator-activated receptor-alpha (PPARalpha) in bezafibrate-induced hepatocarcinogenesis and cholestasis. Carcinogenesis, 26(1), 219-227. https://doi.org/10.1093/carcin/bgh285
Hill, T., Rooney, J., Abedini, J., El-Masri, H., Wood, C. E., & Corton, J. C. (2020). Gene Expression Thresholds Derived From Short-Term Exposures Identify Rat Liver Tumorigens. Toxicol Sci. https://doi.org/10.1093/toxsci/kfaa102
Holden, P. R., Hasmall, S. C., James, N. H., West, D. R., Brindle, R. D., Gonzalez, F. J., Peters, J. M., & Roberts, R. A. (2000). Tumour necrosis factor alpha (TNFalpha): role in suppression of apoptosis by the peroxisome proliferator nafenopin. Cell Mol Biol (Noisy-le-grand), 46(1), 29-39. https://pubmed.ncbi.nlm.nih.gov/10726969
Igarashi, Y., Nakatsu, N., Yamashita, T., Ono, A., Ohno, Y., Urushidani, T., & Yamada, H. (2015). Open TG-GATEs: a large-scale toxicogenomics database. Nucleic Acids Res, 43(Database issue), D921-7. https://doi.org/10.1093/nar/gku955
Isenberg, J. S., Kamendulis, L. M., Smith, J. H., Ackley, D. C., Pugh, G., Lington, A. W., & Klaunig, J. E. (2000). Effects of Di-2-ethylhexyl phthalate (DEHP) on gap-junctional intercellular communication (GJIC), DNA synthesis, and peroxisomal beta oxidation (PBOX) in rat, mouse, and hamster liver. Toxicol Sci, 56(1), 73-85. https://doi.org/10.1093/toxsci/56.1.73
Isenberg, J. S., Kolaja, K. L., Ayoubi, S. A., Watkins, J. B., & Klaunig, J. E. (1997). Inhibition of WY-14,643 induced hepatic lesion growth in mice by rotenone. Carcinogenesis, 18(8), 1511-1519. https://doi.org/10.1093/carcin/18.8.1511
Issemann, I., & Green, S. (1990). Activation of a member of the steroid hormone receptor superfamily by peroxisome proliferators. Nature, 347(6294), 645-650. https://doi.org/10.1038/347645a0
Klaunig, J. E., Babich, M. A., Baetcke, K. P., Cook, J. C., Corton, J. C., David, R. M., DeLuca, J. G., Lai, D. Y., McKee, R. H., Peters, J. M., Roberts, R. A., & Fenner-Crisp, P. A. (2003). PPARalpha agonist-induced rodent tumors: modes of action and human relevance. Crit Rev Toxicol, 33(6), 655-780. https://doi.org/10.1080/713608372
Kondo, M., Miyata, K., Nagahori, H., Sumida, K., Osimitz, T. G., Cohen, S. M., Lake, B. G., & Yamada, T. (2019). Involvement of Peroxisome Proliferator-Activated Receptor-Alpha in Liver Tumor Production by Permethrin in the Female Mouse. Toxicol Sci, 168(2), 572-596. https://doi.org/10.1093/toxsci/kfz012
Koppen, A., Houtman, R., Pijnenburg, D., Jeninga, E. H., Ruijtenbeek, R., & Kalkhoven, E. (2009). Nuclear receptor-coregulator interaction profiling identifies TRIP3 as a novel peroxisome proliferator-activated receptor gamma cofactor. Mol Cell Proteomics, 8(10), 2212-2226. https://doi.org/10.1074/mcp.M900209-MCP200
Kupershmidt, I., Su, Q. J., Grewal, A., Sundaresh, S., Halperin, I., Flynn, J., Shekar, M., Wang, H., Park, J., Cui, W., Wall, G. D., Wisotzkey, R., Alag, S., Akhtari, S., & Ronaghi, M. (2010). Ontology-based meta-analysis of global collections of high-throughput public data. PLoS One, 5(9). https://doi.org/10.1371/journal.pone.0013066
Lalwani, N. D., Reddy, M. K., Qureshi, S. A., & Reddy, J. K. (1981). Development of hepatocellular carcinomas and increased peroxisomal fatty acid beta-oxidation in rats fed [4-chloro-6-(2,3-xylidino)-2-pyrimidinylthio] acetic acid (Wy-14,643) in the semipurified diet. Carcinogenesis, 2(7), 645-650. https://doi.org/10.1093/carcin/2.7.645
Lapinskas, P. J., Brown, S., Leesnitzer, L. M., Blanchard, S., Swanson, C., Cattley, R. C., & Corton, J. C. (2005). Role of PPARalpha in mediating the effects of phthalates and metabolites in the liver. Toxicology, 207(1), 149-163. https://doi.org/10.1016/j.tox.2004.09.008
Laughter, A. R., Dunn, C. S., Swanson, C. L., Howroyd, P., Cattley, R. C., & Corton, J. C. (2004). Role of the peroxisome proliferator-activated receptor alpha (PPARalpha) in responses to trichloroethylene and metabolites, trichloroacetate and dichloroacetate in mouse liver. Toxicology, 203(1-3), 83-98. https://doi.org/10.1016/j.tox.2004.06.014
Lawrence, J. W., Wollenberg, G. K., & DeLuca, J. G. (2001). Tumor necrosis factor alpha is not required for WY14,643-induced cell proliferation. Carcinogenesis, 22(3), 381-386. https://doi.org/10.1093/carcin/22.3.381
Lee, S. S., Pineau, T., Drago, J., Lee, E. J., Owens, J. W., Kroetz, D. L., Fernandez-Salguero, P. M., Westphal, H., & Gonzalez, F. J. (1995). Targeted disruption of the alpha isoform of the peroxisome proliferator-activated receptor gene in mice results in abolishment of the pleiotropic effects of peroxisome proliferators. Mol Cell Biol, 15(6), 3012-3022. https://doi.org/10.1128/mcb.15.6.3012
Lewis, R. W., Hill, T., & Corton, J. C. (2020). A set of six Gene expression biomarkers and their thresholds identify rat liver tumorigens in short-term assays. Toxicology, 443, 152547. https://doi.org/10.1016/j.tox.2020.152547
Li, G., Brocker, C. N., Xie, C., Yan, T., Noguchi, A., Krausz, K. W., Xiang, R., & Gonzalez, F. J. (2018). Hepatic peroxisome proliferator-activated receptor alpha mediates the major metabolic effects of Wy-14643. J Gastroenterol Hepatol, 33(5), 1138-1145. https://doi.org/10.1111/jgh.14046
Liu, M. H., Li, J., Shen, P., Husna, B., Tai, E. S., & Yong, E. L. (2008). A natural polymorphism in peroxisome proliferator-activated receptor-alpha hinge region attenuates transcription due to defective release of nuclear receptor corepressor from chromatin. Mol Endocrinol, 22(5), 1078-1092. https://doi.org/10.1210/me.2007-0547
Maloney, E. K., & Waxman, D. J. (1999). trans-Activation of PPARalpha and PPARgamma by structurally diverse environmental chemicals. Toxicol Appl Pharmacol, 161(2), 209-218. https://doi.org/10.1006/taap.1999.8809
Marsman, D. S., Cattley, R. C., Conway, J. G., & Popp, J. A. (1988). Relationship of hepatic peroxisome proliferation and replicative DNA synthesis to the hepatocarcinogenicity of the peroxisome proliferators di(2-ethylhexyl)phthalate and [4-chloro-6-(2,3-xylidino)-2-pyrimidinylthio]acetic acid (Wy-14,643) in rats. Cancer Res, 48(23), 6739-6744. https://pubmed.ncbi.nlm.nih.gov/3180084
Marsman, D. S., & Popp, J. A. (1994). Biological potential of basophilic hepatocellular foci and hepatic adenoma induced by the peroxisome proliferator, Wy-14,643. Carcinogenesis, 15(1), 111-117. https://doi.org/10.1093/carcin/15.1.111
Moreno, M., Lombardi, A., Silvestri, E., Senese, R., Cioffi, F., Goglia, F., Lanni, A., & de Lange, P. (2010). PPARs: Nuclear Receptors Controlled by, and Controlling, Nutrient Handling through Nuclear and Cytosolic Signaling. PPAR Res, 2010. https://doi.org/10.1155/2010/435689
Program, N. T. (2007). Toxicity studies of WY-14,643 (CAS No. 50892-23-4) administered in feed to male Sprague-Dawley rats, B6C3F1 mice, and Syrian hamsters. Toxic Rep Ser, 62), 1-136. https://pubmed.ncbi.nlm.nih.gov/24743700
Ong, K. M., Blackford, J. A., Kagan, B. L., Simons, S. S., & Chow, C. C. (2010). A theoretical framework for gene induction and experimental comparisons. Proc Natl Acad Sci U S A, 107(15), 7107-7112. https://doi.org/10.1073/pnas.0911095107
Peters, J. M., Cattley, R. C., & Gonzalez, F. J. (1997). Role of PPAR alpha in the mechanism of action of the nongenotoxic carcinogen and peroxisome proliferator Wy-14,643. Carcinogenesis, 18(11), 2029-2033. https://doi.org/10.1093/carcin/18.11.2029
Phillips, C. V., & Goodman, K. J. (2006). Causal criteria and counterfactuals; nothing more (or less) than scientific common sense. Emerg Themes Epidemiol, 3, 5. https://doi.org/10.1186/1742-7622-3-5
Qu, A., Jiang, C., Cai, Y., Kim, J. H., Tanaka, N., Ward, J. M., Shah, Y. M., & Gonzalez, F. J. (2014). Role of Myc in hepatocellular proliferation and hepatocarcinogenesis. J Hepatol, 60(2), 331-338. https://doi.org/10.1016/j.jhep.2013.09.024
Rao, M. S., & Reddy, J. K. (1996). Hepatocarcinogenesis of peroxisome proliferators. Ann N Y Acad Sci, 804, 573-587. https://doi.org/10.1111/j.1749-6632.1996.tb18646.x
Reddy, J. K., Rao, M. S., Azarnoff, D. L., & Sell, S. (1979). Mitogenic and carcinogenic effects of a hypolipidemic peroxisome proliferator, [4-chloro-6-(2,3-xylidino)-2-pyrimidinylthio]acetic acid (Wy-14, 643), in rat and mouse liver. Cancer Res, 39(1), 152-161. https://pubmed.ncbi.nlm.nih.gov/83907
Ren, H., Aleksunes, L. M., Wood, C., Vallanat, B., George, M. H., Klaassen, C. D., & Corton, J. C. (2010). Characterization of peroxisome proliferator-activated receptor alpha--independent effects of PPARalpha activators in the rodent liver: di-(2-ethylhexyl) phthalate also activates the constitutive-activated receptor. Toxicol Sci, 113(1), 45-59. https://doi.org/10.1093/toxsci/kfp251
Ren, H., Vallanat, B., Nelson, D. M., Yeung, L. W. Y., Guruge, K. S., Lam, P. K. S., Lehman-McKeeman, L. D., & Corton, J. C. (2009). Evidence for the involvement of xenobiotic-responsive nuclear receptors in transcriptional effects upon perfluoroalkyl acid exposure in diverse species. Reprod Toxicol, 27(3-4), 266-277. https://doi.org/10.1016/j.reprotox.2008.12.011
Rolfe, M., James, N. H., & Roberts, R. A. (1997). Tumour necrosis factor alpha (TNF alpha) suppresses apoptosis and induces DNA synthesis in rodent hepatocytes: a mediator of the hepatocarcinogenicity of peroxisome proliferators. Carcinogenesis, 18(11), 2277-2280. https://doi.org/10.1093/carcin/18.11.2277
Rooney, J., Hill, T., Qin, C., Sistare, F. D., & Corton, J. C. (2018). Adverse outcome pathway-driven identification of rat liver tumorigens in short-term assays. Toxicol Appl Pharmacol, 356, 99-113. https://doi.org/10.1016/j.taap.2018.07.023
Rosen, M. B., Das, K. P., Rooney, J., Abbott, B., Lau, C., & Corton, J. C. (2017). PPARα-independent transcriptional targets of perfluoroalkyl acids revealed by transcript profiling. Toxicology, 387, 95-107. https://doi.org/10.1016/j.tox.2017.05.013
Rosen, M. B., Lee, J. S., Ren, H., Vallanat, B., Liu, J., Waalkes, M. P., Abbott, B. D., Lau, C., & Corton, J. C. (2008). Toxicogenomic dissection of the perfluorooctanoic acid transcript profile in mouse liver: evidence for the involvement of nuclear receptors PPAR alpha and CAR. Toxicol Sci, 103(1), 46-56. https://doi.org/10.1093/toxsci/kfn025
Rosen, M. B., Schmid, J. R., Corton, J. C., Zehr, R. D., Das, K. P., Abbott, B. D., & Lau, C. (2010). Gene Expression Profiling in Wild-Type and PPARα-Null Mice Exposed to Perfluorooctane Sulfonate Reveals PPARα-Independent Effects. PPAR Res, 2010. https://doi.org/10.1155/2010/794739
Sanderson, L. M., de Groot, P. J., Hooiveld, G. J., Koppen, A., Kalkhoven, E., Müller, M., & Kersten, S. (2008). Effect of synthetic dietary triglycerides: a novel research paradigm for nutrigenomics. PLoS One, 3(2), e1681. https://doi.org/10.1371/journal.pone.0001681
Shah, Y. M., Morimura, K., Yang, Q., Tanabe, T., Takagi, M., & Gonzalez, F. J. (2007). Peroxisome proliferator-activated receptor alpha regulates a microRNA-mediated signaling cascade responsible for hepatocellular proliferation. Mol Cell Biol, 27(12), 4238-4247. https://doi.org/10.1128/MCB.00317-07
Simon, T. W., Budinsky, R. A., & Rowlands, J. C. (2015). A model for aryl hydrocarbon receptor-activated gene expression shows potency and efficacy changes and predicts squelching due to competition for transcription co-activators. PLoS One, 10(6), e0127952. https://doi.org/10.1371/journal.pone.0127952
Simon, T. W., Simons, S. S., Preston, R. J., Boobis, A. R., Cohen, S. M., Doerrer, N. G., Fenner-Crisp, P. A., McMullin, T. S., McQueen, C. A., Rowlands, J. C., & RISK21 Dose-Response Subteam. (2014). The use of mode of action information in risk assessment: Quantitative key events/dose-response framework for modeling the dose-response for key events. Crit Rev Toxicol, 44 Suppl 3, 17-43. https://doi.org/10.3109/10408444.2014.931925
Stäubli, W., Schweizer, W., Suter, J., & Weibel, E. R. (1977). The proliferative response of hepatic peroxidomes of neonatal rats to treatment with SU-13 437 (nafenopin). J Cell Biol, 74(3), 665-689. https://doi.org/10.1083/jcb.74.3.665
Stefanini, S., Nardacci, R., Farioli-Vecchioli, S., Pajalunga, D., & Sartori, C. (1999a). Liver and kidney peroxisomes in lactating rats and their pups after treatment with ciprofibrate. Biochemical and morphometric analysis. Cell Mol Biol (Noisy-le-grand), 45(6), 815-829. https://pubmed.ncbi.nlm.nih.gov/10541478
Stefanini, S., Nardacci, R., Farioli-Vecchioli, S., Pajalunga, D., & Sartori, C. (1999b). Liver and kidney peroxisomes in lactating rats and their pups after treatment with ciprofibrate. Biochemical and morphometric analysis. Cell Mol Biol (Noisy-le-grand), 45(6), 815-829. https://pubmed.ncbi.nlm.nih.gov/10541478
Svoboda, D. L., Saddler, T., & Auerbach, S. S. (2019). An Overview of National Toxicology Program’s Toxicogenomic Applications: DrugMatrix and ToxFX. In Advances in Computational Toxicology (pp. 141-157). Springer. https://link.springer.com/chapter/10.1007/978-3-030-16443-0_8
Takacs, M. L., & Abbott, B. D. (2007). Activation of mouse and human peroxisome proliferator-activated receptors (alpha, beta/delta, gamma) by perfluorooctanoic acid and perfluorooctane sulfonate. Toxicol Sci, 95(1), 108-117. https://doi.org/10.1093/toxsci/kfl135
Tanaka, K., Smith, P. F., Stromberg, P. C., Eydelloth, R. S., Herold, E. G., Grossman, S. J., Frank, J. D., Hertzog, P. R., Soper, K. A., & Keenan, K. P. (1992). Studies of early hepatocellular proliferation and peroxisomal proliferation in Sprague-Dawley rats treated with tumorigenic doses of clofibrate. Toxicol Appl Pharmacol, 116(1), 71-77. https://doi.org/10.1016/0041-008x(92)90146-j
Valles, E. G., Laughter, A. R., Dunn, C. S., Cannelle, S., Swanson, C. L., Cattley, R. C., & Corton, J. C. (2003). Role of the peroxisome proliferator-activated receptor alpha in responses to diisononyl phthalate. Toxicology, 191(2-3), 211-225. https://doi.org/10.1016/s0300-483x(03)00260-9
Wang, Y., Nakajima, T., Gonzalez, F. J., & Tanaka, N. (2020). PPARs as Metabolic Regulators in the Liver: Lessons from Liver-Specific PPAR-Null Mice. Int J Mol Sci, 21(6). https://doi.org/10.3390/ijms21062061
Watt, E. D., & Judson, R. S. (2018). Uncertainty quantification in ToxCast high throughput screening. PLoS One, 13(7), e0196963. https://doi.org/10.1371/journal.pone.0196963
Weglarz, T. C., & Sandgren, E. P. (2004). Cell cross-talk mediates PPARalpha null hepatocyte proliferation after peroxisome proliferator exposure. Carcinogenesis, 25(1), 107-112. https://doi.org/10.1093/carcin/bgg180
Weibel, E. R., Stäubli, W., Gnägi, H. R., & Hess, F. A. (1969). Correlated morphometric and biochemical studies on the liver cell. I. Morphometric model, stereologic methods, and normal morphometric data for rat liver. J Cell Biol, 42(1), 68-91. https://doi.org/10.1083/jcb.42.1.68
West, D. A., James, N. H., Cosulich, S. C., Holden, P. R., Brindle, R., Rolfe, M., & Roberts, R. A. (1999). Role for tumor necrosis factor alpha receptor 1 and interleukin-1 receptor in the suppression of mouse hepatocyte apoptosis by the peroxisome proliferator nafenopin. Hepatology, 30(6), 1417-1424. https://doi.org/10.1002/hep.510300612
Williams GM, Perrone C. (1995). Mechanism based risk assessment of peroxisome proliferating rodent hepatocarcinogens. In: Peroxisomes: Biology and Role in Toxicology and Disease, eds. J.K. Reddy, T.
Wolf, D. C., Moore, T., Abbott, B. D., Rosen, M. B., Das, K. P., Zehr, R. D., Lindstrom, A. B., Strynar, M. J., & Lau, C. (2008). Comparative hepatic effects of perfluorooctanoic acid and WY 14,643 in PPAR-alpha knockout and wild-type mice. Toxicol Pathol, 36(4), 632-639. https://doi.org/10.1177/0192623308318216
Woods, C. G., Kosyk, O., Bradford, B. U., Ross, P. K., Burns, A. M., Cunningham, M. L., Qu, P., Ibrahim, J. G., & Rusyn, I. (2007). Time course investigation of PPARalpha- and Kupffer cell-dependent effects of WY-14,643 in mouse liver using microarray gene expression. Toxicol Appl Pharmacol, 225(3), 267-277. https://doi.org/10.1016/j.taap.2007.08.028
Yamoto, T., Ohashi, Y., Furukawa, T., Teranishi, M., Manabe, S., & Makita, T. (1996). Change of the sex-dependent response to clofibrate in F344 rat liver during postnatal development. Toxicol Lett, 85(2), 77-83. https://doi.org/10.1016/0378-4274(96)03643-0
Yu, S., Cao, W. Q., Kashireddy, P., Meyer, K., Jia, Y., Hughes, D. E., Tan, Y., Feng, J., Yeldandi, A. V., Rao, M. S., Costa, R. H., Gonzalez, F. J., & Reddy, J. K. (2001). Human peroxisome proliferator-activated receptor alpha (PPARalpha) supports the induction of peroxisome proliferation in PPARalpha-deficient mouse liver. J Biol Chem, 276(45), 42485-42491. https://doi.org/10.1074/jbc.M106480200



